Abstract
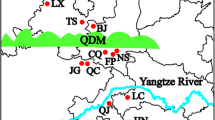
Major histocompatibility complex (MHC) genes are excellent markers for the study of adaptive genetic variation occurring over different geographical scales. The Chinese egret (Egretta eulophotes) is a vulnerable ardeid species with an estimated global population of 2600–3400 individuals. In this study, we sampled 172 individuals of this egret (approximately 6 % of the global population) from five natural populations that span the entire distribution range of this species in China. We examined their population genetic diversity and geographical differentiation at three MHC class II DAB genes by identifying eight exon 2 alleles at Egeu-DAB1, eight at Egeu-DAB2 and four at Egeu-DAB3. Allelic distributions at each of these three Egeu-DAB loci varied substantially within the five populations, while levels of genetic diversity varied slightly among the populations. Analysis of molecular variance showed low but significant genetic differentiation among five populations at all three Egeu-DAB loci (haplotype-based ϕST: 0.029, 0.020 and 0.042; and distance-based ϕST: 0.036, 0.027 and 0.043, respectively; all P < 0.01). The Mantel test suggested that this significant population genetic differentiation was likely due to an isolation-by-distance pattern of MHC evolution. However, the phylogenetic analyses and the Bayesian clustering analysis based on the three Egeu-DAB loci indicated that there was little geographical structuring of the genetic differentiation among five populations. These results provide fundamental population information for the conservation genetics of the vulnerable Chinese egret.




Similar content being viewed by others
References
Acevedo-Whitehouse K, Cunningham AA (2006) Is MHC enough for understanding wildlife immunogenetics? Trends Ecol Evol 21:433–438. doi:10.1016/j.tree.2006.05.010
Addis BR, Lowe WH, Hossack BR, Allendorf FW (2015) Population genetic structure and disease in montane boreal toads: more heterozygous individuals are more likely to be infected with amphibian chytrid. Conserv Genet 16:833–844. doi:10.1007/s10592-015-0704-6
Aguilar A, Smith TB, Wayne RK (2005) A comparison of variation between a MHC pseudogene and microsatellite loci of the little greenbul (Andropadus virens). BMC Evol Biol 5:47. doi:10.1186/1471-2148-5-47
Alcaide M, Edwards SV, Negro JJ, Serrano D, Tella JL (2008) Extensive polymorphism and geographical variation at a positively selected MHC class II B gene of the lesser kestrel (Falco naumanni). Mol Ecol 17:2652–2665. doi:10.1111/j.1365-294X.2008.03791.x
Balakrishnan CN, Ekblom R, Völker M, Westerdahl H, Godinez R, Kotkiewicz H, Burt DW, Graves T, Griffin DK, Warren WC, Edwards SV (2010) Gene duplication and fragmentation in the zebra finch major histocompatibility complex. BMC Biol 8:29. doi:10.1186/1741-7007-8-29
Bevan MJ (1987) Class discrimination in the world of immunology. Nature 325:192–194. doi:10.1038/325192b0
Bichet C, Moodley Y, Penn DJ, Sorci G, Garnier S (2015) Genetic structure in insular and mainland populations of house sparrows (Passer domesticus) and their hemosporidian parasites. Ecol Evol 5:1639–1652. doi:10.1002/ece3.1452
BirdLife International (2015) Species factsheet: Egretta eulophotes. Available via DIALOG. http://www.birdlife.org. Accessed 27 Jan 2015
Corrêa TC, Del Lama SN, De Souza JR, Miño CI (2015) Genetic structuring among populations of the great egret, Ardea alba egretta, in major Brazilian wetlands. Aquat Conserv Mar Freshw Ecosyst 26:333–349. doi:10.1002/aqc.2588
Doherty PC, Zinkernagel RM (1975) Enhanced immunological surveillance in mice heterozygous at H-2 gene complex. Nature 256:50–52. doi:10.1038/256050a0
Ekblom R, Saether SA, Jacobsson P, Fiske P, Sahlman T, Grahn M, Kålås JA, Höglund J (2007) Spatial pattern of MHC class II variation in the great snipe (Gallinago media). Mol Ecol 16:1439–1451. doi:10.1111/j.1365-294X.2007.03281.x
