Abstract
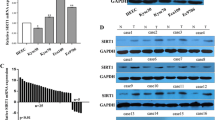
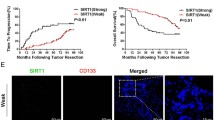
Sirtuin 1 (SIRT1) has been reported to have diverse roles in various biological processes through deacetylation of histone and nonhistone proteins. However, the correlations between SIRT1 protein expression, clinicopathological parameters, and survival of colorectal cancer patients remain unclear. SIRT1 protein expression in a paraffin-embedded tissue microarray, including 13 benign adenomas, nine liver metastasis tissues, and 120 paired colorectal cancer and normal mucosa tissues, was measured by immunohistochemistry. SIRT1 mRNA and protein expression in colon cancer cell lines with different metastatic potential and normal colon cells were detected by real-time reverse transcription polymerase chain reaction (RT-PCR) and Western blotting. The correlations between SIRT1 protein expression, clinicopathological features, and prognosis were analyzed. All samples (100 %) were positive for SIRT1, with variable staining in the cytoplasm rather than the nucleus. There was significant difference in SIRT1 overexpression between adenocarcinomas and normal mucosal tissues (P < 0.01, χ 2 test). SIRT1 overexpression was more frequently observed in advanced-stage tumors and lymph node or liver metastases (P = 0.046, 0.002, and 0.004, respectively, χ 2 test). SIRT1 expression was also significantly elevated in the more aggressive colon cancer cell line SW620. SIRT1 overexpression was significantly correlated with poor overall survival (P = 0.013, log-rank test) and disease-free survival (P = 0.012, log-rank test). SIRT1 overexpression was correlated with advanced-stage and poor prognosis. SIRT1 may play an important role in the progression of colorectal cancer.



Similar content being viewed by others
References
Guarente L, Franklin H. Epstein lecture: sirtuins, aging, and medicine. N Engl J Med. 2011;364:2235–44.
Houtkooper RH, Pirinen E, Auwerx J. Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol. 2012;13:225–38.
Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B, et al. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science. 2004;305:390–2.
Boily G, Seifert EL, Bevilacqua L, He XH, Sabourin G, Estey C, et al. SirT1 regulates energy metabolism and response to caloric restriction in mice. PLoS One. 2008;3:e1759.
Kundu JK, Surh YJ. Emerging avenues linking inflammation and cancer. Free Radic Biol Med. 2012;52:2013–37.
Zhu X, Liu Q, Wang M, Liang M, Yang X, Xu X, et al. Activation of Sirt1 by resveratrol inhibits TNF-alpha induced inflammation in fibroblasts. PLoS One. 2011;6:e27081.
Zhang Z, Lowry SF, Guarente L, Haimovich B. Roles of SIRT1 in the acute and restorative phases following induction of inflammation. J Biol Chem. 2010;285:41391–401.
Ming M, Shea CR, Guo X, Li X, Soltani K, Han W, et al. Regulation of global genome nucleotide excision repair by SIRT1 through xeroderma pigmentosum C. Proc Natl Acad Sci USA. 2010;107:22623–8.
Zhang Y, Zhang M, Dong H, Yong S, Li X, Olashaw N, et al. Deacetylation of cortactin by SIRT1 promotes cell migration. Oncogene. 2009;28:445–60.
Chen HC, Jeng YM, Yuan RH, Hsu HC, Chen YL. SIRT1 promotes tumorigenesis and resistance to chemotherapy in hepatocellular carcinoma and its expression predicts poor prognosis. Ann Surg Oncol. 2012;19:2011–9.
Sung JY, Kim R, Kim JE, Lee J. Balance between SIRT1 and DBC1 expression is lost in breast cancer. Cancer Sci. 2010;101:1738–44.
Feng AN, Zhang LH, Fan XS, Huang Q, Ye Q, Wu HY, et al. Expression of SIRT1 in gastric cardiac cancer and its clinicopathologic significance. Int J Surg Pathol. 2011;19:743–50.
Zhao G, Cui J, Zhang JG, Qin Q, Chen Q, Yin T, et al. SIRT1 RNAi knockdown induces apoptosis and senescence, inhibits invasion and enhances chemosensitivity in pancreatic cancer cells. Gene Ther. 2011;18:920–8.
Stunkel W, Peh BK, Tan YC, Nayagam VM, Wang X, Salto-Tellez M, et al. Function of the SIRT1 protein deacetylase in cancer. Biotechnol J. 2007;2:1360–8.
Kabra N, Li Z, Chen L, Li B, Zhang X, Wang C, et al. SirT1 is an inhibitor of proliferation and tumor formation in colon cancer. J Biol Chem. 2009;284:18210–7.
Jang SH, Min KW, Paik SS, Jang KS. Loss of SIRT1 histone deacetylase expression associates with tumour progression in colorectal adenocarcinoma. J Clin Pathol. 2012;65:735–9.
Haigis MC, Sinclair DA. Mammalian sirtuins: biological insights and disease relevance. Annu Rev Pathol. 2010;5:253–95.
Tanno M, Sakamoto J, Miura T, Shimamoto K, Horio Y. Nucleocytoplasmic shuttling of the NAD+-dependent histone deacetylase SIRT1. J Biol Chem. 2007;282:6823–32.
Byles V, Chmilewski LK, Wang J, Zhu L, Forman LW, Faller DV, et al. Aberrant cytoplasm localization and protein stability of SIRT1 is regulated by PI3K/IGF-1R signaling in human cancer cells. Int J Biol Sci. 2010;6:599–612.
Eades G, Yao Y, Yang M, Zhang Y, Chumsri S, Zhou Q. miR-200a regulates SIRT1 expression and epithelial to mesenchymal transition (EMT)-like transformation in mammary epithelial cells. J Biol Chem. 2011;286:25992–6002.
Chu F, Chou PM, Zheng X, Mirkin BL, Rebbaa A. Control of multidrug resistance gene mdr1 and cancer resistance to chemotherapy by the longevity gene sirt1. Cancer Res. 2005;65:10183–7.
Acknowledgments
This study was supported by Grants from the National Science Foundation of China (81372290 and 81372291), Beijing Municipal Science and Technology Project (2107000074), and Peking University People’s Hospital Research and Development Funds (RDB 2012-18 and RDB 2013-15).
Conflict of interest
There are no conflict of interests to disclose.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Liang Lv and Zhanlong Shen own equal first authorship.
Rights and permissions
About this article
Cite this article
Lv, L., Shen, Z., Zhang, J. et al. Clinicopathological significance of SIRT1 expression in colorectal adenocarcinoma. Med Oncol 31, 965 (2014). https://doi.org/10.1007/s12032-014-0965-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0965-9