Abstract
Purpose
Fibroblast activation protein-α (FAPα) is uniquely expressed in activated fibroblasts, including cancer-associated fibroblasts that populate tumor stroma and contribute to proliferation and immunosuppression. Radiolabeled FAPα inhibitors enable imaging of multiple human cancers, but time-dependent clearance from tumors currently limits their utility as FAPα-targeted radiotherapeutics. We sought to increase the area under the curve (AUC) by constructing a trifunctional ligand that binds FAPα with high affinity and also binds albumin and theranostic radiometals.
Procedures
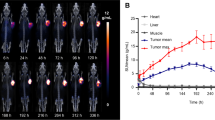
RPS-309 comprised a FAPα-targeting moiety, an albumin-binding group, and 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA). Inhibition of recombinant human FAPα (rhFAPα) was determined by colorimetric assay. Affinity for human serum albumin (HSA) was determined by high-performance affinity chromatography. The tissue distribution of [68Ga]Ga-RPS-309 in SW872 tumor xenograft-bearing mice was imaged by microPET/CT and quantified by biodistribution studies performed from 30 min to 3 h post injection (p.i.). The biodistribution of [177Lu]Lu-RPS-309 was determined at 4, 24, and 96 h p.i.
Results
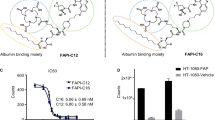
RPS-309 inhibits rhFAPα with IC50 = 7.3 ± 1.4 nM. [68Ga]Ga-RPS-309 is taken up specifically by FAPα-expressing cells and binds HSA with Kd = 4.6 ± 0.1 μM. Uptake of the radiolabeled ligand in tumors was evident from 30 min p.i. (> 5 %ID/g) and was significantly reduced by co-injection of RPS-309. Specific skeletal uptake was also observed. Activity in tumors was constant through 4 h p.i., but cleared significantly by 24 h. The AUC in this period was 127 (%ID/g) × h.
Conclusions
RPS-309 is a high-affinity FAPα inhibitor with prolonged plasma residence. Introduction of the albumin-binding group did not compromise FAPα binding. Although initial tumor uptake was high and FAPα-specific, RPS-309 also progressively cleared from tumors. Nevertheless, RPS-309 incorporates multiple sites in which structural diversity can be introduced, and therefore serves as a platform for future structure-activity relationship studies.





Similar content being viewed by others
References
Liu T, Zhou L, Li D, Andl T, Zhang Y (2019) Cancer-Associated Fibroblast Build and Secure the Tumor Microenvironment. Front Cell Dev Biol 7:60
de Sostoa J, Fajardo CA, Moreno R, Ramos MD, Farrera-Sal M, Alemany R (2019) Targeting the tumor stroma with an oncolytic adenovirus secreting a fibroblast activation protein-targeted bispecific T-cell engager. J ImmunoTherapy. Cancer 7:19
Broekgaarden M, Anbil S, Bulin AL et al (2019) Modulation of redox metabolism negates cancer-associated fibroblasts-induced treatment resistance in a heterotypic 3D culture platform of pancreatic cancer. Biomaterials 222:119421
Kraman M, Bambrough PJ, Arnold JN et al (2010) Suppression of Antitumor Activity Immunity by Stromal Cells Expressing Fibroblast Activation Protein-α. Science 330:827–830
Dvořáková P, Bušek P, Knedlik T et al (2017) Inhibitor-Decorated Polymer Conjugates Targeting Fibroblast Activation Protein. J Med Chem 60:8385–8393
Acharya PS, Zukas A, Chandan V, Katzenstein ALA, Puré E (2006) Fibroblast activation protein: a serine protease expressed at the remodeling surface in idiopathic pulmonary fibrosis. Hum Pathol 37:352–360
Croft AP, Campos J, Jansen K et al (2019) Distinct fibroblast subsets drive inflammation and damage in arthritis. Nature 570:246–251
Laverman P, van der Geest T, Terry SYA et al (2015) Immuno-PET and Immuno-SPECT of Rheumatoid Arthritis with Radiolabeled Anti-Fibroblast Activation Protein Antibody Correlates with Severity of Arthritis. J Nucl Med 56:778–783
Levy MT, McCaughan GW, Marinos G, Gorrell MD (2002) Intrahepatic expression of the hepatic stellate cell marker fibroblast activation protein correlates with the degree of fibrosis in hepatitis C virus infection. Liver 22:93–101
Kalluri R (2016) The biology and function of fibroblasts in cancer. Nat Rev Cancer 16:582–598