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. doi:10.1111/j.1365-294X.2005.02553.x
Excoffier L, Lischer HE (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. doi:10.1111/j.1755-0998.2010.02847.x
Fabiani A, Hoelzel AR, Galimberti F, Muelbert MMC (2003) Long-range paternal gene flow in the southern elephant seal. Science 299:676. doi:10.1126/science.299.5607.676
Falush D, Stephens M, Pritchard JK (2003) Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics 164:1567–1587. doi:10.3410/f.1015548.197423
Funk WC, McKay JK, Hohenlohe PA, Allendorf FW (2012) Harnessing genomics for delineating conservation units. Trends Ecol Evol 27:489–496. doi:10.1016/j.tree.2012.05.012
Germain RN, Castellino F, Han R, Sousa CRE, Romagnoli P, Sadegh-Nasseri S, Zhong G (1996) Processing and presentation of endocytically acquired protein antigens by MHC class II and class I molecules. Immunol Rev 151:5–30. doi:10.1111/j.1600-065X.1996.tb00701.x
Goudet J (1995) FSTAT, a program to estimate and test gene diversities and fixation indices (version 1.2). J Hered 86:485–486
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hughes AL (1991) MHC polymorphism and the design of captive breeding programs. Conserv Biol 5:249–251. doi:10.1111/j.1523-1739.1991.tb00130.x
Hughes AL, Nei M (1992) Maintenance of MHC polymorphism. Nature 355:402–403. doi:10.1038/355402b0
Hull JM, Anderson R, Bradbury M, Estep JA, Ernest HB (2008) Population structure and genetic diversity in Swainson’s Hawks (Buteo swainsoni): implications for conservation. Conserv Genet 9:305–316. doi:10.1007/s10592-007-9342-y
IUCN (2015) IUCN Red list of threatened species. Available via DIALOG. http://www.iucnredlist.org. Accessed 27 Jan 2015
Janeway CA, Murphy K, Travers P, Ehrenstein M (2008) Janeway’s immunobiology. Garland, New York
Jensen JL, Bohonak AJ, Kelley ST (2005) Isolation by distance, web service. BMC Genet 6:13. doi:10.1186/1471-2156-6-13
Jones MR, Cheviron ZA, Carling MD (2014) Variation in positively selected major histocompatibility complex class I loci in rufous-collared sparrows (Zonotrichia capensis). Immunogenetics 66:693–704. doi:10.1007/s00251-014-0800-7
Klareskog L, Sandgerg-Tragardh L, Rask L, Lindblom JB, Curman B, Peterson PA (1977) Chemical properties of human Ia antigens. Nature 265:248–251. doi:10.1038/265248a0
Klein J (1986) Natural history of the major histocompatibility complex. Wiley, New York
Klein J, Bontrop RE, Dawkins RL, Erlich HA, Gyllensten UB, Heise ER, Jones PP, Wakeland EK, Watkins DI (1990) Nomenclature for major histocompatibility complexes of different species: a proposal. Immunogenetics 31:217–219. doi:10.1007/978-3-642-77506-2_32
Klein J, Satta Y, O’hUigin C, Takahata N (1993) The molecular descent of the major histocompatibility complex. Annu Rev Immunol 11:269–295. doi:10.1146/annurev.iy.11.040193.001413
Kohyama TI, Omote K, Nishida C, Takenaka T, Saito K, Fujimoto S, Masuda R (2015) Spatial and temporal variation at major histocompatibility complex class IIB genes in the endangered Blakiston’s fish owl. Zool Lett 1:13. doi:10.1186/s40851-015-0013-4
Koutsogiannouli EA, Moutou KA, Stamatis C, Walter L, Mamuris Z (2014) Genetic variation in the major histocompatibility complex of the European brown hare (Lepus europaeus) across distinct phylogeographic areas. Immunogenetics 66:379–392. doi:10.1007/s00251-014-0772-7
Kushlan JA, Hancock JA (2005) The Herons. Oxford University Press, Oxford
Lanfear R, Calcott B, Ho SY, Guindon S (2012) Partitionfinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol Biol Evol 29:1695–1701. doi:10.1093/molbev/mss020
Lei W, Fang W, Lin Q, Zhou X, Chen X (2015) Characterization of a non-classical MHC class II gene in the vulnerable Chinese egret (Egretta eulophotes). Immunogenetics 67:463–472. doi:10.1007/s00251-015-0846-1
Lei W, Zhou X, Fang W, Lin Q, Chen X (2016) Major histocompatibility complex class II DAB alleles associated with intestinal parasite load in the vulnerable Chinese egret (Egretta eulophotes). Ecol Evol 6:4421–4434. doi:10.1002/ece3.2226
