Abstract
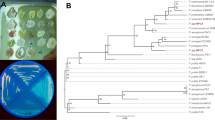
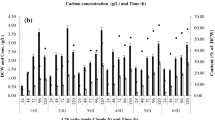
A total of 20 different strains were isolated, purified and screened for polyhydroxyalkanoate (PHA) production. PHA-producing strains were screened by Nile blue staining and confirmed by Sudan Black B staining. Strain 1.1 was selected for further analysis due to its high PHA production ability. PHA production was optimized and time profiling was calculated. PHA production on various different cheap carbon sources, i.e., sugar industry waste (fermented mash, molasses, spent wash) and corn oil, was compared. Cell dry weight and PHA content (%) were calculated and compared. The 12.53 g/L is the CDW of bacterial strain when grown in medium containing corn oil. It was found that corn oil at 12.53 g/L medium can serve as a carbon source for bacterial growth, allowing cells to accumulate PHA up to 35.63 %. The PhaC gene was amplified to confirm the genetic basis for the production of PHAs. Moreover, 16S rRNA gene sequence analysis showed that strain 1.1 belongs to Pseudomonas species.







Similar content being viewed by others
Abbreviations
- PHA:
-
Polyhydroxyalkanoate
- PDA:
-
PHA detection agar
References
Akaraonye E, Keshavarz T, Roy I (2010) Production of polyhydroxyalkanoates: the future green materials of choice. J Chem Technol Biotechnol 85:732–743
Albuquerque MGE, Eiroa M, Torres C, Nunes BR, Reis MAM (2007) Strategies for the development of a side stream process for polyhydroxyalkanoate (PHA) production from sugar cane molasses. J Biotechnol 130:411–421
Anderson AJ, Dawes EA (1990) Occurrence, metabolism, metabolic role, and industrial uses of bacterial polyhydroxyalkanoates. Microbiol Rev 54:450–472
Antonio RV, Steinbuchel A, Rehm BHA (2000) Analysis of in vivo substrate specificity of PHA synthase from Ralstonia eutropha: formation of novel copolyesters in recombinant Eschericia coli. FEMS Microbiol Lett 182:111–117
Beccari M, Bertin L, Dionisi D, Fava F, Lampis S, Majone M, Valentino F, Vallini G, Villano M (2009) Exploiting olive oil mill effluents as a renewable resource for production of biodegradable polymers through a combined anaerobic–aerobic process. J Chem Technol Biotechnol 84:901–908
Bengtsson S, Werker A, Christensson M, Welander T (2008) Production of polyhydroxyalkanoates by activated sludge treating a paper mill wastewater. Bioresour Technol 99:509–516
Bucci DZ, Tavares LBB, Sell I (2005) PHB packaging for storage of food products. Polym Test 24:564–571
Chen GQ (2009) A microbial polyhydroxyalkanoates (PHA) based bio- and materials industry. Chem Soc Rev 38:2434–2446
Chen GQ, Zhang G, Park SJ, Lee SY (2001) Industrial scale production of poly (3-hydroxybutyrate-co-3-hydroxyhexanoate). Appl Microbiol Biotechnol 57:50–55
Fukui T, Doi Y (1998) Efficient production of polyhydroxyalkanoates from plant oils by Alcaligenes eutrophus and its recombinant strain. Appl Microbiol Biotechnol 49:333–336
Gross RA, Kalra B (2002) Biodegradable polymers for the environment. Science 297:803–807
Guillet J (2004) Plastics and environment. In: Scott G (ed) Degradable polymers: principles and applications, 2nd edn. Kluwer, New York, pp 1254–1263
Ha CS, Cho WJ (2002) Miscibility, properties and biodegradability of microbial polyester containing blends. Prog Polym Sci 27:759–809
Hu W, Wang Y, Hua F, Chua H, Sin S, Yu H (2006) Synthesis of poly-hydroxyalkanoates from activated sludge under various oxidation-reduction potentials. Ann Microbiol 56:257–260
Jendrossek D (2001) Microbial degradation of polyesters. Adv Biochem Eng Biotechnol 71:293–325
