Abstract
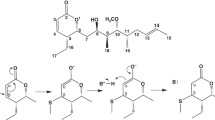
Mammalian brain tubulin is anαΒ heterodimer; bothα andΒ exist in 6–7 isotypic forms which differ in their amino acid sequences. By the use of isotype-specific monoclonal antibodies, we have previously shown that we can purify theαΒ II,αΒ III, andaΒ IV tubulin dimers from bovine brain. We have also observed that these isotypes differ in their distributionin vivo and their polymerization and drug-binding propertiesin vitro. We have now explored the question of whether the isotypically purified dimers differ in their overall conformation using as probes compounds of theN,N′-polymethylenebis (iodoacetamide) series which are known to form discrete intrachain cross-links inΒ-tubulin. These compounds have the structure ICH2CONH(CH2)nNHCOCH2I. One of these cross-links, designatedΒ s, is between cys12 and either cys201 or cys211. The other, designatedΒ *, is between cys239 and cys354. TheΒ * cross-link forms inαΒ II andαΒ IV but not inαΒ III; this is not surprising in view of the fact thatαΒ III has serine at position 239 instead of cysteine. However,αΒ III is also unable to form theΒ s cross-link, although it appears to have all three cysteines which may be involved in the cross-link. This suggests that at least one of the sulfhydryls involved in the cross-link may be inaccessible inαΒ III. Although bothαΒ II andαΒ IV can form theΒ s cross-link, the dependence on cross-linker chain length is different.αΒ II formsΒ s with derivatives in whichn=2, 4, 5, 6, and 7 but not with those in whichn=3 or 10. In contrast,αΒ IV formsΒ s with derivatives in whichn=2, 3, 4, 5, 6, 7, and 10. These results imply that theΒ s sulfhydryls are slightly more accessible inαΒ IV and are therefore less dependent on the conformation of the cross-linker to react with it. It appears, therefore, that theαΒ II,αΒ III, andαΒ IV dimers each have unique conformations. This may help to explain the different assembly and drug-binding properties of these dimers.
Similar content being viewed by others
References
Ahmad, S., Singh, B., and Gupta, R. S. (1991).Biochim. Biophys. Acta 1090, 252–254.
Banerjee, A., Jordan, M. A., Little, M., and Ludueña, R. F. (1987).Eur. J. Biochem. 165, 443–448.
Banerjee, A., and Ludueña, R. F. (1992).J. Biol. Chem. 267, 13335–13339.
Banerjee, A., Roach, M. C., Wall, K. A., Lopata, M. A., Cleveland, D. W., and Ludueña, R. F. (1988).J. Biol. Chem. 263, 3029–3034.
Banerjee, A., Roach, M. C., Trcka, P., and Ludueña, R. F. (1990).J. Biol. Chem. 265, 1794–1799.
Banerjee, A., Roach, M. C., Trcka, P., and Ludueña, R. F. (1992).J. Biol. Chem. 267, 5625–5630.
Cleveland, D. W. (1987).J. Cell Biol. 104, 381–383.
Crestfield, A. M., Moore, A., and Stein, W. H. (1963).J. Biol. Chem. 238, 622–627.
Fellous, A., FranÇon, J., Lennon, A.-M., and Nunez, J. (1977).Eur. J. Biochem. 78, 167–174.
Ikeda, Y., and Steiner, M. (1978).Biochemistry 17, 3454–3459.
Khan, I. A., and Ludueña, R. F. (1991).Biochim. Biophys. Acta 1076, 289–297.
Krauhs, E., Little, M., Kempf, T., Hofer-Warbinek, R., Ade, W., and Ponstingl, H. (1981).Proc. Nat. Acad. Sci. USA 78, 4156–4160.
Laemmli, U. K. (1970).Nature 227, 680–685.
Little, M. (1979).FEBS Letters 108, 283–286.
Little, M., and Ludueña, R. F. (1985).The EMBO Journal 4, 51–56.
Little, M., and Ludueña, R. F. (1987).Biochim. Biophys. Acta 912, 28–33.
Little, M., Krauhs, E., and Ponstingl, H. (1981).Bio Systems 14, 239–246.
Lowry, O., Rosebrough, N., Farr, A., and Randall, R. J. (1951).J. Biol. Chem. 193, 265–27.
Lu, Q., and Ludueña, R. F. (1992).Molecular Biology of the Cell 3, 168a.
Ludueña, R. F., and Roach, M. C. (1981a).Biochemistry 20, 4437–4444.
Ludueña, R. F., and Roach, M. C. (1981b).Biochemistry 20, 4444–4450.
Ludueña, R. F., and Roach, M. C. (1981c).Arch. Biochem. Biophys. 210, 498–504.
Ludueña, R. F., and Roach, M. C. (1991).Pharmac. Ther. 49, 133–152.
Ludueña, R. F., and Woodward, D. O. (1975).Ann. N.Y. Acad. Sci. 253, 272–283.
Ludueña, R. F., Shooter, E. M., and Wilson, L. (1977).J. Biol. Chem. 252, 7006–7014.
Ludueña, R. F., Roach, M. C., Trcka, P. P., Little, M., Palanivelu, P., Binkley, P., and Prasad, V. (1982).Biochemistry 21, 4787–4794.
Ludueña, R. F., Roach, M. C., Jordan, M. A., and Murphy, D. B. (1985).J. Biol. Chem. 260, 1257–1264.
Ludueña, R. F., Roach, M. C., Trcka, P. P., Mallevais, M. L. and MacRae, T. H. (1987).Arch. Biochem. Biophys. 255, 453–459.
Monteiro, M. J., and Cleveland, D. W. (1988).J. Mol. Biol. 198, 439–446.
Renthal, R., Schneider, B. G., Miller, M. M., and Ludueña, R. F. (1993).Cell Motil. Cytoskeleton 25, 19–29.
Roach, M. C., and Ludueña, R. F. (1984).J. Biol. Chem. 259, 12063–12071.
Sharma, J. S., and Ludueña, R. F., (1992).Molecular Biology of the Cell 3, 48a.
Sullivan, K. F. (1988).Ann. Rev. Cell Biol. 4, 687–716.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sharma, J., Ludueña, R.F. Use ofN,N′-polymethylenebis(iodoacetamide) derivatives as probes for the detection of conformational differences in tubulin isotypes. J Protein Chem 13, 165–176 (1994). https://doi.org/10.1007/BF01891975
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01891975