Summary
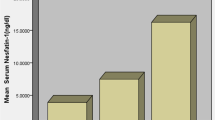
The changes of plasma myostatin levels in patients with type 2 diabetes mellitus (T2D) and their clinical correlation were investigated. We recruited 43 T2D patients and 20 age-matched healthy subjects. Plasma myostatin, lipid and glucose, and serum insulin were determined. T2D patients showed significantly higher fasting plasma glucose (FPG), serum insulin and triglyceride levels, and lower high-density lipoprotein levels than normal control subjects (P<0.01). Mean plasma myostatin level in T2D patients and health controls was (66.5±17.8) and (46.2±13.8) ng/mL, respectively. An unpaired t test showed that the increase of myostatin in the T2D patients was significant (P<0.001). In both healthy control and T2D groups, the female subjects showed higher myostatin levels than the male subjects. In the T2D patients, plasma level of myostatin was negatively correlated with body mass index (BMI, r=−0.42, P<0.01) and FPG (r=−0.51, P〈0.01), but positively correlated with insulin resistance index (HOMA-IR, r=0.48, P<0.01). Up-regulation of plasma myostatin in the T2D patients and its correlation with BMI, FPG and blood insulin sensitivity suggests that plasma myostatin may be implicated in the pathogenesis of T2D and thus presented as a therapeutic target for treating the disease. Furthermore, circulating myostatin levels may be used as a biomarker for the disease.
Similar content being viewed by others
References
Boutayeb A, Boutayeb S. The burden of non communicable diseases in developing countries. Int J Equity Health, 2005,4(1):2
Stumvoll M, Goldstein BJ, van Haeften TW. Type 2 diabetes: principles of pathogenesis and therapy. Lancet, 2005,365(9467):1333–1346
McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature, 1997,387(6628):83–90
Lee SJ, McPherron AC. Regulation of myostatin activity and muscle growth. Proc Natl Acad Sci USA, 2001,98(16): 9306–9311
Philip B, Lu Z, Gao Y. Regulation of GDF-8 signaling by the p38 MAPK. Cell Signal, 2005,17(3):365–375
Yang W, Zhang Y, Li Y, et al. Myostatin induces cyclin D1 degradation to cause cell cycle arrest through a phosphatidylinositol 3-kinase/AKT/GSK-3 beta pathway and is antagonized by insulin-like growth factor 1. J Biol Chem, 2007,282(6):3799–3808
Huang Z, Chen D, Zhang K, et al. Regulation of myostatin signalling by c-Jun N-terminal kinase in C2C12 cells. Cell Signal, 2007,19(11):2286–2295
Zhu X, Topouzis S, Liang LF, et al. Myostatin signaling through Smad2, Smad3 and Smad4 is regulated by the inhibitory Smad7 by a negative feedback mechanism. Cytokine, 2004,26(6):262–272
Kim HS, Liang L, Dean RG, et al. Inhibition of preadipocyte differentiation by myostatin treatment in 3T3-L1 cultures. Biochem Biophys Res Commun, 2001,281(4): 902–906
Chen Y, Ye J, Cao L, et al. Myostatin regulates glucose metabolism via the AMP-activated protein kinase pathway in skeletal muscle cells. Int J Biochem Cell Biol, 2010,42(12):2072–2081
Mitchell MD, Osepchook CC, Leung KC, et al. Myostatin is a human placental product that regulates glucose uptake. J Clin Endocrinol Metab, 2006,91(4):1434–1437
McPherron AC, Lee SJ. Suppression of body fat accumulation in myostatin-deficient mice. J Clin Invest, 2002,109(5):595–601
Lin J, Arnold HB, Della-Fera MA, et al. Myostatin knockout in mice increases myogenesis and decreases adipogenesis. Biochem Biophys Res Commun, 2002,291(3):701–706
Williams MS. Myostatin mutation associated with gross muscle hypertrophy in a child. N Engl J Med, 2004,351(10):1030–1031
Hocquette JF, Bas P, Bauchart D, et al. Fat partitioning and biochemical characteristics of fatty tissues in relation to plasma metabolites and hormones in normal and double-muscled young growing bulls. Comp Biochem Physiol A Mol Integr Physiol, 1999,122(1):127–138
Zhao B, Wall RJ, Yang J. Transgenic expression of myostatin propeptide prevents diet-induced obesity and insulin resistance. Biochem Biophys Res Commun, 2005,337(1):248–255
Mukherjee A, Sidis Y, Mahan A, et al. FSTL3 deletion reveals roles for TGF-β family ligands in glucose and fat homeostasis in adults. Proc Natl Acad Sci USA, 2007,104(4):1348–1353
