Opinion statement
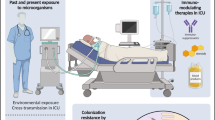
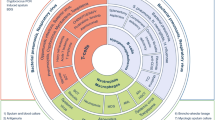
Aspergillus species are septated molds that cause a wide spectrum of clinical syndromes. Among these, invasive aspergillosis (IA) causes very high morbidity and mortality among the most severely immunosuppressed, especially those with profound qualitative or quantitative neutropenia. Empirical, pre-emptive, and targeted approaches have been attempted to blunt establishment of infection with variable success. The preferred treatment of primary IA is voriconazole, which has been found to be superior to amphotericin B. Azoles interfere with the synthesis of ergosterol found in the fungal cell membrane, whereas polyenes—such as amphotericin B—interfere with ergosterol function. An echinocandin that disrupts fungal cell wall synthesis—caspofungin—and itraconazole have been approved for salvage therapy of IA. Lipid amphotericin B formulations are used in those intolerant to amphotericin B deoxycholate. Posaconazole is used for prophylaxis in high-risk groups such as those with acute myelogenous leukemia, myelodysplastic syndrome, and stem cell transplant recipients with graft versus host disease to reduce the incidence of invasive fungal infections (IFI) such as IA. Combining mold-active azoles or a lipid amphotericin B formulation with an echinocandin may have a role in refractory IA. Immunomodulatory properties of antifungals, growth factors, cytokines, and immune cell infusions may enhance host ability to facilitate adjunctive control of infection, but an uncontrolled, exuberant inflammatory response can also cause significant pathology. Surgical resection may be a last resort when angioinvasion of critical structures places a patient at high risk for bleeding, thrombosis, or embolic phenomena, despite medical therapy. Nevertheless, immune reconstitution with myeloid lineage recovery is the key to successful outcomes.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
De Pauw B, Walsh TJ, Donnelly JP, Stevens DA, Edwards JE, et al. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis. 2008;46:1813–21. Consensus case definitions for proven or probable IA are given.
Segal BH, Herbrecht R, Stevens DA, Ostrosky-Zeichner L, Sobel J, et al. Defining responses to therapy and study outcomes in clinical trials of invasive fungal diseases: Mycoses Study Group and European Organization for Research and Treatment of Cancer consensus criteria. Clin Infect Dis. 2008;47:674–83. Consensus definitions for IFI outcome, including IA, such as complete and partial recovery are specified.
Walsh TJ, Anaissie EJ, Denning DW, Herbrecht R, Kontoyiannis DP, et al. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:327–60.Comprehensive U.S. clinical guidelines on the management of IA are provided.
Kontoyiannis DP, Marr KA, Park BJ, Alexander BD, Anaissie EJ, et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001-2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis. 2010;50:1091–100.Active surveillance of IFI among over 20 U.S. HSCT centers demonstrated IA as a leading cause of IFI in this group, occurring later after transplantation, but with high mortality despite current therapies.
Pappas PG, Alexander BD, Andes DR, Hadley S, Kauffman CA, et al. Invasive fungal infections among organ transplant recipients: results of the Transplant-Associated Infection Surveillance Network (TRANSNET). Clin Infect Dis. 2010;50:1101–11.Solid organ transplant IFI surveillance demonstrated IA to occur mostly in lung transplant recipients while IA was second in incidence only to invasive candidiasis among other organ transplant recipients.
Azie N, Neofytos D, Pfaller M, Meier-Kriesche HU, Quan SP, et al. The PATH (Prospective Antifungal Therapy) Alliance(R) registry and invasive fungal infections: update 2012. Diagn Microbiol Infect Dis. 2012;73:293–300. Another prospective surveillance cohort of HSCT and SOT IFI that corroborates findings from TRANSNET, but also comments on antifungal utilization and efficacies in these groups.
Cordonnier C, Pautas C, Maury S, Vekhoff A, Farhat H, et al. Empirical versus preemptive antifungal therapy for high-risk, febrile, neutropenic patients: a randomized, controlled trial. Clin Infect Dis. 2009;48:1042–51.
Morrissey CO, Chen SC, Sorrell TC, Milliken S, Bardy PG, et al. Galactomannan and PCR versus culture and histology for directing use of antifungal treatment for invasive aspergillosis in high-risk haematology patients: a randomised controlled trial. Lancet Infect Dis. 2013;13:519–28.
