Abstract
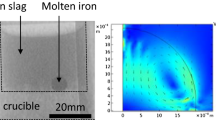
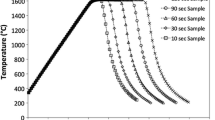
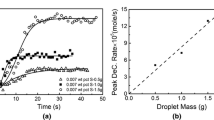
An approach for accommodating the interfacial area changes in kinetic equations for heterogeneous reactions in the presence of spontaneous emulsification has been proposed. The kinetics were analyzed by incorporating time-averaged interfacial areas in the rate equations. The approach was found to be applicable for the experimental data and to satisfactorily describe the reaction kinetics. In the case of a high-temperature reaction between 2.35 g Fe-5 wt pct Al alloy metal droplets with CaO-SiO2-Al2O3 slag at 1650 °C, it was found that the kinetics follow a first-order relationship with respect to aluminum in the metal, and it was concluded that they were controlled by mass transport in the metal phase. The calculated metal mass-transfer coefficient k m was 1.7×10−6 m/s.
Similar content being viewed by others
References
P. Kozakevitch, G. Urbain, and M. Sage: Rev. Metall., 1955, vol. 2, pp. 161–72.
H. Ooi, T. Nozaki, and H. Yoshii: Trans. Iron Steel Inst. Jpn., 1974, vol. 14, pp. 9–16.
P.V. Riboud and L.D. Lucas: Can. Metall. Q., 1981, vol. 20 (2), pp. 199–208.
Y. Chung and A.W. Cramb: Phil. Trans. R. Soc. London A, 1998, vol. 356, pp. 981–93.
Y. Chung and A.W. Cramb: Metall. Mater. Trans. B, 2000, vol. 31B, pp. 957–71.
M.A. Rhamdhani, G.A. Brooks, and K.S. Coley: Proc. Int. Symp. on Oxygen in Steelmaking COM 2004, Aug. 2004, TMSCIM, Hamilton, ON, Canada, 2004, pp. 203–17.
M.A. Rhamdhani, G.A. Brooks, and S.A. Nightingale: Proc. Int. Symp. on Metal/Ceramic Interactions COM 2002, Aug. 2002, TMSCIM, Montreal, 2002, pp. 303–13.
F. Ji, M.A. Rhamdhani, Subagyo, M., Barati, K.S. G.A. Brooks, G.A. Irons, and S.A. Nightingale: High Temp. Mater. Processing, 2003, vol. 22 (5–6), pp. 359–67.
M.A. Rhamdhani and G.A. Brooks: Metall. Mater. Trans. B, 2003, vol. 34B, pp. 355–58.
T. Mori, E. Ichise, and A. Moro-oka: Steelmaking Data Source Book, Gordon and Breach Science Publishers, Montreux, Switzerland, 1988, pp. 45–54.
H. Sakao and T. Fujisawa: Steelmaking Data Source Book, Gordon and Breach Science Publishers, Montreux, Switzerland, 1988, pp. 145–56.
R.J. Pomfret and P. Grieveson: Can. Metall. Q., 1983, vol. 22 (3), pp. 287–99.
B. Deo and R. Boom: Fundamentals of Steelmaking Metallurgy, Prentice-Hall International, New York, NY, 1993, p. 119.
J.W. Robison, Jr. and R.D. Pehlke: Metall. Trans., 1974, vol. 5, pp. 1041–51.
J.J. Moore: Chemical Metallurgy, Butterworth and Co., London, 1990, p. 119.
M. Ashizuka, M. Tokuda, and M. Ohtani: Trans. Iron Steel Inst. Jpn., 1972, vol. 12, pp. 383–92.
A.H.P. Skelland: in Science and Practice of Liquid-Liquid Extraction, J.D. Thornton, ed., Oxford University Press, New York, NY, 1992, pp. 40–156.
R. Higbie: Trans. Inst. Chem. Eng., 1935, vol. 31, pp. 365–89.
S. Glasstone, K.J. Laidler, and H. Eyring: The Theory of Rate Processes, McGraw-Hill, New York, NY, 1941, p. 477.
A.B. Newman: Trans. A.I.Ch.E., 1931, vol. 27, pp. 310–33.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rhamdhani, M.A., Coley, K.S. & Brooks, G.A. Kinetics of metal/slag reactions during spontaneous emulsification. Metall Mater Trans B 36, 219–227 (2005). https://doi.org/10.1007/s11663-005-0023-2
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11663-005-0023-2