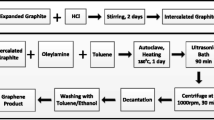
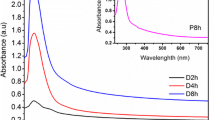
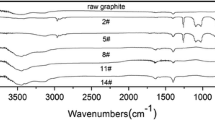
Abstract
A versatile and scalable mixed-solvent strategy, by which two mediocre solvents could be combined into good solvents for exfoliating graphite, is demonstrated for facile and green preparation of graphene by liquid-phase exfoliation of graphite. Mild sonication of crystal graphite powder in a mixture of water and alcohol could yield graphene nanosheets, which formed a highly stable suspension in the mixed solvents. The graphene yield was estimated as ~10 wt%. The optimum mass fraction of ethanol in water–ethanol mixtures and isopropanol in water–isopropanol mixtures was experimentally determined as ~40 and ~55 % respectively, which could be roughly predicted by the theory of Hansen solubility parameters. Statistics based on atomic force microscopic analysis show that up to ~86 % of the prepared nanosheets were less than 10-layer thick with a monolayer fraction of ~8 %. High resolution transmission electron microscopy, infrared spectroscopy, X-ray diffraction, and Raman spectrum analysis of the vacuum-filtered films suggest the graphene sheets to be largely free of defects and oxides. The proposed mixed-solvent strategy here extends the scope for liquid-phase processing graphene and gives researchers great freedom in designing ideal solvent systems for specific applications.







Similar content being viewed by others
References
Abergel DSL, Fal’ko VI (2007) Optical and magneto-optical far-infrared properties of bilayer graphene. Phys Rev B 75(15). doi:10.1103/PhysRevB.75.155430
Bourlinos AB, Georgakilas V, Zboril R, Steriotis TA, Stubos AK (2009) Liquid-phase exfoliation of graphite towards solubilized graphenes. Small 5(16):1841–1845. doi:10.1002/smll.200900242
Coleman JN (2009) Liquid-phase exfoliation of nanotubes and graphene. Adv Funct Mater 19(23):3680–3695. doi:10.1002/adfm.200901640
Cravotto G, Cintas P (2010) Sonication-assisted fabrication and post-synthetic modifications of graphene-like materials. Chemistry 16(18):5246–5259. doi:10.1002/chem.200903259
De S, King PJ, Lotya M, O’Neill A, Doherty EM, Hernandez Y, Duesberg GS, Coleman JN (2010) Flexible, transparent, conducting films of randomly stacked graphene from surfactant-stabilized, oxide-free graphene dispersions. Small 6(3):458–464. doi:10.1002/smll.200901162
Dreyer DR, Park S, Bielawski CW, Ruoff RS (2010) The chemistry of graphene oxide. Chem Soc Rev 39(1):228–240. doi:10.1039/b917103g
Geim AK (2009) Graphene: status and prospects. Science 324(5934):1530–1534. doi:10.1126/science.1158877
Geim AK, Novoselov KS (2007) The rise of graphene. Nat Mater 6(3):183–191. doi:10.1038/nmat1849
Hansen CM (2007) Hansen solubility parameters: a user’s handbook. CRC Press, Boca Raton
Hernandez Y, Nicolosi V, Lotya M, Blighe FM, Sun Z, De S, McGovern IT, Holland B, Byrne M, Gun’Ko YK, Boland JJ, Niraj P, Duesberg G, Krishnamurthy S, Goodhue R, Hutchison J, Scardaci V, Ferrari AC, Coleman JN (2008) High-yield production of graphene by liquid-phase exfoliation of graphite. Nat Nanotechnol 3(9):563–568. doi:10.1038/nnano.2008.215
Hernandez Y, Lotya M, Rickard D, Bergin SD, Coleman JN (2010) Measurement of multicomponent solubility parameters for graphene facilitates solvent discovery. Langmuir 26(5):3208–3213. doi:10.1021/la903188a
Hontoria-Lucas C (1995) Study of oxygen-containing groups in a series of graphite oxides: physical and chemical characterization. Carbon 33(11):1585–1592. doi:10.1016/0008-6223(95)00120-3
Khan U, O’Neill A, Lotya M, De S, Coleman JN (2010) High-concentration solvent exfoliation of graphene. Small 6(7):864–871. doi:10.1002/smll.200902066
Li D, Muller MB, Gilje S, Kaner RB, Wallace GG (2008a) Processable aqueous dispersions of graphene nanosheets. Nat Nanotechnol 3(2):101–105. doi:10.1038/nnano.2007.451
Li X, Zhang G, Bai X, Sun X, Wang X, Wang E, Dai H (2008b) Highly conducting graphene sheets and Langmuir–Blodgett films. Nat Nanotechnol 3(9):538–542. doi:10.1038/nnano.2008.210
Lotya M, King PJ, Khan U, De S, Coleman JN (2010) High-concentration, surfactant-stabilized graphene dispersions. ACS Nano 4(6):3155–3162. doi:10.1021/nn1005304
Lu J, Yang JX, Wang J, Lim A, Wang S, Loh KP (2009) One-pot synthesis of fluorescent carbon nanoribbons, nanoparticles, and graphene by the exfoliation of graphite in ionic liquids. ACS Nano 3(8):2367–2375. doi:10.1021/nn900546b
Malard LM, Pimenta MA, Dresselhaus G, Dresselhaus MS (2009) Raman spectroscopy in graphene. Phys Rep 473(5–6):51–87. doi:10.1016/j.physrep.2009.02.003
