Abstract
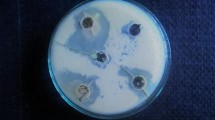
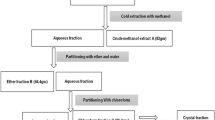
Black pepper (Piper nigrum L.) has gained a global consideration because of its volume in the spice industry. This plant has shown great potential for having medical applications and novel biologically active compounds. In black pepper, natural lipophilic amide piperine was isolated. Piperine was isolated from the fruit using glacial acetic acid as solvent. After isolation and characterization of piperine were done, then it is used to prepare new and novel derivatives of piperine through the experimental procedure. The derivatives of piperine were synthesized by condensation of piperic acid with different substituted aromatic amines, to make a new derivative of piperine, such as ((2E, 4E)-5-(1,3-benzodioxol-5-yl)-N-(1,3-benzothiazol-2-yl)penta-2,4-dienamide) and a novel derivative ((2E, 4E)-5-(1,3-benzodioxol-5-yl)-N-(1,3-benzothiazol-2-yl)penta-2,4-dienehydrazide). The structure of piperine and the derivatives were elucidated through a combination of spectrometric techniques, including ultraviolet spectrophotometer, Mass spectrometry Fourier transform infrared spectroscopy, proton nuclear magnetic resonance.









Similar content being viewed by others
References
R.K. Saxena, P. Venkaiah, L. Anitha, R.M. Venu, Int. J. Food Sci. Nutr. 58, 250–260 (2007)
S. Hartwig, B. Malgorzata, Q. Rolf, S. Wolfgang, L. Gerd, J. Agric. Food Chem. 53, 3358–3363 (2005)
A.C. Nisar, F. Hina, H.A. Bilal, F. Shahid, A. Mohammad, A.K. Mubarak, Asian Pac. J. Trop. Biomed. 2, S1945–S1953 (2012)
T. Jagella, W. Grosch, Eur. Food Res. Technol. 2091, 22–26 (1999)
M. Meghwal, T.K. Goswami, Open Access Scientific Reports 1, 129 (2012). doi:10.4172/scientificreports.129
U. Parimi, D. Kolli, V.R.V. Durvasula, Med. Chem. Res. 22, 5466–5471 (2013)
I. Gulcin, Int. J. Food Sci. Nutr. 56, 491–499 (2005)
Y. Liu, V.R. Yadev, B.B. Aggarwal, M.G. Nair, Nat. Prod. Commun. 5, 1253–1257 (2010)
H.J. Dorman, S. Deans, J. Appl. Microbiol. 88, 308–316 (2000)
M. Vachana, J.P. Samuel, K. Geema, P.S. Evan, KhR Mahaboob, Inflammation 35, 1348–1356 (2012)
R. El-Hamas, M. Idaomar, A. Alonso-Moraga, S.A. Munoz, Food Chem. Toxicol. 41, 41–47 (2003)
G.A. Agbor, J.A. Vinson, J.E. Oben, J.Y. Ngogang, Nutr. Res. 12, 659–663 (2006)
E.S. Sunila, G. Kuttan, J. Ethnopharmacol. 90, 339–346 (2004)
O. Dong-yun, Z. Long-hui, P. Hao, X. Li-hui, W. Yao, L. Kun-peng, H. Xian-hui, Food Chem. Toxicol. 60, 424–430 (2013)
R.S. Vijayakumar, D. Surya, N. Nalini, Redox Rep. 9, 105–110 (2004)
N. Pathak, S. Khandlewal, Eur. J. Pharmacol. 576, 160–170 (2007)
C.R. Pradeep, G. Kuttan, Clin. Exp. Metastasis 19, 703–708 (2002)
S. Panda, A. Kar, Hormon. Metab. Res. 35(9), 523–526 (2003)
N. Pathak, S. Khandelwalm, Environ. Toxicol. Pharmacol. 28, 52–60 (2009)
K. Srinivasan, Food Res. Int. 38, 77–86 (2005)
D. Acharaporn, I. Kornkanok, P. Sakonwun, L. Nanteetip, J. Nat. Med. 67, 303–310 (2013)
M.H. Mehmood, A.H. Gilani, J. Med. Food 13, 1086–1096 (2010)
R.A. Clery, C.J. Hammond, A.C. Wright, J. Essent. Oil Res. 18, 1–3 (2006)
V. Badmaev, M. Majeed, P.N. Edward, Nutr. Res. 19, 381–388 (1999)
B. Vladimir, M. Muhammed, P. Lakshmi, J. Nutr. Biochem. 11(2), 109–113 (2000)
R.K. Bhardwaj, H. Glaesser, L. Becquemont, U. Klotz, S.K. Gupta, M.F. Fromm, J. Pharmacol. Exp. Ther. 302, 645–650 (2002)
R.S. Vijayakumar, N. Nalini, J. Food Biochem. 30, 405–421 (2006)
T. Velpandian, R. Jasuja, R.K. Bhardwaj, J. Jaiswal, S.K. Gupta, Eur. J. Drug Metab. Pharmacokinet. 26, 241–247 (2001)
T. Anette, B. Carin, W. Tina, E. Stéphane, S. Ankie, F. Markus, B. Paul, Food Chem. Toxicol. 66, 350–357 (2014)
P.V. Srinivas, J.M. Rao, Phytochemistry 52, 957–958 (1999)
R. Capasso, A.A. Izzo, F. Borrelli, A. Russo, L. Sautebin, A. Pinto, F. Capasso, N. Mascolo, Life Sci. 71, 2311–2317 (2002)
K.V. Padalkar, V.G. Gaikar, Sep. Sci. Technol. 43(12–11), 3097–3118 (2008)
S.R. Sachin, K.R. Virendra, Ind. Crops Prod. 58, 259–264 (2014)
C. Perakis, V. Louli, K. Magoulas, J. Food Eng. 71(4), 386–393 (2005)
A.C. Kumoro, S. Harcharan, H. Masitah, Chin. J. Chem. Eng. 17(6), 1014–1020 (2009)
A.B. Wood, L.B. Maureen, D.J. James, Flavour Fragr. J. 3, 55–64 (1988)
Acknowledgments
The authors would like to thank technical staff of Chemistry Department at Raparin University and Jamia Hamdard (Hamdard University) to let do the practical work in their chemical laboratories. This work was financed with grants from HCDP programme in Ministry of Higher Education and Scientific Research of Kurdistan – Iraq.
Conflict of interest
The authors declare that they have no conflict of interest. This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aziz, D.M., Hama, J.R. & Alam, S.M. Synthesising a novel derivatives of piperine from black pepper (Piper nigrum L.). Food Measure 9, 324–331 (2015). https://doi.org/10.1007/s11694-015-9239-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-015-9239-2