Abstract
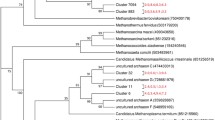
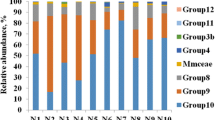
Understanding the methanogen community structure and methanogenesis from Bubalus bubalis in India may be beneficial to methane mitigation. Our current understanding of the microbial processes leading to methane production is incomplete, and further advancement in the knowledge of methanogenesis pathways would provide means to manipulate its emission in the future. In the present study, we evaluated the methanogenic community structure in the rumen as well as their potential genes involved in methanogenesis. The taxonomic and metabolic profiles of methanogens were assessed by shotgun sequencing of rumen metagenome by Ion Torrent semiconductor sequencing. The buffalo rumen contained representative genera of all the families of methanogens. Members of Methanobacteriaceae were found to be dominant, followed by Methanosarcinaceae, Methanococcaceae, Methanocorpusculaceae, and Thermococcaceae. A total of 60 methanogenic genera were detected in buffalo rumen. Methanogens related to the genera Methanobrevibacter, Methanosarcina, Methanococcus, Methanocorpusculum, Methanothermobacter, and Methanosphaera were predominant, representing >70 % of total archaeal sequences. The metagenomic dataset indicated the presence of genes involved in the methanogenesis and acetogenesis pathways, and the main functional genes were those of key enzymes in the methanogenesis. Sequences related to CoB--CoM heterodisulfide reductase, methyl coenzyme M reductase, f420-dependent methylenetetrahydromethanopterin reductase, and formylmethanofuran dehydrogenase were predominant in rumen. In addition, methenyltetrahydrofolate cyclohydrolase, methylenetetrahydrofolate dehydrogenase, 5,10-methylenetetrahydrofolate reductase, and acetyl-coenzyme A synthetase were also recovered.






Similar content being viewed by others
References
Beauchemin KA, Kreuzer M, O’Mara F, McAllister TA (2008) Nutritional management for enteric methane abatement: a review. Anim Prod Sci 48(2):21–27
Brulc JM, Antonopoulos DA, Miller ME, Wilson MK, Yannarell AC, Dinsdale EA, Edwards RE, Frank ED, Emerson JB, Wacklin P, Coutinho PM, Henrissat B, Nelson KE, White BA (2009) Gene-centric metagenomics of the fiber-adherent bovine rumen microbiome reveals forage specific glycoside hydrolases. Proc Natl Acad Sci U S A 106(6):1948–1953
Burke SA, Lo SL, Krzycki JA (1998) Clustered genes encoding the methyltransferases of methanogenesis from monomethylamine. J Bacteriol 180(13):3432–3440
Cadillo-Quiroz H, Yavitt JB, Zinder SH (2009) Methanosphaerula palustris gen. nov., sp. nov., a hydrogenotrophic methanogen isolated from a minerotrophic fen peatland. Int J Syst Evol Microbiol 59(Pt 5):928–935
Chhabra A, Manjunath KR, Panigrahy S, Parihar JS (2009) Spatial pattern of methane emissions from Indian livestock. Curr Sci 96(5):683–689
Chidthaisong A, Conrad R (2000) Turnover of glucose and acetate coupled to reduction of nitrate, ferric iron and sulfate and to methanogenesis in anoxic rice field soil. FEMS Microbiol Ecol 31(1):73–86
Denman SE, Tomkins NW, McSweeney CS (2007) Quantitation and diversity analysis of ruminal methanogenic populations in response to the antimethanogenic compound bromochloromethane. FEMS Microbiol Ecol 62(3):313–322
Dhillon A, Lever M, Lloyd KG, Albert DB, Sogin ML, Teske A (2005) Methanogen diversity evidenced by molecular characterization of methyl coenzyme M reductase A (mcrA) genes in hydrothermal sediments of the Guaymas Basin. Appl Environ Microbiol 71(8):4592–4601
DiMarco AA, Bobik TA, Wolfe RS (1990) Unusual coenzymes of methanogenesis. Annu Rev Biochem 59:355–394
Drake HL, Gössner AS, Daniel SL (2008) Old acetogens, new light. Ann N Y Acad Sci 1125:100–128
Earl J, Hall G, Pickup RW, Ritchie DA, Edwards C (2003) Analysis of methanogen diversity in a hypereutrophic lake using PCR-RFLP analysis of mcr sequences. Microb Ecol 46(2):270–278
Eggerth AH (1935) The gram-positive non-spore-bearing anaerobic bacilli of human feces. J Bacteriol 30(3):277–299
Ferry JG (1992) Methane from acetate. J Bacteriol 174(17):5489–5495
Friedrich MW (2005) Methyl-coenzyme M reductase genes: unique functional markers for methanogenic and anaerobic methane-oxidizing Archaea. Methods Enzymol 397:428–442