Tillmanns J, Hoffmann D, Habbaba Y et al (2015) Fibroblast activation protein alpha expression identifies activated fibroblasts after myocardial infarction. J Mol Cell Cardiol 87:194–203
Park JE, Lenter MC, Zimmerman RN, Garin-Chesa P, Old LJ, Rettig WJ (1999) Fibroblast activation protein, a dual specificity serine protease expressed in reactive human tumor stromal fibroblasts. J Biol Chem 274:36505–36512
Henry LR, Lee H-O, Lee JS et al (2007) Clinical Implications of Fibroblast Activation Protein in Patients with Colon Cancer. Clin Cancer Res 13:1736–1741
Cohen SJ, Alpaugh RK, Palazzo I et al (2008) Fibroblast Activation Protein and Its Relationship to Clinical Outcome in Pancreatic Adenocarcinoma. Pancreas 37:154–158
Puré E, Blomberg R (2018) Pro-tumorigenic roles of fibroblast activation protein in cancer: back to the basics. Oncogene 37:4343–4357
Kratochwil C, Flechsig P, Lindner T et al (2019) 68Ga-FAPI PET/CT: Tracer Uptake in 28 Different Kinds of Cancer. J Nucl Med 60:801–805
Giesel FL, Kratochwil C, Lindner T et al (2019) 68Ga-FAPI PET/CT: Biodistribution and Preliminary Dosimetry Estimate of 2 DOTA-Containing FAP-Targeting Agents in Patients with Various Cancers. J Nucl Med 60:386–392
Lindner T, Loktev A, Altmann A et al (2018) Development of Quinoline-Based Theranostic Ligands for the Targeting of Fibroblast Activation Protein. J Nucl Med 59:1415–1422
Müller C, Struthers H, Winiger C, Zhernosekov K, Schibli R (2013) DOTA Conjugate With an Albumin-Binding Entity Enables the First Folic Acid-Targeted 177Lu-radionuclide Tumor Therapy in Mice. J Nucl Med 54:124–131
Farkas R, Siwowska K, Ametamey SM, Schibli R, van der Muelen NP, Müller C (2016) (64)Cu- and (68)Ga-Based PET Imaging of Folate Receptor-Positive Tumors: Development and Evaluation of an Albumin-Binding NODAGA-Folate. Mol Pharm 13:1979–1987
Kelly JM, Amor-Coarasa A, Nikolopoulou A et al (2017) Dual-Target Binding Ligands with Modulated Pharmacokinetics for Endoradiotherapy of Prostate Cancer. J Nucl Med 58:1442–1449
Kelly J, Amor-Coarasa A, Ponnala S et al (2018) Trifunctional PSMA-targeting constructs for prostate cancer with unprecedented localization to LNCaP tumors. Eur J Nucl Med Mol Imaging 45:1841–1851
Kelly JM, Amor-Coarasa A, Ponnala S et al (2019) Albumin-Binding PSMA Ligands: Implications for Expanding the Therapeutic Window. J Nucl Med 60:656–663
Lindner T, Loktev A, Giesel F, Kratochwil C, Altmann A, Haberkorn U (2019) Targeting of activated fibroblasts for imaging and therapy. EJNMMI Radiopharm Chem 4:16
Loktev A, Lindner T, Mier W et al (2018) A Tumor-Imaging Method Targeting Cancer-Associated Fibroblasts. J Nucl Med 59:1423–1429
Loktev A, Lindner T, Burger EM et al (2019) Development of Fibroblast Activation Protein-Targeted Radiotracers with Improved Tumor Retention. J Nucl Med 60:1421–1429
Busek P, Mateu R, Zubal M, Kotackova L, Sedo A (2018) Targeting Fibroblast Activation in Cancer - Prospects and Caveats. Front Biosci 23:1933–1968
Tsai TY, Yeh TK, Chen X et al (2010) Substituted 4-Carboxymethylpyroglutamic Acid Diamides as Potent and Selective Inhibitors of Fibroblast Activation Protein. J Med Chem 53:6572–6583
Ryabtsova O, Jansen K, Goethem SV et al (2012) Acylated Gly-(2-cyano)pyrrolidines as inhibitors of fibroblast activation protein (FAP) and the issue of FAP/prolyl oligopeptidase (PREP)-selectivity. Bioorg Med Chem Lett 22:3412–3417
Poplawski SE, Lai JH, Li Y et al (2013) Identification of Selective and Potent Inhibitors of Fibroblast Activation Protein and Prolyl Oligopeptidase. J Med Chem 56:3467–3477
Jansen K, Heirbaut L, Cheng JD et al (2013) Selective Inhibitors of Fibroblast Activation Protein (FAP) with a (4-Quinolinoyl)-glycyl-2-cyanopyrrolidine Scaffold. ACS Med Chem Lett 4:491–496
Jansen K, Heirbaut L, Verkerk R et al (2014) Extended Structure-Activity Relationship and Pharmacokinetic Investigation of (4-Quinolinoyl)glycyl-2-cyanopyrrolidine Inhibitors of Fibroblast Activation Protein (FAP). J Med Chem 57:3053–3074
Watabe T, Liu Y, Kaneda-Nakashima K et al (2020) Theranostics Targeting Fibroblast Activation Protein in the Tumor Stroma: 64Cu- and 225Ac-Labeled FAPI-04 in Pancreatic Cancer Xenograft Mouse Models. J Nucl Med 61:563–569
