Abstract
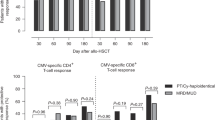
We analyzed cytomegalovirus (CMV) infection risk factors and immune reconstitution kinetics in 89 patients after allogeneic stem cell transplantation (allo-SCT). The use of alemtuzumab for in vivo T cell depletion (TCD) had, besides the donor/recipient CMV serostatus, the strongest influence on the CMV infection risk in univariate and multivariate analyses. In comparison to without use of in vivo TCD, the CMV infection risk [hazard ratio (HR)] was 4.82-fold after TCD with alemtuzumab, but only 1.40-fold after TCD with antithymocyte globulin (ATG). Alemtuzumab strongly depressed CD4+ and CD8+ T cell reconstitution, whereas ATG only delayed CD4+ T cell reconstitution. Considering the reconstitution kinetics of CD4+ and CD8+ T cells, CMV-specific CD8+ T cells, NK cells and the IgG concentration, only a low day +60 NK cell count (≤161 versus >161/μl) was significantly associated with CMV infection development (HR 2.92, p = 0.034). CMV-specific CD8+ T cells were detected in 57% of patients with a CMV-seropositive donor, but in none of the patients with a CMV-seronegative donor on day +30 (p = 0.01). Our data indicate that the type of in vivo TCD (alemtuzumab or ATG) differentially influences both the CMV infection risk and CD4+/CD8+ T cell reconstitution kinetics in patients after allo-SCT.



Similar content being viewed by others
References
Boeckh M, Leisenring W, Riddell SR, Bowden RA, Huang ML, Myerson D, et al. Late cytomegalovirus disease and mortality in recipients of allogeneic hematopoietic stem cell transplants: importance of viral load and T-cell immunity. Blood. 2003;101:407–14.
Bjorklund A, Aschan J, Labopin M, Remberger M, Ringden O, Winiarski J, et al. Risk factors for fatal infectious complications developing late after allogeneic stem cell transplantation. Bone Marrow Transplant. 2007;40:1055–62.
Ozdemir E, Saliba RM, Champlin RE, Couriel DR, Giralt SA, de Lima M, et al. Risk factors associated with late cytomegalovirus reactivation after allogeneic stem cell transplantation for hematological malignancies. Bone Marrow Transplant. 2007;40:125–36.
Storek J, Dawson MA, Storer B, Stevens-Ayers T, Maloney DG, Marr KA, et al. Immune reconstitution after allogeneic marrow transplantation compared with blood stem cell transplantation. Blood. 2001;97:3380–9.
Robin M, Porcher R, De Castro Araujo R, de Latour RP, Devergie A, Rocha V, et al. Risk factors for late infections after allogeneic hematopoietic stem cell transplantation from a matched related donor. Biol Blood Marrow Transplant. 2007;13:1304–12.
Mohty M, Faucher C, Vey N, Stoppa AM, Viret F, Chabbert I, et al. High rate of secondary viral and bacterial infections in patients undergoing allogeneic bone marrow mini-transplantation. Bone Marrow Transplant. 2000;26:251–5.
Junghanss C, Boeckh M, Carter RA, Sandmaier BM, Maris MB, Maloney DG, et al. Incidence and outcome of cytomegalovirus infections following nonmyeloablative compared with myeloablative allogeneic stem cell transplantation, a matched control study. Blood. 2002;99:1978–85.
Mohty M, Jacot W, Faucher C, Bay JO, Zandotti C, Collet L, et al. Infectious complications following allogeneic HLA-identical sibling transplantation with antithymocyte globulin-based reduced intensity preparative regimen. Leukemia. 2003;17:2168–77.
Han XY. Epidemiologic analysis of reactivated cytomegalovirus antigenemia in patients with cancer. J Clin Microbiol. 2007;45:1126–32.
Ljungman P, Brand R, Einsele H, Frassoni F, Niederwieser D, Cordonnier C. Donor CMV serologic status and outcome of CMV-seropositive recipients after unrelated donor stem cell transplantation: an EBMT megafile analysis. Blood. 2003;102:4255–60.
Boeckh M, Nichols WG. The impact of cytomegalovirus serostatus of donor and recipient before hematopoietic stem cell transplantation in the era of antiviral prophylaxis and preemptive therapy. Blood. 2004;103:2003–8.
Hambach L, Stadler M, Dammann E, Ganser A, Hertenstein B. Increased risk of complicated CMV infection with the use of mycophenolate mofetil in allogeneic stem cell transplantation. Bone Marrow Transplant. 2002;29:903–6.
Van Kraaij MG, Verdonck LF, Rozenberg-Arska M, Dekker AW. Early infections in adults undergoing matched related and matched unrelated/mismatched donor stem cell transplantation: a comparison of incidence. Bone Marrow Transplant. 2002;30:303–9.
Chakrabarti S, Milligan DW, Brown J, Osman H, Vipond IB, Pamphilon DH, et al. Influence of cytomegalovirus (CMV) sero-positivity on CMV infection, lymphocyte recovery and non-CMV infections following T-cell-depleted allogeneic stem cell transplantation: a comparison between two T-cell depletion regimens. Bone Marrow Transplant. 2004;33:197–204.