Gagen EJ, Denman SE, Padmanabha J, Zadbuke S, Al Jassim R, Morrison M, McSweeney CS (2010) Functional gene analysis suggests different acetogen populations in the bovine rumen and tammar wallaby forestomach. Appl Environ Microbiol 76(23):7785–7795
Garcia JL (1990) Taxonomy and ecology of methanogens. FEMS Microbiol Lett 87(3–4):297–308
Hallam SJ, Girguis PR, Preston CM, Richardson PM, DeLong EF (2003) Identification of methyl coenzyme M reductase A (mcrA) genes associated with methane-oxidizing archaea. Appl Environ Microbiol 69(9):5483–5491
Hungate RE (1969) Chapter IV A roll tube method for cultivation of strict anaerobes. Methods Microbiol 3:117–132
Jami E, Mizrahi I (2012) Composition and similarity of bovine rumen microbiota across individual animals. PLoS One 7(3):e33306
Jarvis GN, Strömpl C, Burgess DM, Skillman LC, Moore ER, Joblin KN (2000) Isolation and identification of ruminal methanogens from grazing cattle. Curr Microbiol 40(5):327–332
Joblin KN (1999) Ruminal acetogens and their potential to lower ruminant methane emissions. Crop Pasture Sci 50(8):1307–1314
Juottonen H, Galand PE, Yrjälä K (2006) Detection of methanogenic Archaea in peat: comparison of PCR primers targeting the mcrA gene. Res Microbiol 157(10):914–921
Krause L, Diaz NN, Edwards RA, Gartemann KH, Krömeke H, Neuweger H, Pühler A, Runte KJ, Schlüter A, Stoye J, Szczepanowski R, Tauch A, Goesmann A (2008) Taxonomic composition and gene content of a methane-producing microbial community isolated from a biogas reactor. J Biotechnol 136(1–2):91–101
Kröber M, Bekel T, Diaz NN, Goesmann A, Jaenicke S, Krause L, Miller D, Runte KJ, Viehöver P, Pühler A, Schlüter A (2009) Phylogenetic characterization of a biogas plant microbial community integrating clone library 16S-rDNA sequences and metagenome sequence data obtained by 454-pyrosequencing. J Biotechnol 142(1):38–49
Kumar S, Nagarajan M, Sandhu JS, Kumar N, Behl V (2007) Phylogeography and domestication of Indian river buffalo. BMC Evol Biol 7:186
Kumar S, Dagar SS, Puniya AK, Upadhyay RC (2013) Changes in methane emission, rumen fermentation in response to diet and microbial interactions. Res Vet Sci 94(2):263–268
Le Mer J, Roger P (2001) Production, oxidation, emission and consumption of methane by soils: a review. Eur J Soil Biol 37:25–50
Li ZP, Liu HL, Li GY, Bao K, Wang KY, Xu C, Yang YF, Yang FH, Wright AD (2013) Molecular diversity of rumen bacterial communities from tannin-rich and fiber-rich forage fed domestic Sika deer (Cervus nippon) in China. BMC Microbiol 13:151
Liu Y, Whitman WB (2008) Metabolic, phylogenetic, and ecological diversity of the methanogenic archaea. Ann N Y Acad Sci 1125:171–189
López C, Fernández PV, Manso RW, Wallo A, Guevara AV, García D Puente ME, Ramos D, García AM, Díaz BR, Alea JJ, Pérez D, López I, Jam A, Maestrey A, Pena J, Martínez MO (1999) República de Cuba. Inventario Nacional de Emisiones y Absorciones de Gases de Invernadero. Año 1990. CITMA/AMA/Instituto de Meteorología, - CC-TRAIN, La Habana, 401 pp
Luton PE, Wayne JM, Sharp RJ, Riley PW (2002) The mcrA gene as an alternative to 16S rRNA in the phylogenetic analysis of methanogen populations in landfill. Microbiology 148(Pt 11):3521–3530
Lwin KO, Kondo M, Ban-Tokuda T, Lapitan RM, Del-Barrio AN, Fujihara T, Matsui H (2012) Ruminal fermentation and microbial ecology of buffaloes and cattle fed the same diet. Anim Sci J 83(12):767–776
Meyer F, Paarmann D, D’Souza M, Olson R, Glass EM, Kubal M, Paczian T, Rodriguez A, Stevens R, Wilke A, Wilkening J, Edwards RA (2008) The metagenomics RAST server—a public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinformatics 9:386
Miron J, Ben-Ghedalia D, Morrison M (2001) Invited review: adhesion mechanisms of rumen cellulolytic bacteria. J Dairy Sci 84(6):1294–1309
Nelson MC, Morrison M, Yu Z (2011) A meta-analysis of the microbial diversity observed in anaerobic digesters. Bioresour Technol 102(4):3730–3739
Parks DH, Tyson GW, Hugenholtz P, Beiko RG (2014) STAMP: statistical analysis of taxonomic and functional profiles. Bioinformatics 30(21):3123–3124
