Abstract
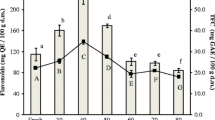
The aim of this study was to assess the efficiency of different drying methods (room, sun, oven, microwave, cross-flow, infra-red, dehumidifier, and freeze-drying) on maximum retention of the nutritional and bioactive compounds profiling of Talinum triangulare, which is a less-known perishable leafy-vegetable. The evaluation of various drying methods is to learn the best appropriate strategy for a post-harvest drying method for retaining all the potential benefits with minor loss. Microwave and freeze-dried samples held the maximum ascorbic acid quantified by HPLC with 1.36 and 1.11 g/100 g DW, respectively. The main carotenoid compounds identified were violaxanthin, lutein, zeaxanthin, β-carotene isomers, trans-β-carotene, and cis-β-carotenes. Gallic, protocatechuic, catechin, para-coumaric, ferulic, rutin, trans-cinnamic, and quercetin are the significant phenolics and flavonoids identified and quantified by liquid chromatography. The efficiency of different solvents on bioactive extractions uncovered that the methanol and 80% aqueous ethanol were good for retention of total phenolics, total flavonoids, and antioxidant compounds, which was affirmed through phosphomolybdate, DPPH, and FRAP assays. Dried T. triangulare foliage could be productively utilized as a promising raw material for food and pharma businesses because of its rich bioactive composition.




Similar content being viewed by others
References
Gupta S, Lakshmi AJ, Manjunath MN, Prakash J (2005) Analysis of nutrient and anti-nutrient content of underutilized green leafy vegetables. LWT-Food Sci Technol 38:339–345. https://doi.org/10.1016/j.lwt.2004.06.012
Gupta S, Prakash J (2009) Studies on Indian green leafy vegetables for their antioxidant activity. Plant Foods Hum Nutr 64:39–45. https://doi.org/10.1007/s11130-008-0096-6
Ezekwe CI, Uzomba CR, Ugwu OPC (2013) The effect of methanol extract of Talinum triangulare (water leaf) on the hematology and some liver parameters of experimental rats. Glob J Biotechnol Biochem 8:51–60. https://doi.org/10.5829/idosi.gjbb.2013.8.2.1109
Fontem DA, Schippers RR (2010) Talinum triangulare (Jacq.) Willd [internet] record from Protabase: Grubben GJH, Denton OA (editors). PROTA (Plant Resources of Tropical Africa/Ressources végétales de l’Afrique tropicale), Wageningen, Netherlands
Ogbonnaya EC, Chinedum EK (2013) Vitamin and carotenoid composition of raw and decoctions of water leaf (Talinum triangulare). Biochem Pharmacol 2:1–3. https://doi.org/10.4172/2167-0501.1000121
Yang RY, Tsou SC, Lee TC, Wu WJ, Hanson PM, Kuo G, Lai PY (2006) Distribution of 127 edible plant species for antioxidant activities by two assays. J Sci Food Agric 86:2395–2403. https://doi.org/10.1002/jsfa.2630
Ezekwe MO, Besong SA, Igbokwe PE, Ezekwe EI (2002) Beneficial influence of Purslane and waterleaf supplementation on cardiovascular disease risk in humans. FASEB J 16:A639–A639
Ibeawuchi II, Nwufo MI, Oti NN, Opara CC, Eshett ET (2007) Productivity of intercropped green (Amaranthus cruentus)/waterleaf (Talinum triangulare) with poultry manure rates in southeastern Nigeria. Plant Sci 2:222–227. https://doi.org/10.3923/jps.2007.222.227
Swarna J, Ravindhran R (2013) Pharmacognostical and phytochemical evaluation of Talinum triangulare (Jacq.) Willd. Int J Pharm Pharm Sci 2:249
AOCS (2003) Official methods and recommended practices of the American Oil Chemist’s Society. In: Moisture content: Da 2a-48, 5th edn. AOCS Press, Champaign
Vega-Galvez A, Di Scala K, Rodriguez K, Lemus-Mondaca R, Miranda M, López J, Pérez-Won M (2009) Effect of air-drying temperature on physico-chemical properties, antioxidant capacity, colour and total phenolic content of red pepper (Capsicum annuum, L. var. Hungarian). Food Chem 117:647–653. https://doi.org/10.1016/j.foodchem.2009.04.066
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1. https://doi.org/10.1104/pp.24.1.1
Lichtenthaler HK (1996) Vegetation stress: an introduction to the stress concept in plants. J Plant Physiol 148:4–14. https://doi.org/10.1016/S0176-1617(96)80287-2
