Abstract
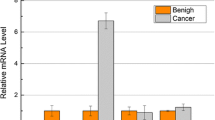
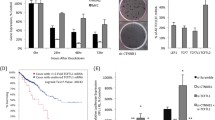
The LHX genes play an important role in a number of developmental processes. Potential roles of LHXs have been demonstrated in various neoplastic tissues as tumor suppressors or promoters depending on tumor status and types. The aim of this study was to investigate the function role of LHXs in the human colorectal cancer (CRC). The gene expression changes of LHXs in CRC tissues compared with noncancerous colorectal tissues was detected using real-time quantitative reverse transcriptase-polymerase chain reaction (QRT-PCR) analysis and immunohositochemistry. And we identified the gene LHX4 that were significantly upregulated in CRC by QRT-PCR analysis and immunohistochemistry. Furthermore, we discovered that LHX4 promoted cancer cell proliferation in vitro, and LHX4 expression correlated with elevated β-catenin levels in CRC and β-catenin function was required for LHX4’s oncogenic effects. Mechanistically, LHX4 facilitate TCF4 to bind to β-catenin and form a stable LHX4/TCF4/β-catenin complex and transactive its downstream target gene. LHX4 mutations that disrupt the LHX4-β-catenin interaction partially prevent its function in tumor cells. All in all, LHX4 is a commonly activated tumor promoter that activate Wnt/β-catenin signaling in cancer cells of CRC.


Similar content being viewed by others
References
Perencevich M, Stoffel EM. A multidisciplinary approach to the diagnosis and management of multiple colorectal polyps. Gastroenterol Hepatol. 2011;7:420.
Fearon ER. Molecular genetics of colorectal cancer. Annu Rev Pathol Mech Dis. 2011;6:479–507.
Gerstung M, Eriksson N, Lin J, Vogelstein B, Beerenwinkel N. The temporal order of genetic and pathway alterations in tumorigenesis. PLoS One. 2011;6:e27136.
Boland CR, Goel A. Microsatellite instability in colorectal cancer. Gastroenterology. 2010;138:2073–87. e2073.
Thompson MD, Monga SP. WNT/beta-catenin signaling in liver health and disease. Hepatology. 2007;45:1298–305.
Klaus A, Birchmeier W. Wnt signalling and its impact on development and cancer. Nat Rev Cancer. 2008;8:387–98.
Pevny LH, Lovell-Badge R. < i> Sox</i>genes find their feet. Curr Opin Genet Dev. 1997;7:338–44.
Kormish JD, Sinner D, Zorn AM. Interactions between SOX factors and Wnt/beta-catenin signaling in development and disease. Dev Dyn. 2010;239:56–68.
Kiefer JC. Back to basics: sox genes. Dev Dyn. 2007;236:2356–66.
Bafico A, Liu G, Goldin L, Harris V, Aaronson SA. An autocrine mechanism for constitutive Wnt pathway activation in human cancer cells. Cancer Cell. 2004;6:497–506.
Liu F, Millar SE. Wnt/beta-catenin signaling in oral tissue development and disease. J Dent Res. 2010;89:318–30.
Franklin MC, Carey KD, Vajdos FF, Leahy DJ, de Vos AM, Sliwkowski MX. Insights into ErbB signaling from the structure of the ErbB2-pertuzumab complex. Cancer Cell. 2004;5:317–28.
Agus DB, Akita RW, Fox WD, Lewis GD, Higgins B, Pisacane PI, et al. Targeting ligand-activated ErbB2 signaling inhibits breast and prostate tumor growth. Cancer Cell. 2002;2:127–37.
Muthuswamy SK, Gilman M, Brugge JS. Controlled dimerization of ErbB receptors provides evidence for differential signaling by homo- and heterodimers. Mol Cell Biol. 1999;19:6845–57.
Acknowledgments
We thank Dr. Yumin Chen, of Institute of Shanghai Springermedia, for the critical review of the manuscript. This work was supported by AfterTumor.com Grant No. CN13424V
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Nier Cha, Wei Liu, and Na Yang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cha, N., Liu, W., Yang, N. et al. Oncogenicity of LHX4 in colorectal cancer through Wnt/β-catenin/TCF4 cascade. Tumor Biol. 35, 10319–10324 (2014). https://doi.org/10.1007/s13277-014-2210-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2210-8