Caillot D, Bassaris H, McGeer A, Arthur C, Prentice HG, et al. Intravenous itraconazole followed by oral itraconazole in the treatment of invasive pulmonary aspergillosis in patients with hematologic malignancies, chronic granulomatous disease, or AIDS. Clin Infect Dis. 2001;33:e83–90.
Denning DW, Lee JY, Hostetler JS, Pappas P, Kauffman CA, et al. NIAID Mycoses Study Group Multicenter Trial of Oral Itraconazole Therapy for Invasive Aspergillosis. Am J Med. 1994;97:135–44.
Nucci M, Biasoli I, Akiti T, Silveira F, Solza C, et al. A double-blind, randomized, placebo-controlled trial of itraconazole capsules as antifungal prophylaxis for neutropenic patients. Clin Infect Dis. 2000;30:300–5.
Gallin JI, Alling DW, Malech HL, Wesley R, Koziol D, et al. Itraconazole to prevent fungal infections in chronic granulomatous disease. N Engl J Med. 2003;348:2416–22.
Segal BH, DeCarlo ES, Kwon-Chung KJ, Malech HL, Gallin JI, et al. Aspergillus nidulans infection in chronic granulomatous disease. Medicine (Baltimore). 1998;77:345–54.
Henriet SS, Hermans PW, Verweij PE, Simonetti E, Holland SM, et al. Human leukocytes kill Aspergillus nidulans by reactive oxygen species-independent mechanisms. Infect Immun. 2011;79:767–73.
Herbrecht R, Denning DW, Patterson TF, Bennett JE, Greene RE, et al. Voriconazole versus amphotericin B for primary therapy of invasive aspergillosis. N Engl J Med. 2002;347:408–15. A pivotal randomized controlled clinical trial that illustrated the superiority of voriconazole over amphotericin B for the treatment of IA, leading to the licensure of voriconazole for the primary treatment of IA -- the “treatment of choice.”
Patterson TF, Boucher HW, Herbrecht R, Denning DW, Lortholary O, et al. Strategy of following voriconazole versus amphotericin B therapy with other licensed antifungal therapy for primary treatment of invasive aspergillosis: impact of other therapies on outcome. Clin Infect Dis. 2005;41:1448–52.
Pasqualotto AC, Denning DW. New and emerging treatments for fungal infections. J Antimicrob Chemother. 2008;61 Suppl 1:i19–30.
Cornely OA, Maertens J, Winston DJ, Perfect J, Ullmann AJ, et al. Posaconazole vs. fluconazole or itraconazole prophylaxis in patients with neutropenia. N Engl J Med. 2007;356:348–59.
Ullmann AJ, Lipton JH, Vesole DH, Chandrasekar P, Langston A, et al. Posaconazole or fluconazole for prophylaxis in severe graft-versus-host disease. N Engl J Med. 2007;356:335–47.
Georgiadou SP, Kontoyiannis DP. The impact of azole resistance on aspergillosis guidelines. Ann N Y Acad Sci. 2012;1272:15–22.
van der Linden JW, Snelders E, Kampinga GA, Rijnders BJ, Mattsson E, et al. Clinical implications of azole resistance in Aspergillus fumigatus, The Netherlands, 2007-2009. Emerg Infect Dis. 2011;17:1846–54. Initial reports of cyp51A mutation conferred azole resistance in the Netherlands that has since had limited geographical distribution elsewhere.
Verweij PE, Snelders E, Kema GH, Mellado E, Melchers WJ. Azole resistance in Aspergillus fumigatus: a side-effect of environmental fungicide use? Lancet Infect Dis. 2009;9:789–95.
Pfaller MA, Diekema DJ, Ghannoum MA, Rex JH, Alexander BD, et al. Wild-type MIC distribution and epidemiological cutoff values for Aspergillus fumigatus and three triazoles as determined by the Clinical and Laboratory Standards Institute broth microdilution methods. J Clin Microbiol. 2009;47:3142–6.
Simitsopoulou M, Roilides E, Paliogianni F, Likartsis C, Ioannidis J, et al. Immunomodulatory effects of voriconazole on monocytes challenged with Aspergillus fumigatus: differential role of Toll-like receptors. Antimicrob Agents Chemother. 2008;52:3301–6.
