Abstract
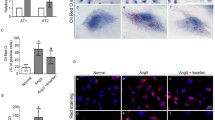
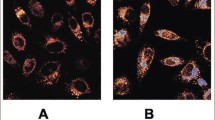
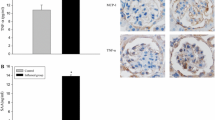
The aim of this article is to investigate whether interleukin-1β (IL-1β) could regulate the intracellular accumulation of cholesterol and the expression of lipid-metabolism-related regulators in podocytes in vitro and the potential mechanisms. Podocytes were treated with 200 μg/ml of low-density protein (LDL), 20 ng/ml of IL-1β, or 200 μg/ml of LDL plus 5–20 ng/ml of IL-1β for 24 h in vitro. The contents of intracellular cholesterol were determined by enzymatic assays and Oil Red O staining. The levels of LDL receptor (LDLr), 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, sterol regulatory element binding protein 2 (SREBP-2), SREBP cleavage activating protein (SCAP), and insulin-induced gene-1 (Insig-1) expression were characterized by real-time polymerase chain reaction (RT-PCR) and Western blot assays. Treatment with IL-1β or LDL alone increased the contents of intracellular cholesterol (P < 0.05 for both) and lipid droplets, and treatment with both IL-1β and LDL further increased the accumulation of intracellular cholesterol in podocytes (P < 0.05 vs. LDL alone). While loading with LDL significantly inhibited the expression of LDLr, HMG-CoA reductase, nuclear SREBP-2 (nSREBP-2), SCAP, and Insig-1 by 40–62% treatment with IL-1β enhanced the expression of LDLr, HMG-CoA reductase and nSREBP-2, but not Insig-1, in podocytes (P < 0.05 vs. control). Treatment with both LDL and IL-1β significantly increased the levels of LDLr and HMG-CoA reductase expression and the ratio of SCAP to Insig-1, as compared with that in the LDL-treated podocytes (P < 0.05 vs. LDL alone). Our data indicated that IL-1β mitigated the LDL-triggered SCAP-SREBP-2-mediated feedback inhibition on the expression of LDLr and HMG-CoA reductase, leading to the intracellular accumulation of LDL-cholesterol in podocytes in vitro.



Similar content being viewed by others
References
Klshr S, Schreiner G, Ichikawa I (1988) The progression of renal disease. N Engl J Med 318:1657–1666
Moorhead JF, Chan MK, El-Nahas M, Varghese Z (1988) Lipid nephrotoxicity in chronic progressive glomerular and tubulointerstitial disease. Lancet 2:1309–1311
Keane WF, Kasiske BL, O’Donnell MP (1988) Lipids and progressive glomerulosclerosis. A model analogous to atherosclerosis. Am J Nephrol 8:261–271
Lee HS, Lee JS, Koh HI, Ko KW (1991) Intraglomerular lipid deposition in routine biopsies. Clin Nephrol 36:67–75
Grone HJ, Walli A, Grone E et al (1990) Receptor mediated uptake of apo B and apo E rich liporloteins by human glomerular epithelial cells. Kidney Int 37:1449–1459
Noel LH (1999) Morphological features of primary focal and segmental glomerulosclerosis. Nephrol Dial Transpl 14(Suppl 3):53–57
Ruan XZ, Varghese Z, Moorhead JF (2003) Inflammation modifies lipid-mediated renal injury. Nephrol Dial Transpl 18:27–32
Brown MS, Goldstein JL (1986) A receptor-mediated pathway for cholesterol homeostasis. Science 232:34–47
Goldstein JL, Brown MS (1985) The LDL receptor and the regulation of cellular cholesterol metabolism. J Cell Sci 3(Suppl):131–137
Gallo LL, Atasoy R, Vahouny GV, Treadwell CR (1978) Enzymatic assay for cholesterol ester hydrolase activity. J Lipid Res 19:913–916
Rawson RB (2003) The SREBP pathway—insights from Insigs and insects. Nat Rev Mol Cell Biol 4:631–640
Yang T, Espenshade PJ, Wright ME et al (2002) Crucial step in cholesterol homeostasis: sterols promote binding of SCAP to INSIG-1, a membrane protein that facilitates retention of SREBPs in ER. Cell 110:489–500
Shankland SJ, Pippin JW, Reiser J, Mundel P (2007) Podocytes in culture: past, present, and future. Kidney Int 72:26–36
Wheeler DC, Chana RS, Topley N, Petersen MM, Davies M, Williams JD (1994) Oxidation of low density lipoprotein by mesangial cells may promote glomerular injury. Kidney Int 45:1628–1636
Ruan XZ, Varghese Z, Fernando R, Moorhead JF (1998) Cytokine regulation of low-density lipoprotein receptor gene transcription in human mesangial cells. Nephrol Dial Transpl 13:1391–1397
