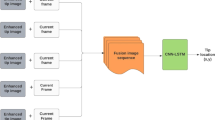
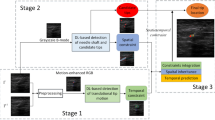
Abstract
Purpose
This paper addresses localization of needles inserted both in-plane and out-of-plane in challenging ultrasound-guided interventions where the shaft and tip have low intensity. Our approach combines a novel digital subtraction scheme for enhancement of low-level intensity changes caused by tip movement in the ultrasound image and a state-of-the-art deep learning scheme for tip detection.
Methods
As the needle tip moves through tissue, it causes subtle spatiotemporal variations in intensity. Relying on these intensity changes, we formulate a foreground detection scheme for enhancing the tip from consecutive ultrasound frames. The tip is augmented by solving a spatial total variation regularization problem using the split Bregman method. Lastly, we filter irrelevant motion events with a deep learning-based end-to-end data-driven method that models the appearance of the needle tip in ultrasound images, resulting in needle tip detection.
Results
The detection model is trained and evaluated on an extensive ex vivo dataset collected with 17G and 22G needles inserted in-plane and out-of-plane in bovine, porcine and chicken phantoms. We use 5000 images extracted from 20 video sequences for training and 1000 images from 10 sequences for validation. The overall framework is evaluated on 700 images from 20 sequences not used in training and validation, and achieves a tip localization error of 0.72 ± 0.04 mm and an overall processing time of 0.094 s per frame (~ 10 frames per second).
Conclusion
The proposed method is faster and more accurate than state of the art and is resilient to spatiotemporal redundancies. The promising results demonstrate its potential for accurate needle localization in challenging ultrasound-guided interventions.





Similar content being viewed by others
References
Elsharkawy H, Babazade R, Kolli S, Kalagara H, Soliman ML (2016) The Infiniti plus ultrasound needle guidance system improves needle visualization during the placement of spinal anesthesia. Korean J Anesthesiol 69(4):417–419
Lu H, Li J, Lu Q, Bharat S, Erkamp R, Chen B, Drysdale J, Vignon F, Jain A (2014) A new sensor technology for 2D ultrasound-guided needle tracking. MICCAI 17(Pt. 2):389–396
Xia W, West S, Finlay M, Mari J, Ourselin S, David A, Desjardins A (2017) Looking beyond the imaging plane: 3D needle tracking with a linear array ultrasound probe. Sci Rep 7(1):3674
Miura M, Takeyama K, Suzuki T (2014) Visibility of ultrasound-guided echogenic needle and its potential in clinical delivery of regional anesthesia. Tokai J Exp Clin Med 39(2):80–86
Arif M, Moelker A, van Walsum T (2018) Needle Tip Visibility in 3D Ultrasound Images. Cardiovasc Interv Radiol 41(1):145–152
Fevre MC, Vincent C, Picard J, Vighetti A, Chapuis C, Detavernier M, Allenet B, Payen JF, Bosson JL, Albaladejo P (2018) Reduced variability and execution time to reach a target with a needle GPS system: comparison between physicians, residents and nurse anaesthetists. Anaesth Crit Care Pain Med 37(1):55–60
Stolka PJ, Foroughi P, Rendina M, Weiss CR, Hager GD, Boctor EM (2014) Needle guidance using handheld stereo vision and projection for ultrasound-based interventions. MICCAI 17(Pt.2):684–691
Priester AM, Natarajan S, Culjat MO (2013) Robotic ultrasound systems in medicine. IEEE EEE Trans Ultrason Ferroelectr Freq Control 60:507–523
Ayvali E, Desai J (2014) Optical flow-based tracking of needles and needle-tip localization using circular hough transform in ultrasound images. Ann Biomed Eng 43(8):1828–1840
Zhao Y, Cachard C, Liebgott H (2013) Automatic needle detection and tracking in 3D ultrasound using an ROI-based RANSAC and Kalman method. Ultrason Imaging 35(4):283–306
Hacihaliloglu I, Beigi P, Ng G, Rohling RN, Salcudean S, Abolmaesumi P (2015) Projection-based phase features for localization of a needle Tip in 2D curvilinear ultrasound. MICCAI 9349:347–354
Hatt CR, Ng G, Parthasarathy V (2015) Enhanced needle localization in ultrasound using beam steering and learning-based segmentation. Comput Med Imaging Graph 41:46–54
Mwikirize C, Nosher JL, Hacihaliloglu I (2018) Signal attenuation maps for needle enhancement and localization in 2D ultrasound. Int J CARS 13(3):363–374
Beigi P, Rohling R, Salcudean S, Ng G (2017) CASPER: computer-aided segmentation of imperceptible motion-a learning-based tracking of an invisible needle in ultrasound. Int J CARS 12(11):1857–1866
Beigi P, Rohling R, Salcudean SE, Ng GC (2016) Spectral analysis of the tremor motion for needle detection in curvilinear ultrasound via spatiotemporal linear sampling. Int J CARS 11(6):1183–1192
Mwikirize C, Nosher JL, Hacihaliloglu I (2018) Convolution neural networks for real-time needle detection and localization in 2D ultrasound. Int J CARS 13(5):647–657
Pourtaherian A, Ghazvinian Zanjani F, Zinger S, Mihajlovic N, Ng G, Korsten H, With P (2017) Improving needle detection in 3D ultrasound using orthogonal-plane convolutional networks. MICCAI 2:610–618
Pourtaherian A, Ghazvinian Zanjani F, Zinger S, Mihajlovic N, Ng G, Korsten H, With P (2018) Robust and semantic needle detection in 3D ultrasound using orthogonal-plane convolutional neural networks. Int J CARS 13(9):1321–1333
Redmon J, Farhadi A (2016) Yolo9000: better, faster, stronger. arXiv:1612.08242
Afonso M, Bioucas-Dias J, Figueiredo M (2010) Fast image recovery using variable splitting and constrained optimization. IEEE Trans Image Process 19(9):2345–2356
Chan S, Khoshabeh R, Gibson K, Gill P, Nguyen T (2011) An augmented lagrangian method for total variation video restoration. IEEE Trans Image Process 20(11):3097–3111
Goldstein T, Osher S (2009) The split Bregman method for L1-regularized problems. SIAM J Imaging Sci 2(2):323–343
Fong D, Saunders M (2011) LSMR: an iterative algorithm for sparse least-squares problems. SIAM J Sci Comput 33(5):2950–2971
Klambauer G, Unterthiner T, Mayr A, Hochreiter S (2017) Self-normalizing neural networks. arXiv:1706.02515v5
Everingham M, Gool LV, Williams CKI, Winn J, Zisserman A (2010) The PASCAL visual object classes (VOC) challenge. Int J Comput Vis 88:303–338
Acknowledgements
This work was accomplished with funding support from the North American Spine Society 2017 young investigator award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
This article does not contain patient data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MP4 51745 kb)
Rights and permissions
About this article
Cite this article
Mwikirize, C., Nosher, J.L. & Hacihaliloglu, I. Learning needle tip localization from digital subtraction in 2D ultrasound. Int J CARS 14, 1017–1026 (2019). https://doi.org/10.1007/s11548-019-01951-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-019-01951-z