Choi JH, Kwon EY, Park CM, Choi SM, Lee DG, et al. Immunomodulatory effects of antifungal agents on the response of human monocytic cells to Aspergillus fumigatus conidia. Med Mycol. 2010;48:704–9.
Gallis HA, Drew RH, Pickard WW. Amphotericin B: 30 years of clinical experience. Rev Infect Dis. 1990;12:308–29.
Falagas ME, Karageorgopoulos DE, Tansarli GS. Continuous versus conventional infusion of amphotericin B deoxycholate: a meta-analysis. PLoS One. 2013;8:e77075. A meta-analysis of the historical “gold standard” in the treatment of IA -- amphotericin B -- that suggest continuous infusion may offer benefit in terms of nephrotoxicity without compromising survival over conventional infusion methods.
Leenders AC, Daenen S, Jansen RL, Hop WC, Lowenberg B, et al. Liposomal amphotericin B compared with amphotericin B deoxycholate in the treatment of documented and suspected neutropenia-associated invasive fungal infections. Br J Haematol. 1998;103:205–12.
Linden PK, Coley K, Fontes P, Fung JJ, Kusne S. Invasive aspergillosis in liver transplant recipients: outcome comparison of therapy with amphotericin B lipid complex and a historical cohort treated with conventional amphotericin B. Clin Infect Dis. 2003;37:17–25.
Bowden R, Chandrasekar P, White MH, Li X, Pietrelli L, et al. A double-blind, randomized, controlled trial of amphotericin B colloidal dispersion versus amphotericin B for treatment of invasive aspergillosis in immunocompromised patients. Clin Infect Dis. 2002;35:359–66.
Cornely OA, Maertens J, Bresnik M, Ebrahimi R, Ullmann AJ, et al. Liposomal amphotericin B as initial therapy for invasive mold infection: a randomized trial comparing a high-loading dose regimen with standard dosing (AmBiLoad trial). Clin Infect Dis. 2007;44:1289–97.
Vogelsinger H, Weiler S, Djanani A, Kountchev J, Bellmann-Weiler R, et al. Amphotericin B tissue distribution in autopsy material after treatment with liposomal amphotericin B and amphotericin B colloidal dispersion. J Antimicrob Chemother. 2006;57:1153–60.
Clemons KV, Schwartz JA, Stevens DA. Experimental central nervous system aspergillosis therapy: efficacy, drug levels and localization, immunohistopathology, and toxicity. Antimicrob Agents Chemother. 2012;56:4439–49.
Steinbach WJ, Benjamin Jr DK, Kontoyiannis DP, Perfect JR, Lutsar I, et al. Infections due to Aspergillus terreus: a multicenter retrospective analysis of 83 cases. Clin Infect Dis. 2004;39:192–8.
Kontoyiannis DP, Lewis RE, May GS, Osherov N, Rinaldi MG. Aspergillus nidulans is frequently resistant to amphotericin B. Mycoses. 2002;45:406–7.
Panackal AA, Imhof A, Hanley EW, Marr KA. Aspergillus ustus infections among transplant recipients. Emerg Infect Dis. 2006;12:403–8.
Lewis RE, Chamilos G, Prince RA, Kontoyiannis DP. Pretreatment with empty liposomes attenuates the immunopathology of invasive pulmonary aspergillosis in corticosteroid-immunosuppressed mice. Antimicrob Agents Chemother. 2007;51:1078–81.
Bellocchio S, Gaziano R, Bozza S, Rossi G, Montagnoli C, et al. Liposomal amphotericin B activates antifungal resistance with reduced toxicity by diverting Toll-like receptor signalling from TLR-2 to TLR-4. J Antimicrob Chemother. 2005;55:214–22.
Kurtz MB, Douglas CM. Lipopeptide inhibitors of fungal glucan synthase. J Med Vet Mycol. 1997;35:79–86.
Maertens J, Raad I, Petrikkos G, Boogaerts M, Selleslag D, et al. Efficacy and safety of caspofungin for treatment of invasive aspergillosis in patients refractory to or intolerant of conventional antifungal therapy. Clin Infect Dis. 2004;39:1563–71.
Lockhart SR, Zimbeck AJ, Baddley JW, Marr KA, Andes DR, et al. In vitro echinocandin susceptibility of Aspergillus isolates from patients enrolled in the Transplant-Associated Infection Surveillance Network. Antimicrob Agents Chemother. 2011;55:3944–6.
