Abstract
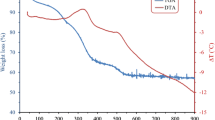
This study aimed to understand the microstructural, optical, and electrical properties of tin-doped indium oxide (ITO) prepared with tetravalent and divalent tin salts. The influence of tin valence on the electrical, optical, structural, and morphological properties of the films were characterized by the mean of four-point probe, thermogravimetric analysis, differential thermal analysis (DTA), UV–Vis spectroscopy, X-ray diffraction (XRD), field emission scanning electron microscopy (FESEM), and X-ray photoelectron spectroscope. XRD results revealed formation of cubic bixbyite structure of In2O3 with a small shift in major peak position toward lower angles with addition of Sn2+ and Sn4+. TG–DTA showed that the optimum heat treatment temperatures for thin films prepared with divalent and tetravalent tin salts are 500 and 450 °C, respectively. FESEM showed that with decreasing tin valence, grain size was gradually increased. The surface coverage for both thin films is similar, without any remarkable cracks. By increasing the tin valence from II to IV, high transparency (88.5 % in the visible region) and low conductivity (10 kΩ/sq.) can be obtained after calcination in air at 450 °C, which can be considered acceptable for electrostatic and antistatic applications of ITO thin films. The Haacke figures of merit at λ = 550 nm are comprised of between 2.94 × 10−5 Ω−1 and 1.83 × 10−5 Ω−1 for ITO prepared from tetravalent and divalent tin salt, respectively.
Graphical Abstract











Similar content being viewed by others
References
Ginley DS, Bright C (2000) Transparent conducting oxides. MRS Bull 25:15–18
Chopra KL, Major S, Pandya DK (1983) Transparent conductors—a status review. Thin Solid Films 102:1–46
Alam MJ, Cameron DC (2000) Optical and electrical properties of transparent conductive ITO thin films deposited by sol–gel process. Thin Solid Films 377–378:455–459
González B, Mason TO, Quintana JP, Warschkow O, Ellis DE, Hwang JH, Hodges JD, Jorgensen JP (2004) Defect structure studies of bulk and nano-indium-tin oxide. J Appl Phys 96:3912–3920
Granqvist CG, Hamberg I (1986) Evaporated Sn-doped In2O3 films: basic optical properties and applications to energy efficient windows. J Appl Phys 60:122–159
Granqvist CG, Hultåker A (2002) Transparent and conducting ITO films: new developments and applications. Thin Solid Films 411:1–5
Hwang JH, Edwards DD, Kammler DR, Mason TO (2000) Point defects and electrical properties of Sn-doped In-based transparent conducting oxides. Solid State Ionics 129:135–144
Sunde TOL, Garskaite E, Otter B, Fossheim HE, Saeterli R, Holmestad R, Einarsrud MA, Grande T (2012) Transparent and conducting ITO thin films by spin coating of an aqueous precursor solution. J Mater Chem 22:15740–15749
Uchihashi H, Touge N, Minami T (1989) Preparation of amorphous Al2O3 thin films from stabilized Al-alkoxides by the sol–gel method. J Ceram Soc Jpn 97:396–399
Houng B (2005) Tin doped indium oxide transparent conducting thin films containing silver nanoparticles by sol–gel technique. Appl Phys Lett 87:251922-1–3
Leite ER, Giraldi TR, Pontes FM, Longo EL, Beltran A, Andres J (2003) Crystal growth in colloidal tin oxide nanocrystals induced by coalescence at room temperature. J Appl Phys Lett 83:1566–1568
Ribeiro C, Lee EJH, Giraldi TR, Longo E, Varela JA, Leite ER (2004) Study of synthesis variables in the nanocrystal growth behavior of tin oxide processed by controlled hydrolysis. J Phys Chem B 108:15612–15617
Rizzato AP, Broussous L, Santilli CV, Pulcinelli SH, Craievich AF (2001) Structure of SnO2 alcohols and films prepared by sol–gel dip coating. J Non-Cryst Solids 284:61–67
Kim DW, Hwang IS, Kwon SJ, Kang HY, Park KS, Choi YJ, Choi KJ, Park JG (2007) Highly conductive coaxial SnO2–In2O3 heterostructured nanowires for Li ion battery electrodes. J Nano Lett 7:3041–3045
