Abstract
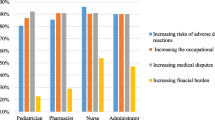
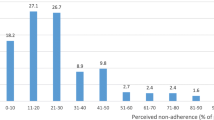
Objective To assess current experiences and attitudes of hospital based paediatricians towards off-label medicine prescribing. Setting Paediatric hospital wards and out-patient clinics. Design A prospective, questionnaire based study. Results A 30 item questionnaire was sent to 300 hospital based paediatricians and 250 (83%) were returned completed. Over 69% of responders were familiar with the term off-label medicines. However, only 28% were knowingly prescribing off-label medicines to children. The majority of respondents (90%) expressed concerns about the safety and efficacy of off-label medicines. Only 15% had observed Adverse Drug Reactions, and 31% a treatment failure. The vast majority of respondents (83%) did not obtain informed consent or tell parents they were prescribing off label medicines to their children. Conclusions Off-label prescribing of medicines to children is a familiar concept to the majority of paediatricians in Jordan although only a smaller number are aware that it is common in their practice. Respondents showed concern about off label prescribing, although the majority do not consider it necessary to inform parents. More comprehensive research is needed in this area in Jordan and other Middle Eastern countries.


Similar content being viewed by others
References
Budetti PP. Ensuring safe and effective medications for children. JAMA. 2003;290(7):950–1.
Pandolfini C, Bonati M. A literature review on off-label drug use in children. Eur J Pediatr. 2005;164(9):552–8.
Conroy S, Choonara I, Impicciatore P, Mohn A, Arnell H, Rane A, et al. Survey of unlicensed and off label drug use in paediatric wards in European countries. ENDIC BMJ. 2000;320(7227):79–82.
Turner S, Longworth A, Nunn AJ, Choonara I. Unlicensed and off label drug use in paediatric wards: prospective study. BMJ. 1998;316(7128):343–5.
Ekins-Daukes S, Helms PJ, Taylor MW, McLay JS. Off-label prescribing to children: attitudes and experience of general practitioners. Br J Clin Pharmacol. 2005;60(2):145–9.
McLay JS, Tanaka M, Ekins-Daukes S, Helms PJ. A prospective questionnaire assessment of attitudes and experiences of off label prescribing among hospital based paediatricians. Arch Dis Child. 2006;91(7):584–7.
Impicciatore P, Choonara I, Clarkson A, Provasi D, Pandolfini C, Bonati M. Incidence of adverse drug reactions in paediatric in/out-patients: a systematic review and meta-analysis of prospective studies. Br J Clin Pharmacol. 2001;52(1):77–83.
Wong I, Sweis D, Cope J, Florence A. Paediatric medicines research in the UK: how to move forward? Drug Saf. 2003;26(8):529–37.
Choonara I, Conroy S. Unlicensed and off-label drug use in children: implications for safety. Drug Saf. 2002;25(1):1–5.
Turner S, Nunn AJ, Fielding K, Choonara I. Adverse drug reactions to unlicensed and off-label drugs on paediatric wards: a prospective study. Acta Paediatr. 1999;88(9):965–8.
Horen B, Montastruc JL, Lapeyre-Mestre M. Adverse drug reactions and off-label drug use in paediatric outpatients. Br J Clin Pharmacol. 2002;54(6):665–70.
Mukattash T, Hawwa AF, Trew K, McElnay JC. Healthcare professional experiences and attitudes on unlicensed/off-label paediatric prescribing and paediatric clinical trials. Eur J Clin Pharmacol 2011;67(5):449–61.
Stewart D, Rouf A, Snaith A, Elliott K, Helms PJ, McLay JS. Attitudes and experiences of community pharmacists towards paediatric off-label prescribing: a prospective survey. Br J Clin Pharmacol. 2007;64(1):90–5.
Mukattash TL, Millership JS, Collier PS, McElnay JC. Public awareness and views on unlicensed use of medicines in children. Br J Clin Pharmacol. 2008;66(6):838–45.
CIA Wold Factbook/Countries of the World/Jordan. Can be retrieved from URL: http://www.theodora.com/wfbcurrent/jordan/jordan_people.html Accessed 29 June 2011.
Conroy S, McIntyre J. The use of unlicensed and off-label medicines in the neonate. Semin Fetal Neonatal Med. 2005;10(2):115–22.
Marchetti F, Bua J, Ventura A, Notarangelo LD, Di Maio S, Migliore G, Bonati M. The awareness among paediatricians of off-label prescribing in children: a survey of Italian hospitals. Eur J Clin Pharmacol. 2007;63(1):81–5.
Jordan Food and Drug Administration (JFDA). Available at: http://www.jfda.jo/en/default/ Accessed July 2011.
Nunn AJ. Making medicines that children can take. Arch Dis Child. 2003;88(5):369–71.
Schirm E, Tobi H, de Vries TW, Choonara I, Jong-van De, den Berg LT. Lack of appropriate formulations of medicines for children in the community. Acta Paediatr. 2003;92(12):1486–9.
Bajcetic M, Jelisavcic M, Mitrovic J, Divac N, Simeunovic S, Samardzic R, Gorodischer R. Off label and unlicensed drugs use in paediatric cardiology. Eur J Clin Pharmacol. 2005;61(10):775–9.
Pasquali SK, Hall M, Slonim AD, Jenkins KJ, Marino BS, Cohen MS, et al. Off-label use of cardiovascular medications in children hospitalized with congenital and acquired heart disease. Circ Cardiovasc Qual Outcomes. 2008;1(2):74–83.
Porta A, Esposito S, Menson E, Spyridis N, Tsolia M, Sharland M, et al. Off-label antibiotic use in children in three European countries. Eur J Clin Pharmacol. 2010;66(9):919–27.
Ceci A, Felisi M, Catapano M, Baiardi P, Cipollina L, Ravera S, Bagnulo S, Reggio S, Rondini G. Medicines for children licensed by the European Agency for the Evaluation of Medicinal Products. Eur J Clin Pharmacol. 2002;58(8):495–500.
Al-Wazaify M, Matowe L, Albsoul-Younes A, Al-Omran OA. Pharmacy education in Jordan, Saudi Arabia, and Kuwait. Am J Pharm Educ. 2006;70(1):18.
Guadagnoli E, Velicer WF. Relation to sample size to the stability of component patterns. Psychol Bull. 1988;103(2):265–75.
Acknowledgments
All the paediatricians who participated in this study.
Funding
No external funding was obtained for this study.
Conflicts of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix



Rights and permissions
About this article
Cite this article
Mukattash, T.L., Wazaify, M., Khuri-Boulos, N. et al. Perceptions and attitudes of Jordanian paediatricians towards off-label paediatric prescribing. Int J Clin Pharm 33, 964–973 (2011). https://doi.org/10.1007/s11096-011-9569-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-011-9569-z