Meyer JC, Geim AK, Katsnelson MI, Novoselov KS, Booth TJ, Roth S (2007) The structure of suspended graphene sheets. Nature 446(7131):60–63. doi:10.1038/nature05545
Morant RA (1970) The crystallite size of pyrolytic graphite. J Phys D Appl Phys 3(9):1367–1373. doi:10.1088/0022-3727/3/9/319
Murugan AV, Muraliganth T, Manthiram A (2009) Rapid, facile microwave-solvothermal synthesis of graphene nanosheets and their polyaniline nanocomposites for energy storage. Chem Mater 21(21):5004–5006. doi:10.1021/cm902413c
Nemesincze P, Osvath Z, Kamaras K, Biro L (2008) Anomalies in thickness measurements of graphene and few layer graphite crystals by tapping mode atomic force microscopy. Carbon 46(11):1435–1442. doi:10.1016/j.carbon.2008.06.022
Nicolosi V, Vrbanic D, Mrzel A, McCauley J, O’Flaherty S, McGuinness C, Compagnini G, Mihailovic D, Blau WJ, Coleman JN (2005) Solubility of Mo6S4.5I4.5 nanowires in common solvents: a sedimentation study. J Phys Chem B 109(15):7124–7133. doi:10.1021/jp045166r
Norimatsu W, Takada J, Kusunoki M (2011) Formation mechanism of graphene layers on SiC (\( 000{\bar{\text{1}}} \)) in a high-pressure argon atmosphere. Phys Rev B 84(3):1–6. doi:10.1103/PhysRevB.84.035424
Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y, Dubonos SV, Grigorieva IV, Firsov AA (2004) Electric field effect in atomically thin carbon films. Science 306(5696):666–669. doi:10.1126/science.1102896
Shapira P, Youtie J, Arora S (2012) Early patterns of commercial activity in graphene. J Nanopart Res 14(4). doi:10.1007/s11051-012-0811-y
Shen Z, Li J, Yi M, Zhang X, Ma S (2011) Preparation of graphene by jet cavitation. Nanotechnology 22(36):365306. doi:10.1088/0957-4484/22/36/365306
Shih CJ, Vijayaraghavan A, Krishnan R, Sharma R, Han JH, Ham MH, Jin Z, Lin S, Paulus GL, Reuel NF, Wang QH, Blankschtein D, Strano MS (2011) Bi- and trilayer graphene solutions. Nat Nanotechnol 6(7):439–445. doi:10.1038/nnano.2011.94
Si Y, Samulski ET (2008) Synthesis of water soluble graphene. Nano Lett 8(6):1679–1682. doi:10.1021/nl080604h
Stankovich S, Piner RD, Chen X, Wu N, Nguyen ST, Ruoff RS (2006) Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly(sodium 4-styrenesulfonate). J Mater Chem 16(2):155. doi:10.1039/b512799h
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammes A, Jia Y, Wu Y, Nguyen ST, Ruoff RS (2007) Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45(7):1558–1565. doi:10.1016/j.carbon.2007.02.034
Sun Z, Yan Z, Yao J, Beitler E, Zhu Y, Tour JM (2010) Growth of graphene from solid carbon sources. Nature 468(7323):549–552. doi:10.1038/nature09579
Suslick KS, Price GJ (1999) Applications of ultrasound to materials chemistry. Annu Rev Mater Sci 29(1):295–326. doi:10.1146/annurev.matsci.29.1.295
Titelman GI, Gelman V, Bron S, Khalfin RL, Cohen Y, Bianco-Peled H (2005) Characteristics and microstructure of aqueous colloidal dispersions of graphite oxide. Carbon 43(3):641–649. doi:10.1016/j.carbon.2004.10.035
Valles C, Drummond C, Saadaoui H, Furtado CA, He M, Roubeau O, Ortolani L, Monthioux M, Penicaud A (2008) Solutions of negatively charged graphene sheets and ribbons. J Am Chem Soc 130(47):15802–15804. doi:10.1021/ja808001a
Whitby RLD, Korobeinyk A, Mikhalovsky SV, Fukuda T, Maekawa T (2011) Morphological effects of single-layer graphene oxide in the formation of covalently bonded polypyrrole composites using intermediate diisocyanate chemistry. J Nanopart Res 13(10):4829–4837. doi:10.1007/s11051-011-0459-z
Wilson NR, Pandey PA, Beanland R, Young RJ, Kinloch IA, Gong L, Liu Z, Suenaga K, Rourke JP, York SJ, Sloan J (2009) Graphene oxide: structural analysis and application as a highly transparent support for electron microscopy. ACS Nano 3(9):2547–2556. doi:10.1021/nn900694t
Yi M, Li J, Shen Z, Zhang X, Ma S (2011) Morphology and structure of mono- and few-layer graphene produced by jet cavitation. Appl Phys Lett 99(12):123112. doi:10.1063/1.3641863
Acknowledgments
This study was supported by the Special Funds for Co-construction Project of Beijing Municipal Commission of Education, the “985” Project of Ministry of Education of China, and the fundamental research funds for the Central Universities.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yi, M., Shen, Z., Ma, S. et al. A mixed-solvent strategy for facile and green preparation of graphene by liquid-phase exfoliation of graphite. J Nanopart Res 14, 1003 (2012). https://doi.org/10.1007/s11051-012-1003-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-012-1003-5