Epifani M, Diaz R, Arbiol J, Siciliano P, Morante JR (2006) Solution synthesis of thin films in the SnO2–In2O3 system: a case study of the mixing of sol–gel and metal-organic solution processes. J Chem Mater 18:840–846
Segmüller A, Noyan IN, Sperious V (1989) X-ray diffraction studies of thin films and multilayer structure. Pergamon Press, Oxford
Wagner CNJ (1965). In: Cohen JB, Hilliard JE (eds.) Local atomic arrangements studied by X-ray diffraction, vol 36. Gordon and Breach, New York, p 51
Sen S, Halder SK, Gupta SPS (1973) An X-ray line shift analysis in vacuum evaporated silver films. J Phys D Appl Phys 6:1978–1985
Shigesato Y, Hayashi Y, Haranoh T (1992) Doping mechanisms of tin doped indium oxide films. J Appl Phys Lett 61:73–75
Ishida T, Kobayashi H, Nakato Y (1993) Structures and properties of electron-beam-evaporated indium tin oxide films as studied by X-ray photoelectron spectroscopy and work-function measurements. J Appl Phys 73:4344–4350
Yamaguchi M, Ektessabi AI, Nomura H, Yasui N (2004) Characteristics of indium tin oxide thin films prepared using electron beam evaporation. Thin Solid Films 447–448:115–118
Pujilaksono B, Klement U, Nyborg L, Jelvestam U, Hill S, Burgard D (2005) X-ray photoelectron spectroscopy studies of indium tin oxide nanocrystalline powder. Mater Charact 54:1–7
Moulder JF, Stickle WF, Sobol PE, Bomben KD (1992) Handbook of X-ray photoelectron spectroscopy. Perkin-Elmer Corporation, Eden Prairie, Minnesota
Bryan JD, Gamelin DR (2005) Doped semiconductor nanocrystals: synthesis, characterization, physical properties and applications. J Prog Org Coat 54:47–126
Leite ER, Giraldi TR, Pontes FM, Longo E, Beltran A, Andres J (2003) Crystal growth in colloidal tin oxide nanocrystals induced by coalescence at room temperature. Appl Phys Lett 83:1566–1568
Ribeiro C, Lee EJH, Giraldi TR, Longo E, Varela JA, Leite ER (2004) Study of synthesis variables in the nanocrystal growth behavior of tin oxide processed by controlled hydrolysis. J Phys Chem B 108:15612–15617
Sakka S (2004) Handbook of sol–gel science and technology: processing, characterization, and applications. Kluwer, Boston
Zhang Y (1982) Electronegativities of elements in valence states and their applications: a scale for strengths of Lewis acids. Inorg Chem 21:3889–3893
Lee JS, Choi SC (2005) Solvent effect on synthesis of indium tin oxide nano-powders by a solvothermal process. J Eur Ceram Soc 25:3307–3314
Kim S, Choi S, Park Ch, Jin H (1999) Transparent conductive ITO thin films through the sol–gel process using metal salts. Thin Solid Films 347:155–160
Kim JH, Jeon KA, Kim GH, Lee SY (2006) Electrical, structural, and optical properties of ITO thin films prepared at room temperature by pulsed laser deposition. Appl Surf Sci 252:4834–4837
Bisht H, Eun HT, Mehrtens A, Aegerter MA (1999) Comparison of spray pyrolyzed FTO, ATO and ITO coatings for flat and bent glass substrate. Thin Solid Films 351:109–114
Haacke G (1976) New figure of merit for transparent conductors. J Appl Phys 47:4086–4089
Tauc J (1974) Amorphous and liquid semiconductors. Plenum Press, New York
Kundu S, Biswas PK (2005) Synthesis and photoluminescence property of nanostructured sol–gel indium tin oxide film on glass. Chem Phys Lett 414:107–110
Burstein E (1954) Anomalous optical absorption limit in InSb. Phys Rev 93:632–633
Acknowledgments
Authors are grateful to the research council of Sharif University of Technology for the financial support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mirzaee, M., Dolati, A. Effects of tin valence on microstructure, optical, and electrical properties of ITO thin films prepared by sol–gel method. J Sol-Gel Sci Technol 75, 582–592 (2015). https://doi.org/10.1007/s10971-015-3729-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-015-3729-x