Kuo H-T, Merkens H, Zhang Z et al (2018) Enhancing Treatment Efficacy of 177Lu-PSMA-617 with the Conjugation of an Albumin-Binding Motif: Preclinical Dosimetry and Endoradiotherapy Studies. Mol Pharm 15:5183–5191
Benešová M, Umbricht CA, Schibli R, Müller C (2018) Albumin-Binding PSMA Ligands: Optimization of the Tissue Distribution Profile. Mol Pharm 15:934–946
Umbricht CA, Benešová M, Schibli R, Müller C (2018) Preclinical Development of Novel PSMA-Targeting Radioligands: Modulation of Albumin-Binding Properties To Improve Prostate Cancer Therapy. Mol Pharm 15:2297–2306
Wang Z, Jacobson O, Tian R et al (2018) Radioligand Therapy of Prostate Cancer with a Long-Lasting Prostate-Specific Membrane Antigen Targeting Agent 90Y-DOTA-EB-MCG. Bioconjug Chem 29:2309–2315
Kramer V, Fernández R, Lehnert W et al (2020) Biodistribution and dosimetry of a single dose of albumin-binding ligand [177Lu]Lu-PSMA-ALB-56 in patients with mCRPC. Eur J Nucl Med Mol Imaging. https://doi.org/10.1007/s00259-020-05022-3
Zang J, Fan X, Wang H et al (2019) First-in-human study of 177Lu-EB-PSMA-617 in patients with metastatic castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging 46:148–158
Gamache RF, Zettlitz KA, Tsai W-TK, Collins J, Wu AM, Murphy JM (2020) Tri-functional platform for construction of modular antibody fragments for in vivo18F-PET or NIRF molecular imaging. Chem Sci 11:1832–1838
Plescia J, Cesco SD, Patrascu MB et al (2019) Integrated Synthetic, Biophysical, and Computational Investigations of Covalent Inhibitors of Prolyl Oligopeptidase and Fibroblast Activation Protein α. J Med Chem 62:7874–7884
Fischer E, Chaitanya K, Wüest T et al (2012) Radioimmunotherapy of Fibroblast Activation Protein Positive Tumors by Rapidly Internalizing Antibodies. Clin Cancer Res 18:6208–6218
Kong F, Pang X, Zhao J et al (2019) Hydrolytic Metabolism of Cyanopyrrolidine DPP-4 Inhibitors Mediated by Dipeptidyl Peptidases. Drug Metab Dispos 47:238–248
van der Geest T, Roeleveld DM, Walgreen B et al (2018) Imaging fibroblast activation protein to monitor therapeutic effects of neutralizing interleukin-22 in collagen-induced arthritis. Rheumatology 57:737–747
Muliaditan T, Caron J, Okesola M et al (2018) Macrophages are exploited from an innate wound healing response to facilitate cancer metastasis. Nat Commun 9:2951
Egger C, Cannet C, Gérard C et al (2017) Effects of the fibroblast activation protein inhibitor, PT100, in a murine model of pulmonary fibrosis. Eur J Pharmacol 809:64–72
Fu X, Khalil H, Kanisicak O et al (2018) Specialized Fibroblast Differentiated States Underlie Scar Formation in the Infarcted Mouse Heart. J Clin Invest 128:2127–2143
Ma Y, Iyer RP, Jung M, Czubryt MP, Lindsey ML (2017) Cardiac Fibroblast Activation Post-Myocardial Infarction: Current Knowledge Gaps. Trends Pharmacol Sci 38:448–458
Terry SYA, Koenders MI, Franssen GM et al (2016) Monitoring Therapy Response of Experimental Arthritis with Radiolabeled Tracers Targeting Fibroblasts, Macrophages, or Integrin αvβ3. J Nucl Med 57:467–472
Varasteh Z, Mohanta S, Robu S et al (2019) Molecular Imaging of Fibroblast Activity After Myocardial Infarction Using a 68Ga-Labeled Fibroblast Activation Protein Inhibitor, FAPI-04. J Nucl Med 60:1743–1749
Acknowledgments
The authors wish to thank Prof. Dr. Uwe Haberkorn for providing the HT-1080-FAP and HT-1080 cells and to acknowledge Dr. J. David Warren for assistance with compound purification and analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
James M. Kelly, Shashikanth Ponnala, and John W. Babich hold equity in Noria Therapeutics, Inc, which has licensed intellectual property rights related to compounds described in this manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 1828 kb)
Rights and permissions
About this article
Cite this article
Kelly, J.M., Jeitner, T.M., Ponnala, S. et al. A Trifunctional Theranostic Ligand Targeting Fibroblast Activation Protein-α (FAPα). Mol Imaging Biol 23, 686–696 (2021). https://doi.org/10.1007/s11307-021-01593-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-021-01593-1