Pope PB, Mackenzie AK, Gregor I, Smith W, Sundset MA, McHardy AC, Morrison M, Eijsink VG (2012) Metagenomics of the Svalbard reindeer rumen microbiome reveals abundance of polysaccharide utilization loci. PLoS One 7(6):e38571
Purushe J, Fouts DE, Morrison M, White BA, Mackie RI; North American Consortium for Rumen Bacteria, Coutinho PM, Henrissat B, Nelson KE (2010) Comparative genome analysis of Prevotella ruminicola and Prevotella bryantii: insights into their environmental niche. Microb Ecol 60(4):721–729
Schmieder R, Edwards R (2011) Quality control and preprocessing of metagenomic datasets. Bioinformatics 27(6):863–864
Shima S, Warkentin E, Thauer RK, Ermler U (2002) Structure and function of enzymes involved in the methanogenic pathway utilizing carbon dioxide and molecular hydrogen. J Biosci Bioeng 93(6):519–530
Singh KM, Pandya PR, Parnerkar S, Tripathi AK, Ramani U, Koringa PG, Rank DN, Joshi CG, Kothari RK (2010) Methanogenic diversity studies within the rumen of Surti buffaloes based on methyl coenzyme M reductase A (mcrA) genes point to Methanobacteriales. Pol J Microbiol 59(3):175–178
Singh KM, Tripathi AK, Pandya PR, Parnerkar S, Rank DN, Kothari RK, Joshi CG (2012) Methanogen diversity in the rumen of Indian Surti buffalo (Bubalus bubalis), assessed by 16S rDNA analysis. Res Vet Sci 92(3):451–455
Singh KM, Tripathi AK, Pandya PR, Parnerkar S, Kothari RK, Joshi CG (2013a) Molecular genetic diversity and quantitation of methanogen in ruminal fluid of buffalo (Bubalus bubalis) fed ration (wheat straw and concentrate mixture diet). Genet Res Int 2013:980191
Singh KM, Tripathi AK, Pandya PR, Parnerkar S, Rank DN, Kothari RK, Joshi CG (2013b) Use of real-time PCR technique in determination of major fibrolytic and non fibrolytic bacteria present in Indian Surti buffaloes (Bubalus bubalis). Pol J Microbiol 62(2):195–200
Steinfeld H, Gerber P, Wassenaar T, Castel V, Rosales M, de Haan C (2006) Livestock’s long shadow: environmental issues and options. Food and Agriculture Organization of the United Nations (FAO)
Stevenson DM, Weimer PJ (2007) Dominance of Prevotella and low abundance of classical ruminal bacterial species in the bovine rumen revealed by relative quantification real-time PCR. Appl Microbiol Biotechnol 75(1):165–174
Thauer RK, Kaster AK, Seedorf H, Buckel W, Hedderich R (2008) Methanogenic archaea: ecologically relevant differences in energy conservation. Nat Rev Microbiol 6(8):579–591
Wedlock DN, Janssen PH, Leahy SC, Shu D, Buddle BM (2013) Progress in the development of vaccines against rumen methanogens. Anim 7(Suppl 2):244–252
White D, Drummond JT, Fuqua C (2011) The physiology and biochemistry of prokaryotes, 4th edn. Oxford University Press, Oxford
Wilke A, Harrison T, Wilkening J, Field D, Glass EM, Kyrpides N, Mavrommatis K, Meyer F (2012) The M5nr: a novel non-redundant database containing protein sequences and annotations from multiple sources and associated tools. BMC Bioinformatics 13:141
Woese CR, Kandler O, Wheelis ML (1990) Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya. Proc Natl Acad Sci U S A 87(12):4576–4579
Wright ADG, Klieve AV (2011) Does the complexity of the rumen microbial ecology preclude methane mitigation? Anim Feed Sci Technol 166–167:248–253
Wright AD, Williams AJ, Winder B, Christophersen CT, Rodgers SL, Smith KD (2004) Molecular diversity of rumen methanogens from sheep in Western Australia. Appl Environ Microbiol 70(3):1263–1270
Acknowledgments
The work was supported by the Niche Area of Excellence project funded by the Indian Council of Agricultural Research, New Delhi. The authors are thankful to the support staff at Livestock Research Station, Sardarkrushinagar Dantiwada Agricultural University for the care and handling of buffaloes and assisting in sample collection.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by: Agnieszka Szalewska-Palasz
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 89 kb)
Rights and permissions
About this article
Cite this article
Singh, K.M., Patel, A.K., Shah, R.K. et al. Potential functional gene diversity involved in methanogenesis and methanogenic community structure in Indian buffalo (Bubalus bubalis) rumen. J Appl Genetics 56, 411–426 (2015). https://doi.org/10.1007/s13353-015-0270-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-015-0270-0