Gallo LL, Atasoy R, Vahouny GV, Treadwell CR (1998) Enzymatic assay for cholesterol ester hydrolase activity. J Lipid Res 19:913–916
Gamble W, Vaughan M, Kruth HS, Avigan J (1998) Procedure for determination of free and total cholesterol in micro- or nanogram amounts suitable for studies with cultured cells. J Lipid Res 19:1068–1070
Markwell MA, Hass SM, Bieber LL, Tolbert NE (1978) A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem 87:206–210
Seliger SL (2006) Inflammation and dyslipidemia in nephropathy: an epidemiologic perspective. Kidney Int 69:206–208
Baud L, Fouqueray B, Bellocq A (1999) Inflammatory mechanisms of renal fibrosis: glomerulonephritis. Bull Acad Natl Med 183:23–31
Ruan XZ, Varghese Z, Powis SH, Moorhead JF (2001) Dysregulation of LDL receptor under the influence of inflammatory cytokines: a new pathway for foam cell formation. Kidney Int 60:1716–1725
Chen Y, Ruan XZ, Li Q et al (2007) Inflammatory cytokines disrupt LDL-receptor feedback regulation and cause statin resistance: a comparative study in human hepatic cells and mesangial cells. Am J Physiol Renal Physiol 293:680–687
Ruan XZ, Moorhead JF, Tao JL, Ma KL, Wheeler DC, Powis SH, Varghese Z (2006) Mechanisms of dysregulation of low-density lipoprotein receptor expression in vascular smooth muscle cells by inflammatory cytokines. Arterioscler Thromb Vasc Biol 26:1150–1155
Kunjathoor VV, Febbraio M, Podrez EA, Moore KJ et al (2002) Scavenger receptors class A-I/II and CD36 are the principal receptors responsible for the uptake of modified low density lipoprotein leading to Lipid loading in macrophages. J Biol Chem 277:49982–49988
Takemura T, Yoshioka K, Aya N et al (1993) Apolipoproteins and lipoprotein receptors in glomeruli in human kidney diseases. Kidney Int 43:918–927
Quaschning T, Koniger M et al (1997) Receptor-mediated lipoprotein uptake by human glomerular cells: comparison with skin fibroblasta and HepG2 cells. Nephrol Dial Transplant 12:2528–2536
Ye Q, Chen Y, Lei H, Liu Q, Moorhead JF, Varghese Z, Ruan XZ (2009) Inflammatory stress increases unmodified LDL uptake via LDL receptor: an alternative pathway for macrophage foam-cell formation. Inflamm Res 58:809–818
Gil G, Faust JR, Chin DJ, Goldstein JL, Brown MS (1985) Membrane-bound domain of HMG CoA reductase is required for sterol-enhanced degradation of the enzyme. Cell 41(1):249–258
DiDonato J, Mercurio F, Rosette C (1996) Mapping of the inducible IkappaB phosphorylation sites that signal its ubiquitination and degradation. Mol Cell Biol 16(4):1295–1304
Ravid T, Doolman R, Avner R, Harats D, Roitelman J (2000) The ubiquitin-proteasome pathway mediates the regulated degradation of mammalian 3-hydroxy-3-methylglutaryl-coenzyme A reductase. J Biol Chem 275(46):35840–35847
Sever N, Yang T, Brown MS, Goldstein JL, DeBose-Boyd RA (2003) Accelerated degradation of HMGCoA reductase mediated by binding of Insig-1 to its sterol-sensing domain. Mol Cell 11(1):25–33
Hua X, Nohturfft A, Goldstein JL, Brown MS (1996) Sterol resistance in CHO cells traced to point mutation in SREBP cleavage-activating protein. Cell 87(3):415–426
Acknowledgments
We would like to thank Dr. Xiong Z Ruan (Renal Research Unit, Department of Nephrology, Royal Free Hospital School of Medicine, London, UK) for his generous provision of key reagents, technical assistance, and critical comments. This study was financially supported by the grants from the National Natural Science Foundation of China (grant no. 30672267) and Natural Science Foundation Project of CQ CSTC (grant no. CSTC, 2009BA5080).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, G., Li, Q., Wang, L. et al. Interleukin-1β enhances the intracellular accumulation of cholesterol by up-regulating the expression of low-density lipoprotein receptor and 3-hydroxy-3-methylglutaryl coenzyme A reductase in podocytes. Mol Cell Biochem 346, 197–204 (2011). https://doi.org/10.1007/s11010-010-0605-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0605-4