Abstract
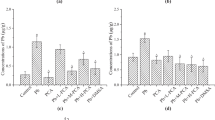
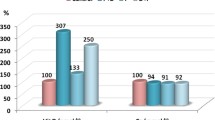
Epidemiological evidence has shown higher susceptibility of Children to the adverse effects of lead (Pb) exposure. However, experimental studies on Pb-induced neurotoxicity in prepubertal (PP) rats are limited. The present study aimed to examine the propensity of ferulic acid (FA), a commonly occurring phenolic acid in staple foods (fruits, vegetables, cereals, coffee etc.) to abrogate Pb-induced toxicity. Initially, we characterized Pb-induced adverse effects among PP rats exposed to Pb acetate (1,000–3,000 ppm in drinking water) for 5 weeks in terms of locomotor phenotype, activity of 5-aminolevulinic acid dehydratase (ALAD) in the blood, blood Pb levels and oxidative stress in brain regions. Further, the ameliorative effects of oral supplements of FA (25 mg/kg bw/day) were investigated in PP rats exposed to Pb (3,000 ppm). Pb intoxication increased the locomotor activity and FA supplements partially reversed the phenotype, while the reduced ALAD activity was also restored. FA significantly abrogated the enhanced oxidative stress in cerebellum (Cb) and hippocampus (Hc) as evidenced in terms of ROS generation, lipid peroxidation and protein carbonyls. Further, Pb-mediated perturbations in the glutathione levels and activity of enzymic antioxidants were also markedly restored. Furthermore, the protective effect of FA was discernible in striatum in terms of reduced oxidative stress, restored cholinergic activity and dopamine levels. Interestingly, reduced activity levels of mitochondrial complex I in Cb and enhanced levels in Hc among Pb-intoxicated rats were ameliorated by FA supplements. FA also decreased the number of damaged cells in cornu ammonis area CA1 and dentate gyrus as reflected by the histoarchitecture of Hc among Pb intoxicated rats. Collectively, our findings in the PP model allow us to hypothesize that ingestion of common phenolics such as FA may significantly alleviate the neurotoxic effects of Pb which may be largely attributed to its ability to abrogate oxidative stress.







Similar content being viewed by others
Abbreviations
- ALAD:
-
Aminolevulinic acid dehydratase
- Pb:
-
Lead
- Cb:
-
Cerebellum
- St:
-
Straitum
- ROS:
-
Reactive oxygen species
- MDA:
-
Malondialdehyde
- HP:
-
Hydroperoxides
- AChE:
-
Acetylcholinesterase
- GSH:
-
Reduced Glutathione
- HPLC:
-
High performance Liquid chromatography
- DA:
-
Dopamine
References
Kim S, Hyun J, Kim H, Kim Y, Kim E, Jang J, Kim K (2011) Effects of lead exposure on nitric oxide-associated gene expression in the olfactory bulb of mice. Biol Trace Elem Res 142:683–692
Dairam A, Limson JL, Watkins GM, Antunes E, Daya S (2007) Curcuminoids, curcumin, and demethoxycurcumin reduce lead-induced memory deficits in male Wistar rats. J Agric Food Chem 55:1039–1044
Prasanthi RPJ, Devi CB, Basha DC, Reddy NS, Reddy GR (2010) Calcium and zinc supplementation protects lead (Pb)-induced perturbations in antioxidant enzymes and lipid peroxidation in developing mouse brain. Int J Dev Neurosci 28:161–167
Jin Y, Yu F, Liao Y, Liu S, Liu M, Xu J, Yang J (2011) Therapeutic efficiency of succimer used with calcium and ascorbic acid in the treatment of mild lead-poisoning. Environ Toxicol Pharmacol 31:137–142
Ahamed M, Siddiqui MKJ (2007) Low level lead exposure and oxidative stress: current opinions. Clin Chim Acta 383:57–64
Bokara KK, Brown E, McCormick R, Yallapragada PR, Rajanna S, Bettaiya R (2008) Lead-induced increase in antioxidant enzymes and lipid peroxidation products in developing rat brain. Biometals 21:9–16
Toscano CD, Guilarte TR (2005) Lead neurotoxicity: from exposure to molecular effects. Brain Res Review 49:529–554
Neal AP, Guilarte TR (2010) Molecular neurobiology of lead (Pb2+): effects on synaptic function. Mol Neurobiol 42:151–160
