Abstract
Objective
Stanniocalcin-1 (STC-1), a kind of glycoprotein hormone, is universally up-regulated in various tumor tissues compared to corresponding normal tissues, suggesting it may be used as a tumor marker, whilst disseminated tumor cells usually exist in peripheral blood. The aim of this study is to investigate the mRNA expression STC-1 in peripheral blood of colorectal cancer (CRC) and analyze its clinicopathological significance.
Methods
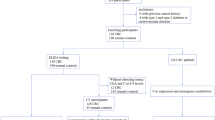
The peripheral blood mononuclear cells (PBMNCs) were isolated from 78 CRC patients and 33 cancer-free controls. The expression status of STC-1 mRNA in PBMNCs was assessed by RT-PCR, its correlation with clinicopathological parameters and 5-year overall survival was analyzed as well.
Results
In the 78 blood samples from CRC patients, 33 (42.31%) showed positive expression of STC-1 mRNA, and all of 15 gastrointestinal tumor tissues were positive for STC-1 mRNA. In contrast, all the blood samples from 14 healthy donors and 19 patients with inflammatory gastrointestinal disease were negative. Furthermore, STC-1 mRNA expression status was associated with patients’ advanced stage, distant metastasis and shortened overall survival.
Conclusion
The detection of STC-1 mRNA in peripheral blood by RT-PCR was highly sensitive and specific for the patients with CRC. STC-1 mRNA may be a potential biomarker for detecting tumor micrometastasis and predicting prognosis.
Similar content being viewed by others
References
Wikman H, Vessella R, Pantel K. Cancer micro-metastasis and tumour dormancy[J]. APMIS 2008; 116:754–770.
Feezor RJ, Copeland EM 3rd, Hochwald SN. Significance of micrometastases in colorectal cancer[J]. Ann Surg Oncol 2002; 9: 944–953.
Wagner GF, Jaworski EM, Haddad M. Stanniocalcin in the seawater salmon: structure, function, and regulation[J]. Am J Physiol 1998; 274:R1177–R1185.
Ishibashi K, Imai M. Prospect of a stanniocalcin endocrine/paracrine system in mammals[J]. Am J Physiol Renal Physiol 2002; 282:F367–F375.
Li K, Dong D, Yao L, et al. Identification of STC-1 as an beta-amyloid activated gene in human brain microvascular endothelial cells using cDNA microarray[J]. Biochem Biophys Res Commun 2008; 376:399–403.
Wu S, Yoshiko Y, De Luca F. Stanniocalcin 1 acts as a paracrine regulator of growth plate chondrogenesis[J]. J Biol Chem 2006; 281: 5120–5127.
Chang AC, Jellinek DA, Reddel RR. Mammalian stanniocalcins and cancer[J]. Endocr Relat Cancer 2003; 10:359–373.
Joensuu K, Heikkilä P, Andersson LC. Tumor dormancy: Elevated expression of stanniocalcins in late relapsing breast cancer[J]. Cancer Lett 2008; 265:76–83.
Fujiwara Y, Sugita Y, Nakamori S, et al. Assessment of stanniocalcin-1 mRNA as a molecular market for micrometastases of various human cancers[J]. Int J Oncol 2000; 16:799–804.
Yang HW, Yang NW, Cao J, et al. Detection of SBEM-mRNA in peripheral blood of patients with breast cancer and its clinical significance[J]. Chin J Cancer Res 2006; 18:294–298.
Huang P, Wang J, Guo Y, et al. Molecular detection of disseminated tumor cells in the peripheral blood in patients with gastrointestinal cancer[J]. J Cancer Res Clin Oncol 2003; 129:192–198.
De Niu P, Radman DP, Jaworski EM, et al. Development of a human stanniocalcin radio-immunoassay: serum and tissue hormone levels and pharmacokinetics in the rat[J]. Mol Cell Endocrinol 2000; 162:131–144.
Deol HK, Varghese R, Wagner GF, et al. Dynamic regulation of mouse ovarian stanniocalcin expression during gestation and lactation[J]. Endocrinology 2000; 141:3412–3421.
McCudden CR, Majewski A, Chakrabarti S, et al. Co-localization of stanniocalcin-1 ligand and receptor in human breast carcinomas[J]. Mol Cell Endocrinol 2004; 213:167–172.
