Abstract
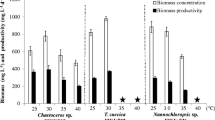
Microalgae are considered as a promising feedstock for biomass production. The selection of the most suitable species is based on several key parameters such as lipid and fatty acid productivity. In the present study, the growth of different microalgae strains was examined in freshwater media for photoautotrophs suited for large-scale applications to identify the most suitable medium for each species. In the optimal medium, Scenedesmus obliquus showed the highest biomass productivity measured as increase of cell dry weight (0.25 g cellu dry weight (CDW) L−1 day−1), while Botryococcus braunii showed the highest lipid and total fatty acid content (430 and 270 mg g−1 CDW, respectively) among the tested species. Regarding lipid and total fatty acid productivity, S. obliquus was the most lipid and total fatty acid productive strain with 41 and 18 mg L−1 day−1 during the exponential phase, respectively. Additionally, the proportion of saturated and monounsaturated fatty acids increased with duration of the incubation in S. obliquus, while polyunsaturated fatty acids decreased. These results nominate S. obliquus as a promising microalga in order to serve as a feedstock for renewable energy production.



Similar content being viewed by others
References
Bigogno C, Khozin-Goldberg I, Boussiba S, Vonshak A, Cohen Z (2002) Lipid and fatty acid composition of the green oleaginous alga Parietochloris incisa, the richest plant source of arachidonic acid. Phytochemistry 60:497–503
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Borowitzka MA (1992) Algal biotechnology products and processes-matching science and economics. J Appl Phycol 4:267–279
Folch J, Lees M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Francisco ÉC, Neves DB, Jacob-Lopes E, Franco TT (2010) Microalgae as feedstock for biodiesel production: carbon dioxide sequestration, lipid production and biofuel quality. J Chem Technol Biotechnol 85:395–403
Griffiths MJ, Harrison STL (2009) Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J Appl Phycol 21:493–507
Hindersin S, Leupold M, Kerner M, Hanelt D (2012) Irradiance optimization of outdoor microalgal cultures using solar tracked photobioreactors. Bioprocess Biosyst Eng. doi:10.1007/s00449-012-0790-5
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54:621–639
Kaczmarzyk D, Fulda M (2010) Fatty acid activation in Cyanobacteria mediated by acyl-acyl carrier protein synthetase enables fatty acid recycling. Plant Physiol 152:1598–1610
Kessler E, Czygan FC (1970) Physiologische und biochemische Beiträge zur Taxonomie der Gattung Chlorella. Arch Mikrobiol 70:211–216
Muñoz R, Guieysse B (2006) Algal-bacterial processes for the treatment of hazardous contamination: a review. Water Res 40:2799–2815
Scharnewski M, Pongdontri P, Mora G, Hoppert M, Fulda M (2008) Mutants of Saccharomyces cerevisiae deficient in acyl-CoA synthetases secrete fatty acids due to interrupted fatty acid recycling. FEBS J 275:2765–2777
Scott SA, Davey MP, Dennis JS, Horst I, Howe CJ, Lea-Smith DJ, Smith AG (2010) Biodiesel from algae: challenges and prospects. Curr Opin Biotechnol 21:277–286
Sheehan J, Dunahay T, Benemann J, Roessler P (1998) A look back at the U.S. Department of Energy’s Aquatic Species Program: biodiesel from algae. Golden, Colorado
Silva TL, Reis A, Medeiros R, Oliveira AC, Gouveia L (2009) Oil production towards biofuel from autotrophic microalgae semicontinuous cultivations monitorized by flow cytometry. Appl Biochem Biotechnol 159:568–578
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. J Biosci Bioeng 101:87–96
Stanier RY, Kunisawa R, Mandel M (1971) Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol Rev 35:171–205
Stein J (ed) (1973) Handbook of phycological methods: culture methods and growth measurements. Cambridge University Press, Cambridge
Yoo C, Jun S, Lee J, Ahn C, Oh H (2010) Selection of microalgae for lipid production under high levels carbon dioxide. Bioresour Technol 101:571–574
Acknowledgments
We thank Sabine Beckmann (Institute for Biogeochemistry and Marine Chemistry, University of Hamburg) for assistance with the GCMS analysis and Sigrid Mörke (Department of Cell Biology and Phycology, University of Hamburg) for her excellent expert technical assistance. Thanks also for Dr. Martin Kerner (SSC GmbH) for his contribution in writing the manuscript. This work was supported by grants from Arbeitsgemeinschaft industrieller Forschungsvereinigungen “Otto von Guericke” e. V. (KF2353701SB9 to D. H.) and by joint grants from Egyptian Ministry of Higher Education and Scientific Research and Deutscher Akademischer Austauschdienst in a German Egyptian Research Long-Term Scholarship program (to A. A.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 8 kb)
Rights and permissions
About this article
Cite this article
Abomohra, A.EF., Wagner, M., El-Sheekh, M. et al. Lipid and total fatty acid productivity in photoautotrophic fresh water microalgae: screening studies towards biodiesel production. J Appl Phycol 25, 931–936 (2013). https://doi.org/10.1007/s10811-012-9917-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-012-9917-y