Baranowska-Bosiacka I, Gutowska I, Marchlewicz M, Marchetti C, Kurzawski M, Dziedziejko V, Kolasa A, Olszewska M, Rybicka M, Safranow K, Nowacki P, Wiszniewska B, Chlubek D (2012) Disrupted pro- and antioxidative balance as a mechanism of neurotoxicity induced by perinatal exposure to lead. Brain Res 1435:56–71
Papanikolaou NC, Hatzidaki EG, Belivanis S, Tzanakakis GN, Tsatsakis AM (2005) Lead toxicity update. A brief review. Med Sci Monit 11:RA329–RA336
Pappas JB, Nuttall KL, Ahlquist JT, Allen EM, Banner W Jr (1995) Oral dimercaptosuccinic acid and ongoing exposure to lead: effects on heme synthesis and lead distribution in a rat model. Toxicol Appl Pharmacol 133:121–129
van Duynhoven J, Vaughan EE, Jacobs DM, Kemperman RA, van Velzen EJ, Gross G, Roger LC, Possemiers S, Smilde AK, Dore J, Westerhuis JA, Van de Wiele T (2011) Metabolic fate of polyphenols in the human superorganism. Proc Natl Acad Sci USA 108:4531–4538
Chang B-J, Jang B-J, Son TG, Cho IH, Quan FS, Choe NH, Nahm SS, Lee JH (2012) Ascorbic acid ameliorates oxidative damage induced by maternal low-level lead exposure in the hippocampus of rat pups during gestation and lactation. Food Chem Toxicol 50:104–108
Liu C-M, Zheng G-H, Cheng C, Sun J-M (2013) Quercetin protects mouse brain against lead-induced neurotoxicity. J Agric Food Chem 61:7630–7635
Reckziegel P, Dias VT, Benvegnú D, Boufleur N, Silva Barcelos RC, Seqat HJ, Pase CS, Dos Santos CM, Flores EM, Burger ME (2011) Locomotor damage and brain oxidative stress induced by lead exposure are attenuated by gallic acid treatment. Toxicol Lett 203:74–81
Khalaf AA, Moselhy WA, Abdel-Hamed MI (2012) The protective effect of green tea extract on lead induced oxidative and DNA damage on rat brain. Neurotoxicology 33:280–289
Gautam P, Flora SJS (2010) Oral supplementation of gossypin during lead exposure protects alteration in heme synthesis pathway and brain oxidative stress in rats. Nutrition 26:563–570
Liu C-M, Ma J-Q, Sun Y-Z (2011) Protective role of puerarin on lead-induced alterations of the hepatic glutathione antioxidant system and hyperlipidemia in rats. Food Chem Toxicol 49:3119–3127
Srinivasan M, Sudheer AR, Menon VP (2007) Ferulic Acid: therapeutic potential through its antioxidant property. J Clin Biochem Nutr 40:92–100
Barone E, Calabrese V, Mancuso C (2009) Ferulic acid and its therapeutic potential as a hormetin for age-related diseases. Biogerontology 10:97–108
Sultana R (2012) Ferulic acid ethyl ester as a potential therapy in neurodegenerative disorders. Biochim Biophys Acta 1822:748–752
Cheng C-Y, Su S-Y, Tang N-Y, Ho TY, Chiang SY, Hsieh CL (2008) Ferulic acid provides neuroprotection against oxidative stress-related apoptosis after cerebral ischemia/reperfusion injury by inhibiting ICAM-1 mRNA expression in rats. Brain Res 1209:136–150
Koh P-O (2012) Ferulic acid prevents the cerebral ischemic injury-induced decrease of Akt and Bad phosphorylation. Neurosci Lett 507:156–160
Yabe T, Hirahara H, Harada N, Ito N, Nagai T, Sanagi T, Yamada H (2010) Ferulic acid induces neural progenitor cell proliferation in vitro and in vivo. Neuroscience 165:515–524
Mori T, Koyama N, Guillot-Sestier MV, Tan J, Town T (2013) Ferulic acid is a nutraceutical β-secretase modulator that improves behavioral impairment and alzheimer-like pathology in transgenic mice. PLoS One 8:e55774
Denny Joseph KM, Muralidhara (2014) Neuroprotective efficacy of a combination of fish oil and ferulic acid against 3-nitropropionic acid-induced oxidative stress and neurotoxicity in rats: behavioural and biochemical evidence. Appl Physiol Nutr Metab 39:487–496
Cauli O, Morelli M (2002) Subchronic caffeine administration sensitizes rats to the motor-activating effects of dopamine D(1) and D(2) receptor agonists. Psychopharmacology 162:246–254