Chakrabarti S, Mackinnon S, Chopra R, Kottaridis PD, Peggs K, O’Gorman P, et al. High incidence of cytomegalovirus infection after nonmyeloablative stem cell transplantation: potential role of Campath-1H in delaying immune reconstitution. Blood. 2002;99:4357–63.
Pérez-Simón JA, Kottaridis PD, Martino R, Craddock C, Caballero D, Chopra R, Spanish and United Kingdom Collaborative Groups for Nonmyeloablative Transplantation, et al. Nonmyeloablative transplantation with or without alemtuzumab: comparison between 2 prospective studies in patients with lymphoproliferative disorders. Blood. 2002;100:3121–7.
Yoon HS, Lee JH, Choi ES, Seo JJ, Moon HN, Kim MN, et al. Cytomegalovirus infection in children who underwent hematopoietic stem cell transplantation at a single center: a retrospective study of the risk factors. Pediatr Transplant. 2009;13:898–905.
Ljungman P, Griffiths P, Paya C. Definitions of cytomegalovirus infection and disease in transplant recipients. Clin Infect Dis. 2002;34:1094–7.
Kern F, Bunde T, Faulhaber N, Kiecker F, Khatamzas E, Rudawski IM, et al. Cytomegalovirus (CMV) phosphoprotein 65 makes a large contribution to shaping the T cell repertoire in CMV-exposed individuals. J Infect Dis. 2002;185:1709–16.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Hebart H, Einsele H. Clinical aspects of CMV infection after stem cell transplantation. Hum Immunol. 2004;65:432–6.
Iversen AC, Norris PS, Ware CF, Benedict CA. Human NK cells inhibit cytomegalovirus replication through a noncytolytic mechanism involving lymphotoxin-dependent induction of IFN-beta. J Immunol. 2005;175:7568–74.
Kuijpers TW, Baars PA, Dantin C, van den Burg M, van Lier RA, Roosnek E. Human NK cells can control CMV infection in the absence of T cells. Blood. 2008;112:914–5.
Boeckh M, Nichols WG. Immunosuppressive effects of beta-herpesviruses. Herpes. 2003;10:12–6.
Ganepola S, Gentilini C, Hilbers U, Lange T, Rieger K, Hofmann J, et al. Patients at high risk for CMV infection and disease show delayed CD8+ T-cell immune recovery after allogeneic stem cell transplantation. Bone Marrow Transplant. 2007;39:293–9.
Ozdemir E, St John LS, Gillespie G, Rowland-Jones S, Champlin RE, Molldrem JJ, et al. Cytomegalovirus reactivation following allogeneic stem cell transplantation is associated with the presence of dysfunctional antigen-specific CD8+ T cells. Blood. 2002;100:3690–7.
Hakki M, Riddell SR, Storek J, Carter RA, Stevens-Ayers T, Sudour P, et al. Immune reconstitution to cytomegalovirus after allogeneic hematopoietic stem cell transplantation: impact of host factors, drug therapy, and subclinical reactivation. Blood. 2003;102:3060–7.
Nakamura R, Cortez K, Solomon S, Battiwalla M, Gill VJ, Hensel N, et al. High-dose acyclovir and pre-emptive ganciclovir to prevent cytomegalovirus disease in myeloablative and non-myeloablative allogeneic stem cell transplantation. Bone Marrow Transplant. 2002;30:235–42.
Busca A, Lovisone E, Aliberti S, Locatelli F, Serra A, Scaravaglio P, et al. Immune reconstitution and early infectious complications following nonmyeloablative hematopoietic stem cell transplantation. Hematology. 2003;8:303–11.
Jiménez M, Ercilla G, Martínez C. Immune reconstitution after allogeneic stem cell transplantation with reduced-intensity conditioning regimens. Leukemia. 2007;21:1628–37.
Kalpoe JS, van der Heiden PL, Vaessen N, Claas EC, Barge RM, Kroes AC. Comparable incidence and severity of cytomegalovirus infections following T cell-depleted allogeneic stem cell transplantation preceded by reduced intensity or myeloablative conditioning. Bone Marrow Transplant. 2007;40:137–43.
Maris M, Boeckh M, Storer B, Dawson M, White K, Keng M, et al. Immunologic recovery after hematopoietic cell transplantation with nonmyeloablative conditioning. Exp Hematol. 2003;31:941–52.
Schulenburg A, Fischer M, Kalhs P, Mitterbauer M, Rabitsch W, Greinix HT, et al. Immune recovery after conventional and non-myeloablative allogeneic stem cell transplantation. Leuk Lymphoma. 2005;46:1755–60.
Acknowledgments
We thank Ms. C. Grobecker for her excellent data documentation. Dr. J. Weirowski made valuable grammatical and stylistic suggestions.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Schmidt-Hieber, M., Schwarck, S., Stroux, A. et al. Immune reconstitution and cytomegalovirus infection after allogeneic stem cell transplantation: the important impact of in vivo T cell depletion. Int J Hematol 91, 877–885 (2010). https://doi.org/10.1007/s12185-010-0597-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-010-0597-6