Gerritsen ME, Soriano R, Yang S, et al. In silico data filtering to identify new angiogenesis targets from a large in vitro gene profiling data set[J]. Physiol Genomics 2002; 10:13–20.
Koide Y, Sasaki T. Stanniocalcin-1 (STC-1) as a molecular marker for human cancer[J]. Rinsho Byori 2006; 54:213–220.
Wascher RA, Huynh KT, Giuliano AE, et al. Stanniocalcin-1: a novel molecular blood and bone marrow marker for human breast cancer[J]. Clin Cancer Res 2003; 9:1427–1435.
Tohmiya Y, Koide Y, Fujimaki S, et al. Stanniocalcin-1 as a novel marker to detect minimal residual disease of human leukemia[J]. Tohoku J Exp Med 2004; 204:125–133.
Yamashita K, Watanabe M. Clinical significance of tumor markers and an emerging perspective on colorectal cancer[J]. Cancer Sci 2009; 100:195–199.
Filvaroff EH, Guillet S, Zlot C, et al. Stanniocalcin 1 alters muscle and bone structure and function in transgenic mice[J]. Endocrinology 2002; 143:3681–3690.
Paciga M, Watson AJ, DiMattia GE, et al. Ovarian stanniocalcin is structurally unique in mammals and its production and release are regulated through the luteinizing hormone receptor[J]. Endocrinology 2002; 143:3925–3934.
Kahn J, Mehraban F, Ingle G, et al. Gene expression profiling in an in vitro model of angiogenesis[J]. Am J Pathol 2000; 156:1887–1900.
Wary KK, Thakker GD, Humtsoe JO, et al. Analysis of VEGF-responsive genes involved in the activation of endothelial cells[J]. Mol Cancer 2003; 2:25.
Zlot C, Ingle G, Hongo J, et al. Gerritsen, Stanniocalcin 1 is an autocrine modulator of endothelial angiogenic responses to hepatocyte growth factor[J]. J Biol Chem 2003; 278:47654–47659.
Chakraborty A, Brooks H, Zhang P, et al. Stanniocalcin-1 regulates endothelial gene expression and modulates transendothelial migration of leukocytes[J]. Am J Physiol Renal Physiol 2007; 292:F895–F904.
Ellard JP, McCudden CR, Tanega C, et al. The respiratory effects of stanniocalcin-1 (STC-1) on intact mitochondria and cells: STC-1 uncouples oxidative phosphorylation and its actions are modulated by nucleotide triphosphates[J]. Mol Cell Endocrinol 2007; 264:90–101.
Macartney-Coxson DP, Hood KA, Shi HJ, et al. Metastatic susceptibility locus, an 8p hot-spot for tumour progression disrupted in colorectal liver metastases: 13 candidate genes examined at the DNA, mRNA and protein level[J]. BMC Cancer 2008; 8:187.
Lai KP, Law AY, Yeung HY, et al. Induction of stanniocalcin-1 expression in apoptotic human nasopharyngeal cancer cells by p53[J]. Biochem Biophys Res Commun 2007; 356:968–975.
Law AY, Lai KP, Lui WC, et al. Histone deacetylase inhibitor-induced cellular apoptosis involves stanniocalcin-1 activation[J]. Exp Cell Res 2008; 314:2975–2984.
McCudden CR, James KA, Hasilo C, et al. Characterization of mammalian stanniocalcin receptors. Mitochondrial targeting of ligand and receptor for regulation of cellular metabolism[J]. J Biol Chem 2002; 277:45249–45258.
Yeung HY, Lai KP, Chan HY, et al. Hypoxia-inducible factor-1-mediated activation of stanniocalcin-1 in human cancer cells[J]. Endocrinology 2005; 146:4951–4960.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the grants from the Department of Health, Jiangsu Province, China (No.H200116).
Rights and permissions
About this article
Cite this article
Ping-ping, W., Peng, W., Pei-lin, H. et al. Stanniocalcin-1 detection of peripheral blood in patients with colorectal cancer. Chin. J. Cancer Res. 22, 274–279 (2010). https://doi.org/10.1007/s11670-010-0274-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11670-010-0274-6