Pfaller MA, Boyken L, Hollis RJ, Kroeger J, Messer SA, et al. In vitro susceptibility of clinical isolates of Aspergillus spp. to anidulafungin, caspofungin, and micafungin: a head-to-head comparison using the CLSI M38-A2 broth microdilution method. J Clin Microbiol. 2009;47:3323–5.
Walsh TJ, Teppler H, Donowitz GR, Maertens JA, Baden LR, et al. Caspofungin versus liposomal amphotericin B for empirical antifungal therapy in patients with persistent fever and neutropenia. N Engl J Med. 2004;351:1391–402.
van Burik JA, Ratanatharathorn V, Stepan DE, Miller CB, Lipton JH, et al. Micafungin versus fluconazole for prophylaxis against invasive fungal infections during neutropenia in patients undergoing hematopoietic stem cell transplantation. Clin Infect Dis. 2004;39:1407–16.
Kubiak DW, Bryar JM, McDonnell AM, Delgado-Flores JO, Mui E, et al. Evaluation of caspofungin or micafungin as empiric antifungal therapy in adult patients with persistent febrile neutropenia: a retrospective, observational, sequential cohort analysis. Clin Ther. 2010;32:637–48.
Seibel NL, Schwartz C, Arrieta A, Flynn P, Shad A, et al. Safety, tolerability, and pharmacokinetics of Micafungin (FK463) in febrile neutropenic pediatric patients. Antimicrob Agents Chemother. 2005;49:3317–24.
Lafaurie M, Lapalu J, Raffoux E, Breton B, Lacroix C, et al. High rate of breakthrough invasive aspergillosis among patients receiving caspofungin for persistent fever and neutropenia. Clin Microbiol Infect. 2010;16:1191–6.
Warn PA, Sharp A, Morrissey G, Denning DW. Activity of aminocandin (IP960; HMR3270) compared with amphotericin B, itraconazole, caspofungin and micafungin in neutropenic murine models of disseminated infection caused by itraconazole-susceptible and -resistant strains of Aspergillus fumigatus. Int J Antimicrob Agents. 2010;35:146–51.
Mikulska M, Viscoli C. Current role of echinocandins in the management of invasive aspergillosis. Curr Infect Dis Rep. 2011;13:517–27.
Moretti S, Bozza S, Massi-Benedetti C, Prezioso L, Rossetti E, et al. An immunomodulatory activity of micafungin in preclinical aspergillosis. J Antimicrob Chemother. 2014;69(4):1065–74.
Hohl TM, Feldmesser M, Perlin DS, Pamer EG. Caspofungin modulates inflammatory responses to Aspergillus fumigatus through stage-specific effects on fungal beta-glucan exposure. J Infect Dis. 2008;198:176–85.
Safdar A. Fungal cytoskeleton dysfunction or immune activation triggered by beta-glucan synthase inhibitors: potential mechanisms for the prolonged antifungal activity of echinocandins. Cancer. 2009;115:2812–5.
Kirkpatrick WR, Perea S, Coco BJ, Patterson TF. Efficacy of caspofungin alone and in combination with voriconazole in a Guinea pig model of invasive aspergillosis. Antimicrob Agents Chemother. 2002;46:2564–8.
Nagasaki Y, Eriguchi Y, Uchida Y, Miyake N, Maehara Y, et al. Combination therapy with micafungin and amphotericin B for invasive pulmonary aspergillosis in an immunocompromised mouse model. J Antimicrob Chemother. 2009;64:379–82.
Elefanti A, Mouton JW, Verweij PE, Tsakris A, Zerva L, et al. Amphotericin B- and voriconazole-echinocandin combinations against Aspergillus spp.: effect of serum on inhibitory and fungicidal interactions. Antimicrob Agents Chemother. 2013;57:4656–63.
Kontoyiannis DP, Hachem R, Lewis RE, Rivero GA, Torres HA, et al. Efficacy and toxicity of caspofungin in combination with liposomal amphotericin B as primary or salvage treatment of invasive aspergillosis in patients with hematologic malignancies. Cancer. 2003;98:292–9.
