Abstract
Academic-industry collaborations are an emerging format of translational stroke research. Next to classic contract research models, a multitude of collaboration models has been developed, some of which even allowing for multinational or intercontinental research programs. This development has recently been paralleled by first successful attempts to overcome the translational stroke research road block, such as the unprecedented success of novel endovascular approaches or the advent of the multicenter preclinical trial concept. While the first underlines the role of the industry as a major innovation driver in stroke research, the latter will require enrollment of industrial partners for optimal output. Moreover, academic-industry partnerships are invaluable to bridge the translational “valley of death” as well as funding gaps in times of dwindling public funding and declining high risk capital investments. However, these collaborations are also subject to relevant challenges because interests, values, and aims often significantly differ between cademia and industry. Here, we describe common academic-industry collaboration models as well as associated benefits and challenges in the stroke research arena. We also suggest strategies for improved planning, implementation, guidance, and utilization of academic-industry collaborations to the maximum mutual benefit.

Similar content being viewed by others
References
George PM, Steinberg GK. Novel stroke therapeutics: unraveling stroke pathophysiology and its impact on clinical treatments. Neuron. 2015;87(2):297–309.
Feigin VL, Forouzanfar MH, Krishnamurthi R, Mensah GA, Connor M, Bennett DA, et al. Global and regional burden of stroke during 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet. 2014;383(9913):245–54.
Matchar DB, Nguyen HV, Tian Y. Bundled payment and care of acute stroke: what does it take to make it work? Stroke. 2015;46(5):1414–21.
Auriel E, Bornstein NM. Neuroprotection in acute ischemic stroke—current status. J Cell Mol Med. 2010;14(9):2200–2.
Gladstone DJ, Black SE, Hakim AM. Heart and Stroke Foundation of Ontario Centre of Excellence in stroke recovery. Toward wisdom from failure: lessons from neuroprotective stroke trials and new therapeutic directions. Stroke. 2002;33(8):2123–36.
Pendlebury ST. Worldwide under-funding of stroke research. Int J Stroke. 2007;2(2):80–4.
Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015;372(24):2296–306.
Berkhemer OA, Fransen PS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015;372(1):11–20.
Campbell BC, Donnan GA, Lees KR, Hacke W, Khatri P, Hill MD, et al. Endovascular stent thrombectomy: the new standard of care for large vessel ischaemic stroke. Lancet Neurol. 2015;14(8):846–54.
Amar AP, Griffin JH, Zlokovic BV. Combined neurothrombectomy or thrombolysis with adjunctive delivery of 3K3A-activated protein C in acute ischemic stroke. Front Cell Neurosci. 2015;9:344.
Martin JB. Academic-industrial collaboration: the good, the bad, and the ugly. Trans Am Clin Climatol Assoc. 2002;113:227–40.
Borsody MK, Yamada C, Bielawski D, Heaton T, Castro Prado F, et al. Effects of noninvasive facial nerve stimulation in the dog middle cerebral artery occlusion model of ischemic stroke. Stroke. 2014;45(4):1102–7.
Macleod MR, van der Worp HB, Sena ES, Howells DW, Dirnagl U, Donnan GA. Evidence for the efficacy of NXY-059 in experimental focal cerebral ischaemia is confounded by study quality. Stroke. 2008;39(10):2824–9.
Boltze J, Lukomska B, Jolkkonen J. MEMS–IRBI consortium. Mesenchymal stromal cells in stroke: improvement of motor recovery or functional compensation? J Cereb Blood Flow Metab. 2014;34(8):1420–1.
Hess DC, Sila CA, Furlan AJ, Wechsler LR, Switzer JA, Mays RW. A double-blind placebo-controlled clinical evaluation of MultiStem for the treatment of ischemic stroke. Int J Stroke. 2014;9(3):381–6.
Terpolilli NA, Kim SW, Thal SC, Kataoka H, Zeisig V, Nitzsche B, et al. Inhalation of nitric oxide prevents ischemic brain damage in experimental stroke by selective dilatation of collateral arterioles. Circ Res. 2012;110(5):727–38.
Borsody MK, Yamada C, Bielawski D, Heaton T, Lyeth B, Garcia A, et al. Effect of pulsed magnetic stimulation of the facial nerve on cerebral blood flow. Brain Res. 2013;1528:58–67.
Yoo AJ, Verduzco LA, Schaefer PW, Hirsch JA, Rabinov JD, González RG. MRI-based selection for intra-arterial stroke therapy: value of pretreatment diffusion-weighted imaging lesion volume in selecting patients with acute stroke who will benefit from early recanalization. Stroke. 2009;40(6):2046–54.
Campbell BC, Purushotham A, Christensen S, Desmond PM, Nagakane Y, Parsons MW, et al. The infarct core is well represented by the acute diffusion lesion: sustained reversal is infrequent. J Cereb Blood Flow Metab. 2012;32(1):50–6.
Wheeler HM, Mlynash M, Inoue M, Tipirneni A, Liggins J, Zaharchuk G, et al. Early diffusion-weighted imaging and perfusion-weighted imaging lesion volumes forecast final infarct size in DEFUSE 2. Stroke. 2013;44(3):681–5.