Berlin A, Schaller KH (1974) European standardized method for the determination of delta-aminolevulinic acid dehydratase activity in blood. Z Klin Chem Klin Biochem 12:389–390
Peixoto NC, Rocha LC, Moraes DP, Bebianno MJ, Dressler VL, Flores EM, Pereira ME (2008) Changes in levels of essential elements in suckling rats exposed to zinc and mercury. Chemosphere 72:1327–1332
Moreadith RW, Fiskum G (1984) Isolation of mitochondria from ascites tumor cells permeabilized with digitonin. Anal Biochem 137:360–367
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Girish C, Muralidhara (2012) Propensity of Selaginella delicatula aqueous extract to offset rotenone-induced oxidative dysfunctions and neurotoxicity in Drosophila melanogaster: implications for Parkinson’s disease. NeuroToxicology 33:444–456
Farant JP, Wigfield DC (1982) Biomonitoring lead exposure with delta-aminolevulinate dehydratase (ALA-D) activity ratios. Int Arch Occup Environ Health 51:15–24
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG, Ahn BW, Shaltiel S, Stadtman ER (1990) Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478
Mokrasch LC, Teschke EJ (1984) Glutathione content of cultured cells and rodent brain regions: a specific fluorometric assay. Anal Biochem 140:506–509
Kostyuk VA, Potapovich AI (1989) Superoxide–driven oxidation of quercetin and a simple sensitive assay for determination of superoxide dismutase. Biochem Int 19:1117–1124
Luthman M, Holmgren A (1982) Rat liver thioredoxin and thioredoxin reductase: purification and characterization. Biochemistry (Mosc) 21:6628–6633
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490
Guthenberg C, Alin P, Mannervik B (1985) Glutathione transferase from rat testis. Methods Enzymol 113:507–510
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–121
Berridge MV, Tan AS (1993) Characterization of the cellular reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT): subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Arch Biochem Biophys 303:474–482
Navarro A, Sánchez Del Pino MJ, Gómez C, Peralta JL, Boveris A (2002) Behavioral dysfunction, brain oxidative stress, and impaired mitochondrial electron transfer in aging mice. Am J Physiol Regul Integr Comp Physiol 282:R985–R992
Navarro A, Gomez C, López-Cepero JM, Boveris A (2004) Beneficial effects of moderate exercise on mice aging: survival, behavior, oxidative stress, and mitochondrial electron transfer. Am J Physiol Regul Integr Comp Physiol 286:R505–R511
Ellman GL, Courtney KD, Andres V, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Dalpiaz A, Filosa R, de Caprariis P, Conte G, Bortolotti F, Biondi C, Scatturin A, Prasad PD, Pavan B (2007) Molecular mechanism involved in the transport of a prodrug dopamine glycosyl conjugate. Int J Pharm 336:133–139
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lanphear BP, Hornung R, Khoury J, Yolton K, Baghurst P, Bellinger DC, Canfield RL, Dietrich KN, Bornschein R, Greene T, Rothenberg SJ, Needleman HL, Schnaas L, Wasserman G, Graziano J, Roberts R (2005) Low-level environmental lead exposure and children’s intellectual function: an international pooled analysis. Environ Health Perspect 113:894–899
Pande M, Flora SJS (2002) Lead induced oxidative damage and its response to combined administration of alpha-lipoic acid and succimers in rats. Toxicology 177:187–196
Hermes-Lima M, Pereira B, Bechara EJ (1991) Are free radicals involved in lead poisoning? Xenobiotica 21:1085–1090
Yusof M, Yildiz D, Ercal N (1999) N-acetyl-l-cysteine protects against delta-aminolevulinic acid-induced 8-hydroxydeoxyguanosine formation. Toxicol Lett 106:41–47
Velaga MK, Basuri CK, Robinson Taylor KS, Yallapragada PR, Rajanna S, Rajanna B (2014) Ameliorative effects of Bacopa monniera on lead-induced oxidative stress in different regions of rat brain. Drug Chem Toxicol 37:357–364
Vaziri ND, Lin C-Y, Farmand F, Sindhu RK (2003) Superoxide dismutase, catalase, glutathione peroxidase and NADPH oxidase in lead-induced hypertension. Kidney Int 63:186–194