Marr KA, Schlamm H, Rottinghaus ST, Jagannatha S, Bow EJ, et al. A randomised, double-blind study of combination antifungal therapy with voriconazole and anidulafungin versus voriconazole monotherapy for primary treatment of invasive aspergillosis [poster no. LB2812]. Special Issue: Abstracts of the 22nd European Congress of Clinical Microbiology and Infectious Diseases, London, United Kingdom, 31 March – 3 April 2012. Clin Microbiol Infect Suppl. 2012;s3:713. Randomized clinical trial comparing voriconazole and anidulafungin to voriconazole alone for primary IA treatment that demonstrates marginal effect in the combination arm over monotherapy.
Panackal AA, Parisini E, Proschan M. Salvage Combination antifungal therapy for acute invasive aspergillosis may improve outcomes: a systematic review and meta-analysis. Manuscript in preparation. Recent meta-analysis that demonstrated improved 12-week survival and clinical response in those receiving combination therapy with a mold active triazole or a lipid amphotericin B product with an echinocandin compared with non-echinocandin monotherpay for salvage IA therapy, after restricting to high quality studies and applying a variety of sensitivity analyses. Caution is advised in interpreting these results, given the inherent limitations of meta-analysis and the changing management strategies in the affected host, which may also improve outcomes.
Roilides E, Uhlig K, Venzon D, Pizzo PA, Walsh TJ. Enhancement of oxidative response and damage caused by human neutrophils to Aspergillus fumigatus hyphae by granulocyte colony-stimulating factor and gamma interferon. Infect Immun. 1993;61:1185–93.
Sionov E, Mendlovic S, Segal E. Experimental systemic murine aspergillosis: treatment with polyene and caspofungin combination and G-CSF. J Antimicrob Chemother. 2005;56:594–7.
Graybill JR, Bocanegra R, Najvar LK, Loebenberg D, Luther MF. Granulocyte colony-stimulating factor and azole antifungal therapy in murine aspergillosis: role of immune suppression. Antimicrob Agents Chemother. 1998;42:2467–73.
Heil G, Hoelzer D, Sanz MA, Lechner K, Liu Yin JA, et al. A randomized, double-blind, placebo-controlled, phase III study of filgrastim in remission induction and consolidation therapy for adults with de novo acute myeloid leukemia. The International Acute Myeloid Leukemia Study Group. Blood. 1997;90:4710–8.
Godwin JE, Kopecky KJ, Head DR, Willman CL, Leith CP, et al. A double-blind placebo-controlled trial of granulocyte colony-stimulating factor in elderly patients with previously untreated acute myeloid leukemia: a Southwest oncology group study (9031). Blood. 1998;91:3607–15.
Lyman GH, Kleiner JM. Summary and comparison of myeloid growth factor guidelines in patients receiving cancer chemotherapy. J Natl Compr Canc Netw. 2007;5:217–28.
Dornbusch HJ, Urban CE, Pinter H, Ginter G, Fotter R, et al. Treatment of invasive pulmonary aspergillosis in severely neutropenic children with malignant disorders using liposomal amphotericin B (AmBisome), granulocyte colony-stimulating factor, and surgery: report of five cases. Pediatr Hematol Oncol. 1995;12:577–86.
Flynn TN, Kelsey SM, Hazel DL, Guest JF. Cost effectiveness of amphotericin B plus G-CSF compared with amphotericin B monotherapy: treatment of presumed deep-seated fungal infection in neutropenic patients in the UK. Pharmacoeconomics. 1999;16:543–50.
Choi JH, Brummer E, Kang YJ, Jones PP, Stevens DA. Inhibitor kappaB and nuclear factor kappaB in granulocyte-macrophage colony-stimulating factor antagonism of dexamethasone suppression of the macrophage response to Aspergillus fumigatus conidia. J Infect Dis. 2006;193:1023–8.
Roilides E, Holmes A, Blake C, Venzon D, Pizzo PA, et al. Antifungal activity of elutriated human monocytes against Aspergillus fumigatus hyphae: enhancement by granulocyte-macrophage colony-stimulating factor and interferon-gamma. J Infect Dis. 1994;170:894–9.
Safdar A, Rodriguez G, Zuniga J, Al Akhrass F, Georgescu G, et al. Granulocyte macrophage colony-stimulating factor in 66 patients with myeloid or lymphoid neoplasms and recipients of hematopoietic stem cell transplantation with invasive fungal disease. Acta Haematol. 2013;129:26–34.