Allen DL, Cleary AS, Speaker KJ, et al. Myostatin, activin receptor IIb, and follistatin-like-3 gene expression are altered in adipose tissue and skeletal muscle of obese mice. Am J Physiol Endocrinol Metab, 2008,294(5): E918–927
Milan G, Dalla Nora E, Pilon C, et al. Changes in muscle myostatin expression in obese subjects after weight loss. J Clin Endocrinol Metab, 2004,89(6):2724–2727
Hittel DS, Berggren JR, Shearer J, et al. Increased secretion and expression of myostatin in skeletal muscle from extremely obese women. Diabetes, 2009,58(1):30–38
Zachwieja JJ, Smith SR, Sinha-Hikim I, et al. Plasma myostatin-immunoreactive protein is increased after prolonged bed rest with low-dose T3 administration. J Gravit Physiol, 1999,6(2):11–15
Reardon KA, Davis J, Kapsa RM, et al. Myostatin, insulin like growth factor-1, and leukemia inhibitory factor mRNAs are upregulated in chronic human disuse muscle atrophy. Muscle Nerve, 2001,24(7):893–899
Hayot M, Rodriguez J, Vernus B, et al. Myostatin up-regulation is associated with the skeletal muscle response to hypoxic stimuli. Mol Cell Endocrinol, 2011,332(1–2):38–47
Yarasheski KE, Bhasin S, Sinha-Hikim I, et al. Serum myostatin immunoreactive protein is increased in 60–92 year old women and men with muscle wasting. J Nutr Health Aging, 2002,6(5):343–348
Matthews DR, Hosker JP, Rudenski AS, et al. Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia, 1985,28(7):412–419
Lenk K, Schur R, Linke A, et al. Impact of exercise training on myostatin expression in the myocardium and skeletal muscle in a chronic heart failure model. Eur J Heart Fail, 2009,11(4):342–348
Gonzalez-Cadavid NF, Taylor WE, Yarasheski K, et al. Organization of the human myostatin gene and expression in healthy men and HIV-infected men with muscle wasting. Proc Natl Acad Sci USA, 1998,95(25):14938–14943
Mendler L, Baka Z, Kovács-Simon A, et al. Androgens negatively regulate myostatin expression in an androgen-dependent skeletal muscle. Biochem Biophys Res Commun, 2007,361(1):237–242
Hill JJ, Qiu Y, Hewick RM, et al. Regulation of myostatin in vivo by growth and differentiation factor-associated serum protein-1: a novel protein with protease inhibitor and follistatin domains. Mol Endocrinol, 2003,17(6):1144–1154
Lee SJ. Genetic analysis of the role of proteolysis in the activation of latent myostatin. PLoS One, 2008,3(2): e1628
Morley JE, Thomas DR, Wilson MM. Cachexia: pathophysiology and clinical relevance. Am J Clin Nutr, 2006,83(4):735–743
Chen Y, Cao L, Ye J, et al. Upregulation of myostatin gene expression in streptozotocin-induced type 1 diabetes mice is attenuated by insulin. Biochem Biophys Res Commun, 2009,388(1):112–116
Charlton M, Nair KS. Protein metabolism in insulin-dependent diabetes mellitus. J Nutr, 1998,128(2): 323S–327S
Welle S, Burgess K, Mehta S. Stimulation of skeletal muscle myofibrillar protein synthesis, p 70 S6 kinase phosphorylation, and ribosomal protein S6 phosphorylation by inhibition of myostatin in mature mice, Am J Physiol Endocrinol Metab, 2009,296(3):E567–E572
Allen DL, Unterman TG. Regulation of myostatin expression and myoblast differentiation by FoxO and SMAD transcription factors. Am J Physiol Cell Physiol, 2007,292(1):C188–C199
Liu CM, Yang Z, Liu CW, et al. Effect of RNA oligonucleotide targeting Foxo-1 on muscle growth in normal and cancer cachexia mice. Cancer Gene Ther, 2007,14(12):945–952
Wagner KR, Fleckenstein JL, Amato AA, et al. A phase I/II trial of MYO-029 in adult subjects with muscular dystrophy. Ann Neurol, 2008,63(5):561–571
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors contributed equally to this work.
This project was supported by grants from the Educational Bureau of Hubei, China (No. Q20092403, and No. B20082405).
Rights and permissions
About this article
Cite this article
Wang, F., Liao, Y., Li, X. et al. Increased circulating myostatin in patients with type 2 diabetes mellitus. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 32, 534–539 (2012). https://doi.org/10.1007/s11596-012-0092-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-012-0092-9