Abstract
Background
Membranous nephropathy (MN) can be idiopathic (iMN) or manifest as a result of systemic underlying conditions as a secondary epiphenomenon. For the prognostic and predictive consequences of this discrimination, the routine use of reliable markers is crucial. This large MN series aimed to evaluate the routine and standardized immunohistochemical (IHC) employment of a panel of 3 biomarkers—phospholipase A2 receptor (PLA2R), thrombospondin type-1 domain-containing 7A (THSD7A), and immunoglobulin (Ig)G4—in the differential diagnosis of MN forms, contributing to the validation of the technique and the correct interpretation of reproducible patterns of reactivity.
Methods
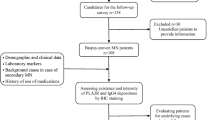
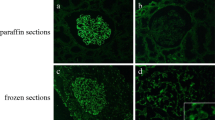
We classified 95 patients with a biopsy proven diagnosis of MN as primary (n = 72) or secondary (n = 23) cases based on clinical data. After performing an IHC assay directed against PLA2R, THSD7A and IgG4 antigens, samples were interpreted by three different nephropathologists to assess the positivity/negativity of the staining according to new interpretation criteria.
Results
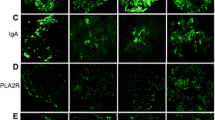
Useful interpretation criteria were introduced to exclude false positive patterns of reactivity and to identify only true granular membranous or mesangial deposits in MN. The IHC directed against PLA2R resulted positive in 51 iMN cases and negative in 21, while 4/23 secondary forms were considered positive. Based on these data the technique showed a sensitivity of 71% and specificity of 83%. On the other hand, the IHC analysis for IgG4 resulted positive in 44 cases of iMN and negative in 28 cases, while only 4/23 secondary forms were positive (same cases positive to PLA2R). Finally, THSD7A was found to be positive only in 1 case, which was negative to PLA2R and IgG4. The combination of the results allowed a classification of the series into two major groups: “double-positive” (PLA2R+/IgG4+/THSD7A−) and “triple-negative” (PLA2R−/IgG4−/THSD7A−) cases.
Conclusions
Based on these data, the diagnostic performance of the three biomarkers used in a “tandem fashion” can reach 79% sensitivity and 83% specificity, significantly reducing the risk of a false-positive or false-negative result and improving the routine characterization of this frequent glomerulonephritis.



Similar content being viewed by others
References
Hsu HC, Lin GH, Chang MH, Chen CH (1983) Association of hepatitis B surface (HBs) antigenemia and membranous nephropathy in children in Taiwan. Clin Nephrol 20:121–129
Slusarczyk J, Michalak T, Nazarewicz-de Mezer T et al (1980) Membranous glomerulopathy associated with hepatitis B core antigen immune complexes in children. Am J Pathol 98:29–43
Takekoshi Y, Tanaka M, Shida N et al (1978) Strong association between membranous nephropathy and hepatitis-B surface antigenaemia in Japanese children. Lancet 2:1065–1068
Kleinknecht C, Levy M, Gagnadoux MF, Habib R (1979) Membranous glomerulonephritis with extra-renal disorders in children. Medicine (Baltimore) 58:219–228
Ronco P, Debiec H (2014) Anti-phospholipase A2 receptor antibodies and the pathogenesis of membranous nephropathy. Nephron Clin Pract 128:232–237. https://doi.org/10.1159/000368588
Ponticelli C, Zucchelli P, Passerini P et al (1995) A 10-year follow-up of a randomized study with methylprednisolone and chlorambucil in membranous nephropathy. Kidney Int 48:1600–1604
Branten AJ, Reichert LJ, Koene RA, Wetzels JF (1998) Oral cyclophosphamide versus chlorambucil in the treatment of patients with membranous nephropathy and renal insufficiency. QJM Mon J Assoc Physicians 91:359–366