A controlled trial of interferon gamma to prevent infection in chronic granulomatous disease. The International Chronic Granulomatous Disease Cooperative Study Group. N Engl J Med. 1991;324:509–16.
Safdar A, Rodriguez G, Ohmagari N, Kontoyiannis DP, Rolston KV, et al. The safety of interferon-gamma-1b therapy for invasive fungal infections after hematopoietic stem cell transplantation. Cancer. 2005;103:731–9.
Bernhisel-Broadbent J, Camargo EE, Jaffe HS, Lederman HM. Recombinant human interferon-gamma as adjunct therapy for Aspergillus infection in a patient with chronic granulomatous disease. J Infect Dis. 1991;163:908–11.
Yamashita K, Miyoshi T, Arai Y, Mizugishi K, Takaori-Kondo A, et al. Enhanced generation of reactive oxygen species by interferon-gamma may have contributed to successful treatment of invasive pulmonary aspergillosis in a patient with chronic granulomatous disease. Int J Hematol. 2013;97:505–10.
Bandera A, Trabattoni D, Ferrario G, Cesari M, Franzetti F, et al. Interferon-gamma and granulocyte-macrophage colony stimulating factor therapy in three patients with pulmonary aspergillosis. Infection. 2008;36:368–73.
Dignani MC, Anaissie EJ, Hester JP, O'Brien S, Vartivarian SE, et al. Treatment of neutropenia-related fungal infections with granulocyte colony-stimulating factor-elicited white blood cell transfusions: a pilot study. Leukemia. 1997;11:1621–30.
Safdar A, Rodriguez GH, Lichtiger B, Dickey BF, Kontoyiannis DP, et al. Recombinant interferon gamma1b immune enhancement in 20 patients with hematologic malignancies and systemic opportunistic infections treated with donor granulocyte transfusions. Cancer. 2006;106:2664–71.
Nichols WG, Price T, Boeckh M. Cytomegalovirus infections in cancer patients receiving granulocyte transfusions. Blood. 2002;99:3483–4.
Danielson C, Benjamin RJ, Mangano MM, Mills CJ, Waxman DA. Pulmonary pathology of rapidly fatal transfusion-related acute lung injury reveals minimal evidence of diffuse alveolar damage or alveolar granulocyte infiltration. Transfusion. 2008;48:2401–8.
Bozza S, Perruccio K, Montagnoli C, Gaziano R, Bellocchio S, et al. A dendritic cell vaccine against invasive aspergillosis in allogeneic hematopoietic transplantation. Blood. 2003;102:3807–14.
Shao C, Qu J, He L, Zhang Y, Wang J, et al. Dendritic cells transduced with an adenovirus vector encoding interleukin-12 are a potent vaccine for invasive pulmonary aspergillosis. Genes Immun. 2005;6:103–14.
Hot A, Mazighi M, Lecuit M, Poiree S, Viard JP, et al. Fungal internal carotid artery aneurysms: successful embolization of an Aspergillus-associated case and review. Clin Infect Dis. 2007;45:e156–61.
Khalil A, Fartoukh M, Bazot M, Parrot A, Marsault C, et al. Systemic arterial embolization in patients with hemoptysis: initial experience with ethylene vinyl alcohol copolymer in 15 cases. AJR Am J Roentgenol. 2010;194:W104–10.
Cesaro S, Pegoraro A, Tridello G, Pillon M, Cannata E, et al. The role of surgery in the treatment of invasive fungal infection in paediatric haematology patients: a retrospective single-centre survey. Mycoses. 2014. doi:10.1111/myc.12172.
Nebiker CA, Lardinois D, Junker L, Gambazzi F, Matt P, et al. Lung resection in hematologic patients with pulmonary invasive fungal disease. Chest. 2012;142:988–95.
Acknowledgments
This research was supported in part by the Intramural Research Program of the NIH/NIAID/DIR/LCID/TMU. The views herein do not reflect the official opinions of the Uniformed Services University or the Department of Defense.
Compliance with Ethics Guidelines
Conflict of Interest
Peter Williamson declares no conflicts of interest.
Anil Panackal is employed at the NIH and has stock in AMCAP.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panackal, A.A., Bennett, J.E. & Williamson, P.R. Treatment Options in Invasive Aspergillosis. Curr Treat Options Infect Dis 6, 309–325 (2014). https://doi.org/10.1007/s40506-014-0016-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-014-0016-2