Abstract
Purpose
Radiosurgery (RS) is a well-established treatment in selected patients with brain metastasis. The aim of this study is to compare the differences between CyberKnife (CK) and TomoTherapy (HT) treatment plans of RS of single brain metastasis (BM) to define when HT should be used in cases beyond Cyberknife—when both systems are readily available for the radiation oncologist.
Methods and materials
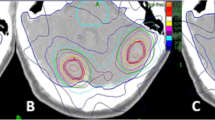
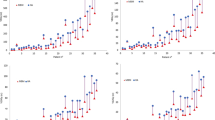
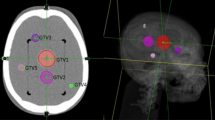
Nineteen patients with single brain metastasis treated with CK were re-planned for radiosurgery using TomoTherapy Hi-ART system. Two planning approaches have been used for TomoTherapy plans: the classical one (HT) and the improved conformity (icHT) that produces dose distributions more similar to those of RS plans. PTV coverage, Conformity Index (CI), Paddick Conformity Index (nCI), Homogeneity Index (HI), Gradient Index (GI), and beam on time of CK, HT, and icHT plans were evaluated and compared.
Results
A good coverage was found for CK, HT, and icHT plans. A difference between mean HI of CK and icHT plans was observed (p = 0.007). Better dose gradients compared to both icHT and HT modalities were observed in CK plans. icHT modality showed improved mean CI respect to HT modality, similar to that obtained in CK plans.
Conclusions
CK plans show higher conformity and lower GI than icHT and HT plans. TomoTherapy demonstrates the advantage of being a device capable to reach different clinical objectives depending on the different planning modality employed. CyberKnife and TomoTherapy are both optimal RS devices, the choice to use one over another has to be clinically guided.
Similar content being viewed by others
References
Eichler AF, Loeffler JS (2007) Multidisciplinary management of brain metastases. Oncologist 12(7):884–898
Sperduto PW, Chao ST, Sneed PK et al (2010) Diagnosis-specific prognostic factors, indexes, and treatment outcomes for patients with newly diagnosed brain metastases: a multi-institutional analysis of 4259 patients. Int J Radiat Oncol Biol Phys 77:655–661
Aoyama H, Shirato H, Tago M et al (2006) Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. JAMA 295:2483–2491
Kocher M, Soffietti R, Abacioglu U et al (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol 29:134–141
Tsao MN, Rades D, Wirth A et al (2012) Radiotherapeutic and surgical management for newly diagnosed brain metastasis (es): an American Society for Radiation Oncology evidence-based guideline. Pract Radiat Oncol 2:210–225
Brown PD, Brown CA, Pollock BE et al (2002) Stereotactic radiosurgery for patients with “radioresistant”brain metastases. Neurosurgery 51:656–665
Chang EL, Selek U, Hassenbusch SJIII et al (2005) Outcome variation among so called “radioresistant” brain metastases treated with stereotactic radiosurgery. Neurosurgery 56:936–945
Yamamoto M, Serizawa T, Shuto T et al (2014) Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901):a multi institutional prospective observational study. Lancet Oncol 15(4):387–395
Soisson ET, Hoban PW, Kammeyer T et al (2011) A technique for stereotactic radiosurgery treatment planning with helical TomoTherapy. Med Dosim 36(1):46–56
Baumert BG, Rutten I, Dehing-Oberije C et al (2006) A pathology-based substrate for target definition in radiosurgery of brain metastases. Int J Radiat Oncol Biol Phys 66(1):187–194
Hoogeman MS, Nuyttens JJ, Levenda G et al (2008) Time dependence of intrafraction patient motion assessed by repeat stereoscopic imaging. Int J Radiat Oncol Biol Phys 70:1313–1319
Drabick DM, MacKenzie MA, Fallone GB (2007) Quantifying appropriate PTV setup margins: analysis of patient setup fidelity and intrafraction motion using post-treatment megavoltage computed tomography scans. Int J Radiat Oncol Biol Phys 68:1222–1228
Shaw E, Scott C, Souhami L et al (2000) Single dose radiosurgical treatment of recurrent previously irradiated primary brain tumors and brainmetastases: final report of RTOG protocol 90-05. Int J Radiat Oncol Biol Phys 47(2):291–298
Gladwish A, Oliver M, Craig J et al (2007) Segmentation ad leaf sequencing for intensity modulated arc therapy. Med Phys 34(5):1779–1788
Lomax NJ, Scheib SG (2003) Quantifying the degree of conformity in radiosurgery treatment panning. Int J Radiat Oncol Biol Phys 55(5):1409–1419
Kubo HD, Wilder RB, Pappas CTE (1999) Impact of collimator leaf width on stereotactic radiosurgery and 3D conformal radiotherapy treatment plans. Int J Radiat Oncol Biol Phys 44:937–945
Han C, Liu A, Schultheiss TE et al (2006) Dosimetric comparisons of Helical Tomotherapy treatment plans and step-and-shoot intensity modulated radiosurgery treatment plans in intracranial stereotactic radiosurgery. Int J Radiat Oncol Biol Phys 65(2):608–616
Bauman G, Yartsev S, Fisher B et al (2007) Simultaneous infield boost with helical TomoTherapy for patients with 1 to 3 brain metastases. Am J Clin Oncol 30(1):38–40
McGuinnes CM, Gottschalk AR, Lessard E et al (2015) Investigating the clinical advantages of a robotic linac equipped with a multileaf collimator in the treatment of brain and prostate cancer patients. J Appl Clin Med Phys 16(5):284–295
Nakamura JL, Verhey LJ, Smith V et al (2001) Dose conformity of gamma knife radiosurgery and risk factors for complications. Int J Radiat Oncol Biol Phys 51(5):1313–1319
Leith JT, Cook S, Chougule P et al (1994) Intrinsic and extrinsic characteristics of human tumors relevant to radiosurgery: comparative cellular radiosensitivity and hypoxic percentages. Acta Neurochir Suppl 62:18–27
Tome W, Fowler J (2000) Selective boosting of tumor sub-volumes. Int J Radiat Oncol Biol Phys 48(2):593–599
Kohutek ZA, Yamada Y, Chan TA et al (2015) Long-term risk of radionecrosis and imaging changes after stereotactic radiosurgery for brain metastases. J Neurooncol 125(1):149–156
Flickinger JC, Lunsford LD, Kondziolka D et al (1992) Radiosurgery and brain tolerance: an analysis of neurodiagnostic imaging changes after gamma knife radiosurgery for arteriovenous malformations. Int J Radiat Oncol Biol Phys 23:19–26
Blonigen BJ, Steinmetz RD, Levin L et al (2010) Irradiated volume as a predictor of brain radionecrosis after linear accelerator stereotactic radiosurgery. Int J Radiat Oncol Biol Phys 77:996–1001
Minniti G, Clarke E, Lanzetta G et al (2011) Stereotactic radiosurgery for brain metastases: analysis of outcome and risk of brain radionecrosis. Radiat Oncol 6:48
Acknowledgements
The abstract was presented in the poster session of the ESTRO 33 Congress, 4–8 April 2014, Barcelona, Spain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Funding
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Greto, D., Pallotta, S., Masi, L. et al. A dosimetric comparison between CyberKnife and tomotherapy treatment plans for single brain metastasis. Radiol med 122, 392–397 (2017). https://doi.org/10.1007/s11547-017-0735-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-017-0735-9