Vanderslice JT, Higgs DJ, Hayes JM, Block G (1990) Ascorbic acid and dehydroascorbic acid content of foods-as-eaten. J Food Compos Anal 3:105–118. https://doi.org/10.1016/0889-1575(90)90018-H
Simon DP, Anila N, Gayathri K, Sarada R (2016) Heterologous expression of β-carotene hydroxylase in Dunaliella salina by agrobacterium-mediated genetic transformation. Algal Res 18:257–265. https://doi.org/10.1016/j.algal.2016.06.017
Kumar SS, Manoj P, Shetty NP, Giridhar P (2015) Effect of different drying methods on chlorophyll, ascorbic acid and antioxidant compounds retention of leaves of Hibiscus sabdariffa L. J Sci Food Agric 95:1812–1820. https://doi.org/10.1002/jsfa.6879
Sadasivam S, Manickam A (2008) Biochemical methods, 8th edn. New Age International Publisher, New Delhi
Djeridane A, Yousfi M, Nadjemi B, Boutassouna D, Stocker P, Vidal N (2006) Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem 97:654–660. https://doi.org/10.1016/j.foodchem.2005.04.028
Burin VM, Arcari SG, Bordignon-Luiz AMT, Costa LLF (2011) Determination of some phenolic compounds in red wine by RP-HPLC: method development and validation. J Chromatogr Sci 49:647–651. https://doi.org/10.1093/chrsci/49.8.647
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269:337–341. https://doi.org/10.1006/abio.1999.4019
Khan MI, Harsha PS, Giridhar P, Ravishankar GA (2012) Pigment identification, nutritional composition, bioactivity, and in vitro cancer cell cytotoxicity of Rivina humilis L. berries, potential source of betalains. LWT-Food Sci Technol 47:315–323. https://doi.org/10.1016/j.lwt.2012.01.025
Oyiazu M (1986) Studies on products of browning reactions: anti-oxidative activities of products of browning reaction prepared from glucosamine. Jpn J Nutr 44:307–315. https://doi.org/10.5264/eiyogakuzashi.44.307
Saini RK, Shetty NP, Prakash M, Giridhar P (2014) Effect of dehydration methods on retention of carotenoids, tocopherols, ascorbic acid and antioxidant activity in Moringa oleifera leaves and preparation of a RTE product. J Food Sci Technol 51:2176–2182. https://doi.org/10.1007/s13197-014-1264-3
Addo AA (2004) Seasonal availability of dietary ascorbic acid and incidence of scurvy in northern state of Nigeria. PhD Thesis, Department of Biochemistry, Ahmadu Bello University, Zaria
Van Duyne FO, Bruckart SM, Chase JT, Simpson JI (1945) Ascorbic acid content of freshly harvested vegetables. J Am Diet Assoc 21:153–154. https://www.cabdirect.org/cabdirect/abstract/19451400391
Raju M, Varakumar S, Lakshminarayana R, Krishnakantha TP, Baskaran V (2007) Carotenoid composition and vitamin a activity of medicinally important green leafy vegetables. Food Chem 101:1598–1605. https://doi.org/10.1016/j.foodchem.2006.04.015
Khoo HE, Prasad KN, Kong KW, Jiang Y, Ismail A (2011) Carotenoids and their isomers: color pigments in fruits and vegetables. Molecules 16:1710–1738. https://doi.org/10.3390/molecules16021710
Daniel DL, Huerta BB, Sosa IA, Mendoza MV (2012) Effect of fixed bed drying on the retention of phenolic compounds, anthocyanins and antioxidant activity of roselle (Hibiscus sabdariffa L.). Ind Crop Prod 40:268–276. https://doi.org/10.1016/j.indcrop.2012.03.015
Eleazu CO, Eleazu KC (2013) Bioactive constituents and in vitro antioxidant capacity of water leaf (Talinum triangulare) as affected by domestic cooking. Eur J Med Plants 3:540
Acknowledgements
The author (SSK) is grateful to CSIR New Delhi for the award of SRF fellowship and Department of Biotechnology (BT/PR16902/NER/95/422/2015), Government of India, New Delhi, for financial assistance. We greatly acknowledge the Director, CSIR-CFTRI, for his kind support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Human and Animal Participants
This article does not contain any studies with human or animal subjects.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumar, S.S., Arya, M., Nagbhushan, P. et al. Evaluation of Various Drying Methods on Bioactives, Ascorbic Acid and Antioxidant Potentials of Talinum triangulare L., foliage. Plant Foods Hum Nutr 75, 283–291 (2020). https://doi.org/10.1007/s11130-020-00804-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-020-00804-4