Hofstra JM, Fervenza FC, Wetzels JFM (2013) Treatment of idiopathic membranous nephropathy. Nat Rev Nephrol 9:443–458. https://doi.org/10.1038/nrneph.2013.125
Barbari A (2017) Continuing the paradigm shift in the treatment of idiopathic membranous nephropathy. Nat Rev Nephrol 13:720–720. https://doi.org/10.1038/nrneph.2017.134
Fiorentino M, Tondolo F, Bruno F et al (2016) Treatment with rituximab in idiopathic membranous nephropathy. Clin Kidney J 9:788–793. https://doi.org/10.1093/ckj/sfw091
Moroni G, Depetri F, Del Vecchio L et al (2016) Low-dose rituximab is poorly effective in patients with primary membranous nephropathy. Nephrol Dial Transplant 32:gfw251. https://doi.org/10.1093/ndt/gfw251
Matsui S, Tsuji H, Takimoto Y, Ono S (2011) Clinical improvement of membranous nephropathy after endoscopic resection of double early gastrointestinal cancers. Clin Exp Nephrol 15:285–288. https://doi.org/10.1007/s10157-010-0389-6
Zheng X-Y, Wei R-B, Tang L et al (2012) Meta-analysis of combined therapy for adult hepatitis B virus-associated glomerulonephritis. World J Gastroenterol 18:821–832. https://doi.org/10.3748/wjg.v18.i8.821
Beck LH, Bonegio RGB, Lambeau G et al (2009) M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med 361:11–21. https://doi.org/10.1056/NEJMoa0810457
Radice A, Pieruzzi F, Trezzi B et al (2017) Diagnostic specificity of autoantibodies to M-type phospholipase A2 receptor (PLA2R) in differentiating idiopathic membranous nephropathy (IMN) from secondary forms and other glomerular diseases. J Nephrol. https://doi.org/10.1007/s40620-017-0451-5
Debiec H, Ronco P (2014) Immunopathogenesis of membranous nephropathy: an update. Semin Immunopathol 36:381–397. https://doi.org/10.1007/s00281-014-0423-y
Tomas NM, Beck LH, Meyer-Schwesinger C et al (2014) Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. N Engl J Med 371:2277–2287. https://doi.org/10.1056/NEJMoa1409354
Hoxha E, Wiech T, Stahl PR et al (2016) A mechanism for cancer-associated membranous nephropathy. N Engl J Med 374:1995–1996. https://doi.org/10.1056/NEJMc1511702
Stahl PR, Hoxha E, Wiech T et al (2017) THSD7A expression in human cancer. Genes Chromosom Cancer 56:314–327. https://doi.org/10.1002/gcc.22440
Larsen CP, Messias NC, Silva FG et al (2013) Determination of primary versus secondary membranous glomerulopathy utilizing phospholipase A2 receptor staining in renal biopsies. Mod Pathol 26:709–715. https://doi.org/10.1038/modpathol.2012.207
Svobodova B, Honsova E, Ronco P et al (2013) Kidney biopsy is a sensitive tool for retrospective diagnosis of PLA2R-related membranous nephropathy. Nephrol Dial Transplant 28:1839–1844. https://doi.org/10.1093/ndt/gfs439
Hoxha E, Kneißler U, Stege G et al (2012) Enhanced expression of the M-type phospholipase A2 receptor in glomeruli correlates with serum receptor antibodies in primary membranous nephropathy. Kidney Int 82:797–804. https://doi.org/10.1038/ki.2012.209
Lefaucheur C, Stengel B, Nochy D et al (2006) Membranous nephropathy and cancer: epidemiologic evidence and determinants of high-risk cancer association. Kidney Int 70:1510–1517. https://doi.org/10.1038/sj.ki.5001790
Petri M, Orbai A-M, Alarcón GS et al (2012) Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 64:2677–2686. https://doi.org/10.1002/art.34473
Aletaha D, Neogi T, Silman AJ et al (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62:2569–2581. https://doi.org/10.1002/art.27584
Dai H, Zhang H, He Y (2015) Diagnostic accuracy of PLA2R autoantibodies and glomerular staining for the differentiation of idiopathic and secondary membranous nephropathy: an updated meta-analysis. Sci Rep 5:8803. https://doi.org/10.1038/srep08803
Hara S, Goto S, Kamiura N et al (2015) Reappraisal of PLA2R1 in membranous nephropathy: immunostaining method influence and association with IgG4-dominant phenotype. Virchows Arch An Int J Pathol. https://doi.org/10.1007/s00428-015-1754-3