Jiang Y, Song X, Gong L, Li P, Dai C, Shao W (2008) High poly (bhydroxybutyrate) production by Pseudomonas fluorescens A2a5 from inexpensive substrates. Enzyme Microb Technol 42:167–172
Khardenavis AA, Vaidya AN, Kumar MS, Chakrabarti T (2009) Utilization of molasses spentwash for production of bioplastics by waste activated sludge. Waste Manage 29:2558–2565
Kim BS (2000) Production of poly (3-hydroxybutyrate) from inexpensive substrates. Enzyme Microb Technol 27:774–777
Koller M, Bona R, Braunegg G, Hermann C, Horvat P, Kroutil M, Martinz J, Neto J, Pereira L, Varila P (2005) Production of polyhydroxyalkanoates from agricultural waste and surplus materials. Biomacromolecules 6:561–565
Kung SS, Chuang YC, Chen CH, Chien CC (2007) Isolation of polyhydroxyalkanoates-producing bacteria using a combination of phenotypic and genotypic approach. Lett Appl Microbiol 44:364–371
Lee SY, Choi JI (2004) Polyhydroxyalkanoates: biodegradeable polymer. In: Demain AL, Davies JE, Atlas RM, Cohen G, Hershberger CL, Hu WS, Sherman DH, Willson RC, David JH (eds) Manual of indusrial microbiology and biotechnology, 2nd edn. American Society of Microbiology, Washington, pp 616–624
Marsudi S, Unno H, Hori K (2008) Palm oil utilization for the simultaneous production of polyhydroxyalkanoates and rhamnolipids by Pseudomonas aeruginosa. Appl Microbiol Biotechnol 78:955–961
Mazur LP, da Silva DD, Grigull VH, Garcia MCF, Magalhães TO, Wagner TM, Einloft S, Dullius J, Schneider AL, Pezzin APT (2009) Strategies of biosynthesis of poly (3-hydroxybutyrate) supplemented with biodiesel obtained from rice bran oil. Mater Sci Eng C 29:583–587
Page WJ (1989) Production of poly-β-hydroxybutyrate by Azotobacter vinelandii strain UWD during growth on molasses and other complex carbon sources. Appl Microbiol Biotechnol 31:329–333
Ramsay JA, Berger E, Ramsay BA, Chavarie C (1990) Recovery of poly-3-hydroxyalkanoic acid granules by a surfactant hypochlorite treatment. Biotechnol Tech 4:221–226
Rehman S, Jamil N, Husnain S (2007) Screening of different contaminated environments for polyhydroxyalkanoates-producing bacterial strains. Bioloogia 62:650–656
Rhu D, Lee W, Kim J, Choi E (2003) Polyhydroxyalkanoate (PHA) production from waste. Water Sci Technol 48:221–228
Shang LG, Jiang M, Yun Z, Yan HQ, Chang HN (2008) Mass production of medium-chain-length poly(3-hydroxyalkanoates) from hydrolyzed corn oil by fed-batch culture of Pseudomonas putida. World J Microbiol Biotechnol 24:2783–2787
Solaiman DKY, Ashby RD, Foglia TA (2000) Rapid and specific identification of medium-chain length polyhydroxyalkanoate synthase gene by polymerase chain reaction. Appl Microbiol Biotechnol 53:690–694
Spiekermann P, Rehm BHA, Kalscheuer R, Baumeister D, Steinbuchel A (1999) A sensitive, viable-colony staining method using Nile red for direct screening of bacteria that accumulate polyhydroxyalkanoic acids and other lipid storage compounds. Arch Microbiol 17:73–80
Tsuge T (2002) Metabolic improvements and use of inexpensive carbon sources in microbial production of polyhydroxyalkanoates. J Biosci Bioeng 94:579–584
Acknowledgment
We are grateful to the Higher Education Commission (HEC), Pakistan, for providing grants under the Presidential Young Innovator research project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaudhry, W.N., Jamil, N., Ali, I. et al. Screening for polyhydroxyalkanoate (PHA)-producing bacterial strains and comparison of PHA production from various inexpensive carbon sources. Ann Microbiol 61, 623–629 (2011). https://doi.org/10.1007/s13213-010-0181-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-010-0181-6