Abstract
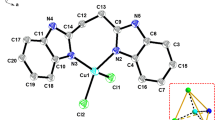
Divalent metal complexes of general formula [M(2-nb)2(mc)2].2(2-nbH), where M = Co(II), Ni(II), Cu(II) or Zn(II), 2-nbH = 2-nitrobenzoic acid and mc = methyl carbazate (NH2NHCOOCH3), have been prepared and characterized by physicochemical and spectroscopic methods. Single-crystal X-ray study of the Cu(II) complex revealed that the molecule is centrosymmetric, with two N,O-chelating mc ligands in equatorial positions and a pair of monodentate 2-nb anions in the axial positions. The lattice 2-nbH molecules help to establish the packing of monomers through hydrogen-bonding interactions. Thermal stability and reactivity of the complexes were studied by TG–DTA. Emission studies show that these complexes are fluorescent.



Similar content being viewed by others
References
Wieghardt K (1989) Angew Chem Int Ed (Engl) 28:1153
Pecoraro VL, Baldwin MJ, Gelasco A (1994) Chem Rev 94:807
Rane KS, Verenkar VMS (2001) Bull Mater Sci 24:39
Edwards DA, Keily JF, Mahon MF, Molloy KC and Thompsett D (1993) J Chem Soc Dalton Trans 3471 and references therein
Patil KC, Soundararajan R, Goldberg EP (1983) Synth React Inorg Met Org Chem 13:29
Ravindranathan P, Patil KC (1985) Proc Indian Acad Sci (Chem Sci) 95:345
Mahesh GV, Ravindranathan R, Patil KC (1986) Proc Indian Acad Sci (Chem Sci) 97:117
Tey SL, Reddy MV, Subba Rao GV, Chowdari BVR, Yi J, Ding J, Vital JJ (2006) Chem Mater 18:1587
Ekambaram S, Patil KC, Maaza M (2005) J Alloys Compd 393:81
Patil KC, Aruna ST, Ekambaram S (1997) Curr Opin Mater Sci 2:158 References therein
Ma G, Zhang T, Yu K (2005) J Braz Chem Soc 16:796
Song JC, Zhang TL, Zhang JG, Ma GX, Li YF, Yu KB (2003) Acta Chim Sin 61:1444
Li GX (2002) Chin J Fine Chem 19:336
Dong GJ, Wang ZP, Zhang ML (2002) Chin J Applied Sci Tech 29:54
Jiang Q (2000) Chin J Petroleum Chem 29:144
Premkumar T, Govindarajan S, Xie R, Pan WP (2003) J Therm Anal Calorim 74:325
Premkumar T, Govindarajan S (2005) J Therm Anal Calorim 79:115
Premkumar T, Govindarajan S (2006) J Therm Anal Calorim 84:395
Kuppusamy K, Govindarajan S (1996) Thermochim Acta 279:143
Baruah AM, Karmakar A, Baruah JB (2008) Inorg Chim Acta 361:2777
Karmakar A, Sarma RJ, Baruah JB (2007) Eur J Inorg Chem 5:643
Karmakar A, Bania K, Baruah AM, Baruah JB (2007) Inorg Chem Commun 10:959
Stachova P, Valigura D, Koman M, Melnık M, Korabik M, Mrozinski J, Glowiak T (2004) Polyhedron 23:1303
Stachova P, Korabik M, Koman M, Melnık M, Mrozinski J, Glowiak T, Mazur M, Valigura D (2006) Inorg Chim Acta 359:1275
Stachova P, Melnık M, Korabik M, Mrozinski J, Koman M, Glowiak T, Valigura D (2007) Inorg Chim Acta 360:1517
Kavalirova J, Korabik M, Stachova P, Moncol J, Sillanpaa R, Lis T, Miklos D, Melnık M, Mrozinski J, Valigura D (2008) Polyhedron 27:1333
Vaskova Z, Padelkova Z, Mazur M, Valigura D, Moncol J (2011) Transition Met Chem 36:883
Srinivasan BR, Sawant SC (2003) Thermochim Acta 402:45
Vogel AI (1961) A textbook of quantitative inorganic analysis including elementary instrumental analysis, 3rd edn. Longman, London
Blessing RH (1995) Acta Cryst A 51:33
Sheldrick GM (1997) SHELXS-97 and SHELXL-97. University of Göttingen, Göttingen
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kathiresan, A., Srinivasan, K., Brinda, S. et al. Synthesis and characterization of cobalt(II), nickel(II), copper(II) and zinc(II) complexes of 2-nitrobenzoic acid with methyl carbazate as ancillary ligand. Crystal structure of the copper(II) complex. Transition Met Chem 37, 393–397 (2012). https://doi.org/10.1007/s11243-012-9601-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-012-9601-8