Abstract
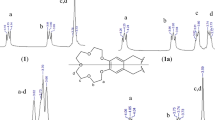
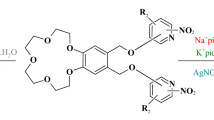
Hexadecanoyl, dihexadecanoyl, dioctadecaneoyl, di-10-undecenoyl, and dicis-9-octadecenoyl derivatives of benzo[18]crown-6, dibenzo[18]crown-6 and dibenzo[24]-8 were synthesized by the condensation of carboxylic acids (palmitic, stearic, oleic and undecenoic acid) with benzo and dibenzo crown ethers in the presence of zinc chloride. The extraction equilibrium constants of such macrocyclic ethers with long side chains were estimated using chloroform/water and dichloromethane/water membranes transfer of Na-PAR (4-(2-pyridylazo)-resorcinol mono sodium monohydrate) with UV–Vis spectroscopy. It was found that they were in the range of 10.88–11.71 in dichloromethane and 8.04–11.77 in chloroform. These results actually show that the Na+ binding effect of macrocyclic ethers depends on the type and the length of side chains. The geometrical properties of the molecules were studied employing semi-empirical calculations by simulated annealing technique. The frontier molecular orbital energies and dipole moments were also examined. The biological activity results showed that the synthesized crown ethers have no activity against the studied microorganisms.
Similar content being viewed by others
References
Pedersen C.J., (1967) J. Am. Chem. Soc.89: 7017
Ü. Çakı ır, H.İ. Uğraş, and Ç. Erk: Supramol. Chem. 16, 193 (2004) references cited
R.L. Bruening, R.M. Izatt, and J.S. Bradshaw: in Y. Inoue and G.W. Gokel (eds.), Cation Binding by Macrocycles, Marcel Dekker. New York (1990), p. 111
G.W. Gokel and J.E. Trafton: in Y. Inoue and G.W. Gokel (eds.), Cation Binding by Macrocycles, Marcel Dekker, New York (1990), p. 253
Namor D.A.F., Sigstad E., (1986) Polyhedron 5: 839
Izatt R.M., Bradshaw J.S., Bruening R.L., Bruening M.L., (1994) Am. Lab. 28: 28c–28m
Nazarenko A.Y., Huszthy P., Bradshaw J.S., Lamb J.D., Izatt R.M., (1994) J. Incl. Phenom. 20: 13
Nakatsuji Y., Nakamura T., Yonetan M., Yuya H., Okahara M., (1988) J. Am. Chem. Soc. 110: 531
Arnold K.A., Echegoyen L., Gokel G.W., (1987) J. Am. Chem. Soc. 107: 3713
McGaw L.C., Jager A.K., Van Staden J.S., (2002) Afr. J. Bot. 68: 417
Kilic T., Dirmenci T., Satil F., Bilsel G., Kocagöz T., Altun M., Gören A.C., (2005) Chem. Nat. Comp.41: 276
Takeda Y., Kato H., (1979) Bull. Chem. Soc. Jpn. 52(4): 1027
Kolthoff I.M., Chantooni M.K., (1997) J. Chem. Eng. Data. 42: 49
Sachleben R.A., Deng Y.P., Bailey D.R., Moyer B.A., (1996) Solvent Extract. Ion Exchange 14: 995
Kimura K., Tsuchida T., Maeda T., Shono T., (1980) Talanta27: 801
Quochi M., İnoue Y., Sakamoto H., Yamahira I., Yoshinaga M., Hakushi T., (1983) J. Org. Chem. 48: 3168
Gaikwade A.G., Noguchi H., Yoshio M., (1991) Anal. Lett. 24: 1625
Sanz-Medel A., Gomis B.D., Garcia A.J.R., (1981) Talanta 28:425
Abrodo P.A., Gomis B.D., Sanz-Medel A., (1984) Microchem. J. 30: 58
Escobar R., Lamoneda C., Pablos F., Guiraum A., (1989) Analyst 4: 114
Takeda Y., Takagi C., (1994) J. Incl. Phenom. 17: 93
Takeda Y., Kawarabayashi A., Takahashi K., Kudo Y., (1995) Bull. Chem. Soc. Jpn. 68: 1309
Çakı ır Ü., Çiçek B., Yı ıldı ız Y.K., Alkan M., (1999) J. Incl. Phenom. 34: 153
Namor D.A.F., Puglies A., Casal A.R., Lierena M.B., (2000) Phys. Chem. Chem. Phys.2: 4355
Perez-Caballero M.G., Amirez-Galicia R.G., Hernandez P., Rubio M.F., (2001) J. Mol. Struct. (THEOCHEM) 535: 131
Zyablikova T.A., Ishmaeva E.A., Kataev V.E., Vereshchagina Y.A., Bazhanova Z.G., Il’yasov A.V., Terent’eva S.A., Pudovik M.A., (2004) Russ. J. Gen. Chem. 74: 1171
Hay B.P., Rustad J.R., (1994) J. Am. Chem. Soc. 116: 6316
Hancock R.D., (1990) Acc. Chem. Res. 23: 253
Vaden T.D., Lisy J.M., (2005) J. Phys. Chem. A. 109: 3880
Okano K., Tsukube H., Hori K., (2004) Tetrahedron 60: 10877
Freidzon A.Y., Bagatur’yants A.A., Gromov S.P., Alfimov M.V., (2003) Russ. Chem. Bull. Int. Ed. 52: 2646
Xu J.W., Lin T.T., Lai Y.H., (2005) Tetrahedron 61: 2431
Didier S., Gaudel-Siri A., Pons J.M., Liotard D., Rajzmann M., (2002) J. Mol. Struct. (THEOCHEM) 588: 71
(a) R.B. de Alencastro, J.A.S. Bomfim, C.A.L. Filgueiras, R.A. Howie, and J.L. Wardell: Appl. Organometal. Chem. 19, 479 (2005); (b) A. Azizoglu: Struct. Chem. 14, 575 (2003)
Khohlova S.S., Lebedev N.G., Bondarev S.L., Knyukshto V.N., Turban A.A., Mikhailova V.A., Ivanov A.I., (2005) Int. J. Quant. Chem. 104: 189
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
UĞRAŞ, H., ÇAKIR, Ü., AZİZOĞLU, A. et al. Experimental, Theoretical and Biological Activity Study on the Acyl-Substituted Benzo-18-crown-6, Dibenzo-18-crown-6 and Dibenzo-24-crown-8. J Incl Phenom Macrocycl Chem 55, 159–165 (2006). https://doi.org/10.1007/s10847-005-9032-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-005-9032-7