Ding G, Zhang Z, Chopp M, Li L, Zhang L, Li Q, et al. MRI evaluation of BBB disruption after adjuvant AcSDKP treatment of stroke with tPA in rat. Neuroscience. 2014;271:1–8.
Arnberg F, Grafstrom J, Lundberg J, Nikkhou-Aski S, Little P, Damberg P, et al. Imaging of a clinically relevant stroke model: glucose hypermetabolism revisited. Stroke. 2015;46(3):835–42.
Ay I, Blasi F, Rietz TA, Rotile NJ, Kura S, Brownell AL, et al. In vivo molecular imaging of thrombosis and thrombolysis using a fibrin-binding positron emission tomographic probe. Circ Cardiovasc Imaging. 2014;7(4):697–705.
Weber RA, Hui ES, Jensen JH, Nie X, Falangola MF, Helpern JA, et al. Diffusional kurtosis and diffusion tensor imaging reveal different time-sensitive stroke-induced microstructural changes. Stroke. 2015;46(2):545–50.
Endres M, Engelhardt B, Koistinaho J, Lindvall O, Meairs S, Mohr JP, et al. Improving outcome after stroke: overcoming the translational roadblock. Cerebrovasc Dis. 2008;25(3):268–78.
Kimmelman J, Mogil JS, Dirnagl U. Distinguishing between exploratory and confirmatory preclinical research will improve translation. PLoS Biol. 2014;12(5), e1001863.
Dirnagl U, Hakim A, Macleod M, Fisher M, Howells D, Alan SM, et al. A concerted appeal for international cooperation in preclinical stroke research. Stroke. 2013;44(6):1754–60.
Dirnagl U, Fisher M. International, multicenter randomized preclinical trials in translational stroke research: it’s time to act. J Cereb Blood Flow Metab. 2012;32(6):933–5.
Howells DW, Porritt MJ, Rewell SS, O’Collins V, Sena ES, van der Worp HB, et al. Different strokes for different folks: the rich diversity of animal models of focal cerebral ischemia. J Cereb Blood Flow Metab. 2010;30(8):1412–31.
Rewell SS, Fernandez JA, Cox SF, Spratt NJ, Hogan L, Aleksoska E, et al. Inducing stroke in aged, hypertensive, diabetic rats. J Cereb Blood Flow Metab. 2010;30(4):729–33.
Buga AM, Di Napoli M, Popa-Wagner A. Preclinical models of stroke in aged animals with or without comorbidities: role of neuroinflammation. Biogerontology. 2013;14(6):651–62.
Quillinan N, Deng G, Grewal H, Herson PS. Androgens and stroke: good, bad or indifferent? Exp Neurol. 2014;259:10–5.
Cai B, Wang N. Large animal stroke models vs. Rodent stroke models, pros and cons, and combination? Acta Neurochir Suppl. 2016;121:77–81.
Boltze J, Ayata C, Wagner DC, Plesnila N. Preclinical phase III trials in translational stroke research: call for collective design of framework and guidelines. Stroke. 2014;45(2):357.
Boltze J, Wagner DC, Henninger N, Plesnila N, Ayata C. Preclinical phase III trials in translational stroke research: community response on framework and guidelines. Transl Stroke Res. 2016. doi:10.1007/s12975-016-0474-6.
Sandercock P. Negative results: why do they need to be published? Int J Stroke. 2012;7(1):32–3.
Feuerstein GZ, Chavez J. Translational medicine for stroke drug discovery: the pharmaceutical industry perspective. Stroke. 2009;40(3 Suppl):S121–5.
Carpenter Jr WT, Koenig JI, Bilbe G, Bischoff S. At issue: a model for academic/industry collaboration. Schizophr Bull. 2004;30(4):997–1004.
Wang L, Plump A, Ringel M. Racing to define pharmaceutical R&D external innovation models. Drug Discov Today. 2015;20(3):361–70.
Hodson R. Open innovation. Nature. 2016;533(7602):S53.
Frei P, Dev KK. Drug dealers: $20 trillion of in-licensing payments. Drug Discov Today. 2013;18(21–22):1027–9.
Comanor WS, Scherer FM. Mergers and innovation in the pharmaceutical industry. J Health Econ. 2013;32(1):106–13.
Wong TY. How to bridge the “valley of death” between a research discovery and clinical application? Ann Acad Med Singapore. 2014;43(8):422–4.
Acknowledgments
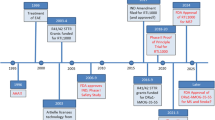
We thank Professor Charli Kruse for providing input and valuable perspectives on academic-industry collaborations. Illustrative elements in Figure 1 were either taken from inhouse material or purchased from the fotolia webpage (rodent pictures only, www.fotolia.com).
Author contribution
JB, DCW, and MJG wrote the manuscript. All authors corrected and improved the draft, and approved the final manuscript version.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Funding
Only institutional funds were used for this project.
Rights and permissions
About this article
Cite this article
Boltze, J., Wagner, DC., Barthel, H. et al. Academic-industry Collaborations in Translational Stroke Research. Transl. Stroke Res. 7, 343–353 (2016). https://doi.org/10.1007/s12975-016-0475-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-016-0475-5