Abstract
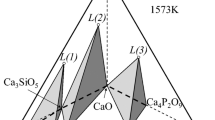
Calcium sulfate (CaSO4) is proposed as an alternative sulfur source to convert the Fe-Ni-Cu-Co alloy to the matte phase. Solid carbon was used as a reducing agent and the influence of oxide fluxes on the sulfurization efficiency at 1673 K (1400 °C) in a CO-CO2-SO2-Ar atmosphere was investigated. When CaSO4 was equilibrated with the Fe-Ni-Cu-Co alloy without any reducing agent, it was reduced by Fe in the liquid alloy, resulting in the formation of FeS. The sulfurization efficiency was about 56 pct, even though an excess amount of CaSO4 (gypsum equivalent, G eq = 1.7) was added. Adding solid carbon as the reducing agent significantly shortened the equilibration time from 36 to 3.5 hours and increased the sulfurization efficiency from 56 to 91 pct, even though the amount of carbon was lower than the theoretical equivalent for carbothermic reduction of CaSO4, viz. C eq = 0.7. Although CaS (not FeS) was formed as a primary reaction product, it continuously reacted with CaSO4, forming CaO-rich slag. Neither the carbothermic reduction time nor the sulfurization efficiency were affected by the addition of Al2O3 (-SiO2) fluxes, but the equilibration time fell to 2.5 hours with the addition of Al2O3-Fe2O3 flux because the former systems produced primarily calcium silicate and calcium aluminate, which have relatively high melting points, whereas the latter system produced calcium ferrite, which has a lower melting point. Consequently, calcium sulfate (waste gypsum) can replace expensive pure sulfur as a raw material in the sulfurization of Fe-Ni-Cu-Co alloy with small amounts of iron oxide (Fe2O3) as a flux material. The present results can be used to improve the recovery of rare metals, such as Ni and Co, from deep sea manganese nodules.













Similar content being viewed by others
References
K.H. Park, D. Mohapatra, and B.R. Reddy: Sep. Purif. Technol., 2006, vol. 51, pp. 332-37.
P.A. Rona: Science, 2003, vol. 299, pp. 673-74.
C.W. Nam, B.S. Kim, and K.H. Park: Kor. Soc. Geosyst. Eng., 2003, vol. 40, pp. 191-97.
K.H. Park, D. Mohapatra, H.I. Kim, and X. Guo: Sep. Purif. Technol., 2007, vol. 56, pp. 303-10.
K.H. Park, C.W. Nam, J.S. Chang, S.C. Ahan, and H.I. Kim: J. Kor. Inst. Resources Recycling, 2008, vol. 17, pp. 51-58.
K.H. Park and C.W. Nam: Trends in Met. & Mater. Eng., 2002, vol. 15, pp. 15-23.
R. Kuusik, P. Saikkonen, and L. Niinisto: J. Therm. Anal., 1985, vol. 30, pp.187-93.
T.D. Wheelock and C.E. Morris: TIZ-Fachberichte., 1986, vol. 110, pp. 37-46.
J.S. Oh and T.D. Wheelock: Ind. Eng. Chem. Res., 1990, vol. 29, pp. 544-50.
X. Yang, Z. Zhang, X. Wang, L. Yang, B. Zhong, and J. Liu: J. Chem. Thermodyn., 2013, vol. 57, pp. 39-45.
X. Zhang, X. Song, Z. Sun, P. Li, and J. Yu: Ind. Eng. Chem. Res., 2012, vol. 51, pp. 6563-70.
B.B. Kale, A.R. Pande, and A. N. Gokarn: Metall. Trans. B., 1992, vol 23B, pp. 567-72.
L. Ma, P. Ning, S. Zheng, X. Niu, W. Zhang, and Y. Du: Ind. Eng. Chem. Res., 2010, vol. 49, pp. 3597-3602.
N. Mihara, D. Kuchar, Y. Kojima, and H. Matsuda: J. Mater. Cycles. Waste Manag., 2007, vol. 9, pp. 21-26.
E.S. Newman: J. Res. Natl Bur Stand., 1941, vol. 27, pp. 191–96.
T.D. Wheelock and D.R. Boylan: Ind. Eng. Chem.., 1960, vol. 52, pp. 215-18.
Y. Liu, H. Zhou, Y.H. Liu, R. Stanger, L. Elliot, T. Wall, and K.F. Cen: Cleaner Combustion and Sustainable World., H. Qi and B. Zhao ed., Tsinghua University Press, Springer, 2013, pp. 323–29.
L.M. Diaz-Bossio, S.E. Squier, and A.H. Pulsifer: Chem. Eng. Sci., 1985, vol. 40, pp. 319-24.
E.M. van der Merwe, C.A. Strydom, and J.H. Potgieter: Thermochim. Acta., 1999, vol. 340-341, pp. 431-37.
J. Talukdar, P. Basu, and J.H. Greenblatt: Fuel, 1996, vol. 75, pp. 1115-23.
B. Kamphuis, A.W. Potma, W. Prins, and W.P.M. van Swaaij: Chem. Eng. Sci., 1993, vol. 48, pp. 105-16.
E.T. Turkdogan and J.V. Vinters: Trans. Inst. Mining & Metall. Section C, 1976, vol. 85, pp. 117-23.
Iv. Gruncharov, Y. Pelovski, G. Bechev, Iv. Dombalov, and Pl. Kirilov: J. Therm. Anal., 1988, vol. 33, pp. 597-602
J.H. Park, J.G. Park, D.J. Min, Y.E. Lee, and Y.B. Kang: J. Eur. Ceram. Soc., 2010, vol. 30, pp. 3181-86.
www.factsage.com (May 2015).
J.H. Park and G.H. Park: ISIJ Int., 2012, vol. 52, pp. 764-69.
K.Y. Ko and J.H. Park: Metall. Mater. Trans. B, 2012, vol. 43B, pp. 440-42.
D.J. Kim and J.H. Park: Metall. Mater. Trans. B, 2012, vol. 43B, pp. 875-86.
J.H. Heo, S.S. Park, and J.H. Park: Metall. Mater. Trans. B, 2012, vol. 43B, pp. 1098-1105.
J.H. Park: ISIJ Int., 2012, vol. 52, pp. 2303-04.
J.H. Park: Met. Mater. Int., 2013, vol. 19, pp. 577-84.
J.H. Heo, B.S. Kim, and J.H. Park: Metall. Mater. Trans. B, 2013, vol. 44B, pp. 1352-63.
S.K. Kwon, Y.M. Kong, and J.H. Park: Met. Mater. Int., 2014, vol. 20, pp. 959-66.
Y. Kang, J. Jang, J.H. Park, and C. Lee: Met. Mater. Int., 2014, vol. 20, pp. 119-27.
Y.S. Han and J.H. Park: Metall. Mater. Trans. B, 2015, vol. 46B, pp. 235-42.
E.T. Turkdogan: Physical Chemistry of High Temperature Technology, Academic Press, New York, NY, 1980, pp. 1–24.
Acknowledgment
This work was financially supported by Ministry of Oceans and Fisheries, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted June 4, 2015.
Rights and permissions
About this article
Cite this article
Jeong, E.H., Nam, C.W., Park, K.H. et al. Sulfurization of Fe-Ni-Cu-Co Alloy to Matte Phase by Carbothermic Reduction of Calcium Sulfate. Metall Mater Trans B 47, 1103–1112 (2016). https://doi.org/10.1007/s11663-016-0590-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0590-4