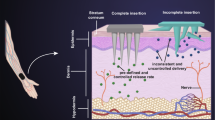
Abstract
Adapalene (ADP), a topically administered antiacne drug, finds limitation due to poor penetration, limited localization, and associated incompatibility of photosensitization and skin irritation. To explicate an innovative and safe method for ADP administration and alleviating the associated limitations, solid lipid nanoparticles (SLN) of ADP have been fabricated and evaluated for efficacy in the present work. The SLN were prepared using pre-emulsion sonication method and incorporated into convenient topical dosage form, hydrogels. In vitro permeation studies of the hydrogels through HCS indicated gel containing ADP-SLN showed 2-fold more accumulation in skin layers as compared to conventional ADP gel. Rheological studies demonstrated ADP-SLN gel to possess pseudoplastic behavior, occlusion and hydration studies revealed permeation effectiveness of ADP-SLN gel over conventional ADP gel while primary skin irritation studies established safety of the ADP-SLN gel upon topical application. Hence, it was concluded that the studied ADP-SLN formulation with skin localizing ability may be a promising carrier for topical delivery of ADP.






Similar content being viewed by others
Abbreviations
- ADP:
-
Adapalene
- SLN:
-
Solid lipid nanoparticles
- ADP-SLN:
-
Adapalene-loaded solid lipid nanoparticles
- GMS:
-
Glyceryl monosterate
- PXRD:
-
Powder X-ray diffraction
- FTIR:
-
Fourier transform infrared
- HCS:
-
Human cadaver skin
- PS:
-
Particle size
- PDI:
-
Polydispersity index
- EE:
-
Entrapment efficiency
- RSM:
-
Response surface methodology
- TEWL:
-
Trans-epidermal water loss
References
Irby CE, Yentzer BA, Feldman SR. A review of ADP in the treatment of acne vulgaris. Drugs. 2004;43:421–4.
Kawashima M, Harada S, Loesche C, Miyachi Y. ADP gel 0.1% is effective and safe for Japanese patients with acne vulgaris: a randomized, multicenter, investigator-blinded, controlled study. J Dermatol Sci. 2008;49:241–8.
Sato T, Akimoto N, Kitamura K, Kurihara H, Hayashi N, Ito A. ADP suppresses sebum accumulation via the inhibition of triacylglycerol biosynthesis and perilipin expression in differentiated hamster sebocytes in-vitro. J Dermatol Sci. 2013;70:204–10.
Piskin S, Uzunali E. A review of the use of adapalene for the treatment of acne vulgaris. Ther Clin Risk Manag. 2007;3:621–4.
Muller RH, Mader K, Gohla S. Solid lipid nanoparticles (SLN) for controlled drug delivery—a review of the state of the art. Eur J Pharm Biopharm. 2000;50:161–78.
Santos MC, Mehnert W, Schaller M, Korting HC, Gysler A, Haberland A, et al. Drug targeting by solid lipid nanoparticles for dermal use. J Drug Target. 2002;10:489–95.
Shah KA, Date AA, Joshi MD, Patravale VB. Solid lipid nanoparticle of tretinoin: potential in topical delivery. Int J Pharm. 2007;345:163–71.
Leyden JJ, Shalita A, Thiboutot D, Washenik K, Webster G. Topical retinoids in inflammatory acne: a retrospective, investigator-blinded, vehicle-controlled, photographic assessment. Clin Ther. 2005;27:216–24.
Jenning V, Gysler A, Schafer-Korting M, Gohla SH. Vitamin A loaded solid lipid nanoparticles for topical use: occlusive properties and drug targeting to the upper skin. Eur J Pharm Biopharm. 2003;49:211–8.
Vyas SP, Khar RK. Nanoparticles. In: Vyas SP, Khar RK, editors. Targeted and controlled drug delivery-noval carrier systems. New Delhi: CBS; 2002. p. 331–8.
Wissing SA, Muller RH. The influence of solid lipid nanoparticles on skin hydration and viscoelasticity-in vivo study. Eur J Pharm Biopharm. 2003;56:67–72.
Maia CS, Mehnert W, Schafer-Korting M. Solid lipid nanoparticles as drug carriers for topical glucocorticoids. Int J Pharm. 2000;96:165–7.
Muller RH, Wissing SA, Souto EB, Barbosa CM. Development of a controlled release formulation based on SLN and NLC for topical clotrimazole delivery. Int J Pharm. 2004;278:71–7.
Jensen LB, Petersson K, Neilson HM. In-vitro penetration properties of solid lipid nanoparticles in intact and barrier-impaired skin. Eur J Pharm Biopharm. 2008;79:68–75.
Patel NA, Patel NJ, Patel RP. Formulation and evaluation of curcumin gel for topical evaluation. Pharm Dev Technol. 2009;14:83–92.
Bhalekar MR, Pokharkar V, Madgulkar A, Patil N, Patil N. Preparation and evaluation of miconazole nitrate-loaded solid lipid nanoparticles for topical delivery. AAPS Pharm Sci Technol. 2009;10(1):289–96.
Bhalekar MR, Upadhaya PG, Nalawade SD, Madgulkar AR, Kshirsagar SJ. Anti-rheumatic activity of Chloroquine-SLN gel on wistar rats using complete Freund’s adjuvant (CFA) model. Ind j rheumatol. 2015 (in press).
Muller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54:131–55.
Gohla S, Mader K, Muller RH. Solid lipid nanoparticles (SLN) for controlled drug delivery—a review of the state of the art. Eur J Pharm Biopharm. 2000;50:161–77.
Solanki AB, Parikh JR, Parikh RH. Formulation and optimization of piroxicam proniosomes by 3-factor, 3-level Box-Behnken design. AAPS Pharm Sci Technol. 2007;8:1–7.
Pachuau L, Mazumder B. A study on the effects of different surfactants on ethylcellulose microspheres. Int J PharmTechnol Res. 2009;1:966–71.
Gadhiri M, Vatanara A. Loading hydrophilic drug in solid lipid media as nanoparticles: statistical modeling of entrapment efficiency and particle size. Int J Pharm. 2012;424:128–37.
Yuan H, Huang LF. Solid lipid nanoparticles prepared by solvent diffusion method in a nanoreactor system. Colloids Surfaces B. 2008;61:132–7.
Wissing S, Muller RH. Solid lipid nanoparticles (SLN)—a novel carrier for UV-blockers, die pharmazie. 2001;56:783–786.
Wissing SA, Muller RH. The influence of the crystallinity of lipid nanoparticles on their occlusive properties. Int J Pharm. 2002;242:377–9.
Muller RH, Hommoss A, Pardieke J. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int J Pharm. 2009;366:170–84.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no declarations of interest.
Rights and permissions
About this article
Cite this article
Bhalekar, M., Upadhaya, P. & Madgulkar, A. Formulation and evaluation of Adapalene-loaded nanoparticulates for epidermal localization. Drug Deliv. and Transl. Res. 5, 585–595 (2015). https://doi.org/10.1007/s13346-015-0261-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-015-0261-z