Adam A, Crespy V, Levrat-Verny MA, Leenhardt F, Leuillet M, Demigne C, Remesy C (2002) The bioavailability of ferulic acid is governed primarily by the food matrix rather than its metabolism in intestine and liver in rats. J Nutr 132:1962–1968
Kanski J, Aksenova M, Stoyanova A, Butterfield DA (2002) Ferulic acid antioxidant protection against hydroxyl and peroxyl radical oxidation in synaptosomal and neuronal cell culture systems in vitro: structure-activity studies. J Nutr Biochem 13:273–281
Nehru B, Kanwar SS (2004) N-acetylcysteine exposure on lead-induced lipid peroxidative damage and oxidative defense system in brain regions of rats. Biol Trace Elem Res 101:257–264
Zhang J, Wang X-F, Lu ZB, Liu NQ, Zhao BL (2004) The effects of meso-2,3-dimercaptosuccinic acid and oligomeric procyanidins on acute lead neurotoxicity in rat hippocampus. Free Radic Biol Med 37:1037–1050
Zhang L, Corona-Morales AA, Vega-González A, Garcia-Estrada J, Escobar A (2009) Dietary tryptophan restriction in rats triggers astrocyte cytoskeletal hypertrophy in hippocampus and amygdala. Neurosci Lett 450:242–245
Wang J, Wu J, Zhang Z (2006) Oxidative stress in mouse brain exposed to lead. Ann Occup Hyg 50:405–409
Yin S-T, Tang M-L, Su L, Chen L, Hu P, Wang HL, Wang M, Ruan DY (2008) Effects of Epigallocatechin-3-gallate on lead-induced oxidative damage. Toxicology 249:45–54
Xu Y, Zhang L, Shao T, Ruan L, Wang L, Sun J, Li J, Zhu X, O’Donnell JM, Pan J (2013) Ferulic acid increases pain threshold and ameliorates depression-like behaviors in reserpine-treated mice: behavioral and neurobiological analyses. Metab Brain Dis 28:571–583
Moreira EG, Vassilieff I, Vassilieff VS (2001) Developmental lead exposure: behavioral alterations in the short and long term. Neurotoxicol Teratol 23:489–495
Ramesh GT, Manna SK, Aggarwal BB, Jadhav AL (2001) Lead exposure activates nuclear factor kappa B, activator protein-1, c-Jun N-terminal kinase and caspases in the rat brain. Toxicol Lett 123:195–207
NourEddine D, Miloud S, Abdelkader A (2005) Effect of lead exposure on dopaminergic transmission in the rat brain. Toxicology 207:363–368
Cory-Slechta DA, Stern S, Weston D, Allen JL, Liu S (2010) Enhanced learning deficits in female rats following lifetime pb exposure combined with prenatal stress. Toxicol Sci 117:427–438
Basha DC, Rani MU, Devi CB, Kumar MR, Reddy GR (2012) Perinatal lead exposure alters postnatal cholinergic and aminergic system in rat brain: reversal effect of calcium co-administration. Int J Dev Neurosci 30:343–350
Lasley SM, Gilbert ME (2000) Glutamatergic components underlying lead-induced impairments in hippocampal synaptic plasticity. NeuroToxicology 21:1057–1068
Nowak P, Szczerbak G, Nitka D, Kostrzewa RM, Sitkiewicz T, Brus R (2008) Effect of prenatal lead exposure on nigrostriatal neurotransmission and hydroxyl radical formation in rat neostriatum: dopaminergic-nitrergic interaction. Toxicology 246:83–89
Luo Y, Zhao HP, Zhang J, Wang J, Yang WL, Yang M, Liao ZG (2012) Effect of ferulic acid on learning and memory impairments of vascular dementia rats and its mechanism of action. Yao Xue Xue Bao 47:256–260
Acknowledgments
We wish to thank the Director, CFTRI for his keen interest in this study. The first author thank the Indian Council Medical Research (ICMR), New Delhi, for the award of a Senior Research Fellowship.
Conflict of interest
The authors declare that there are no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lalith Kumar, V., Muralidhara Ameliorative Effects of Ferulic Acid Against Lead Acetate-Induced Oxidative Stress, Mitochondrial Dysfunctions and Toxicity in Prepubertal Rat Brain. Neurochem Res 39, 2501–2515 (2014). https://doi.org/10.1007/s11064-014-1451-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-014-1451-7