Qin W, Beck LH, Zeng C et al (2011) Anti-phospholipase A2 receptor antibody in membranous nephropathy. J Am Soc Nephrol 22:1137–1143. https://doi.org/10.1681/ASN.2010090967
Xie Q, Li Y, Xue J et al (2015) Renal phospholipase A2 receptor in hepatitis B virus-associated membranous nephropathy. Am J Nephrol 41:345–353. https://doi.org/10.1159/000431331
Murtas C, Ghiggeri GM (2016) Membranous glomerulonephritis: histological and serological features to differentiate cancer-related and non-related forms. J Nephrol 29:469–478. https://doi.org/10.1007/s40620-016-0268-7
De Vriese AS, Glassock RJ, Nath KA et al (2017) A proposal for a serology-based approach to membranous nephropathy. J Am Soc Nephrol 28:421–430. https://doi.org/10.1681/ASN.2016070776
Behnert A, Fritzler MJ, Teng B et al (2013) An anti-phospholipase A2 receptor quantitative immunoassay and epitope analysis in membranous nephropathy reveals different antigenic domains of the receptor. PLoS One 8:e61669. https://doi.org/10.1371/journal.pone.0061669
Fresquet M, Jowitt TA, Gummadova J et al (2015) Identification of a major epitope recognized by PLA2R autoantibodies in primary membranous nephropathy. J Am Soc Nephrol 26:302–313. https://doi.org/10.1681/ASN.2014050502
Kao L, Lam V, Waldman M et al (2015) Identification of the immunodominant epitope region in phospholipase A2 receptor-mediating autoantibody binding in idiopathic membranous nephropathy. J Am Soc Nephrol 26:291–301. https://doi.org/10.1681/ASN.2013121315
Huang CC, Lehman A, Albawardi A et al (2013) IgG subclass staining in renal biopsies with membranous glomerulonephritis indicates subclass switch during disease progression. Mod Pathol 26:799–805. https://doi.org/10.1038/modpathol.2012.237
Larsen CP, Cossey LN, Beck LH (2016) THSD7A staining of membranous glomerulopathy in clinical practice reveals cases with dual autoantibody positivity. Mod Pathol 29:421–426. https://doi.org/10.1038/modpathol.2016.32
Polak JM, Varndell IM (1984) Immunolabeling for electron microscopy. Elsevier, Amsterdam
Kim YG, Choi Y-W, Kim S-Y et al (2015) Anti-phospholipase A2 receptor antibody as prognostic indicator in idiopathic membranous nephropathy. Am J Nephrol 42:250–257. https://doi.org/10.1159/000440983
Timmermans SAMEG., Abdul Hamid MA, Cohen Tervaert JW et al (2015) Anti-PLA2R antibodies as a prognostic factor in PLA2R-related membranous nephropathy. Am J Nephrol 42:70–77. https://doi.org/10.1159/000437236
Ramachandran R, Hn H, Kumar V et al (2015) Tacrolimus combined with corticosteroids versus modified ponticelli regimen in treatment of idiopathic membranous nephropathy: randomized control trial. Nephrology (Carlton). https://doi.org/10.1111/nep.12569
Ruggenenti P, Debiec H, Ruggiero B et al (2015) Anti-phospholipase A2 receptor antibody titer predicts post-rituximab outcome of membranous nephropathy. J Am Soc Nephrol 26:2545–2558. https://doi.org/10.1681/ASN.2014070640
Glassock RJ (1992) Secondary membranous glomerulonephritis. Nephrol Dial Transplant 7(Suppl 1):64–71
Acknowledgements
Our special thanks go to Carla Scalia, Loredana Tusa and Lorella Riva for their technical support and to nephrological team of the San Gerardo Hospital for the routine execution of the renal biopsies.
Funding
No support was used for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
L’Imperio, V., Pieruzzi, F., Sinico, R.A. et al. Routine immunohistochemical staining in membranous nephropathy: in situ detection of phospholipase A2 receptor and thrombospondin type 1 containing 7A domain. J Nephrol 31, 543–550 (2018). https://doi.org/10.1007/s40620-018-0489-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-018-0489-z