Li L, Zhou X, Chen X (2011) Characterization and evolution of MHC class II B genes in ardeid birds. J Mol Evol 72:474–483. doi:10.1007/s00239-011-9446-3
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. doi:10.1093/bioinformatics/btp187
Lillie M, Grueber CE, Sutton JT, Howitt R, Bishop PJ, Gleeson D, Belov K (2015) Selection on MHC class II supertypes in the New Zealand endemic Hochstetter’s frog. BMC Evol Biol 15:63. doi:10.1186/s12862-015-0342-0
Luo M, Pan H, Liu Z, Li M (2012) Balancing selection and genetic drift at major histocompatibility complex class II genes in isolated populations of golden snub-nosed monkey (Rhinopithecus roxellana). BMC Evol Biol 12:207. doi:10.1186/1471-2148-12-207
Martínez-Cruz B, Godoy JA, Negro JJ (2004) Population genetics after fragmentation: the case of the endangered Spanish imperial eagle (Aquila adalberti). Mol Ecol 13:2243–2255. doi:10.1111/j.1365-294X.2004.02220.x
Meyers LA, Bull JJ (2002) Fighting change with change: adaptive variation in an uncertain world. Trends Ecol Evol 17:551–557. doi:10.1016/S0169-5347(02)02633-2
Miller HC, Lambert DM (2004) Genetic drift outweighs balancing selection in shaping post-bottleneck major histocompatibility complex variation in New Zealand robins (Petroicidae). Mol Ecol 13:3709–3721. doi:10.1111/j.1365-294X.2004.02368.x
Morin PA, Luikart G, Wayne RK, The SNP workshop group (2004) SNPs in ecology, evolution and conservation. Trends Ecol Evol 19:208–216. doi:10.1016/j.tree.2004.01.009
Nei M, Rooney AP (2005) Concerted and birth-and-death evolution of multigene families. Annu Rev Genet 39:121–152. doi:10.1146/annurev.genet.39.073003.112240
Nguyen-Phuc H, Fulton JE, Berres ME (2016) Genetic variation of major histocompatibility complex (MHC) in wild Red Junglefowl (Gallus gallus). Poult Sci 95:400–411. doi:10.3382/ps/pev364
Penn DJ, Potts WK (1999) The evolution of mating preferences and major histocompatibility genes. Am Nat 153:145–164. doi:10.1086/303166
Piertney SB (2003) Major histocompatibility complex B-LB gene variation in red grouse Lagopus lagopus scoticus. Wildl Biol 9:251–259
Piertney SB, Oliver MK (2006) The evolutionary ecology of the major histocompatibility complex. Heredity 96:7–21. doi:10.1038/sj.hdy.6800724
Poelstra JW, Vijay N, Bossu CM, Lantz H, Ryll B, Muller I, Baglione V, Unneberg P, Wikelski M, Grabherr MG, Wolf JBW (2014) The genomic landscape underlying phenotypic integrity in the face of gene flow in crows. Science 344:1410–1414. doi:10.1126/science.1253226
Promerová M, Králová T, Bryjová A, Albrecht T, Bryja J (2013) MHC class IIB exon 2 polymorphism in the Grey partridge (Perdix perdix) is shaped by selection, recombination and gene conversion. PLoS One 8:e69135. doi:10.1371/journal.pone.0069135
Rousset F (2008) GENEPOP’007: a complete re-implementation of the GENEPOP software for Windows and Linux. Mol Ecol Resour 8:103–106. doi:10.1111/j.1471-8286.2007.01931.x
Schut E, Rivero-de Aguilar J, Merino S, Magrath MJ, Komdeur J, Westerdahl H (2011) Characterization of MHC-I in the blue tit (Cyanistes caeruleus) reveals low levels of genetic diversity and trans-population evolution across European populations. Immunogenetics 63:531–542. doi:10.1007/s00251-011-0532-x
Slatkin M (1993) Isolation by distance in equilibrium and non-equilibrium populations. Evolution 47:264–279. doi:10.2307/2410134
Smith S, Goüy de Bellocq J, Suchentrunk F, Schaschl H (2011) Evolutionary genetics of MHC class II beta genes in the brown hare, Lepus europaeus. Immunogenetics 63:743–751. doi:10.1007/s00251-011-0539-3
Sommer S (2005) The importance of immune gene variability (MHC) in evolutionary ecology and conservation. Front Zool 2:16. doi:10.1186/1742-9994-2-16
Spurgin LG, Richardson DS (2010) How pathogens drive genetic diversity: MHC, mechanisms and misunderstandings. Proc R Soc B 277:979–988. doi:10.1098/rspb.2009.2084
Spurgin LG, van Oosterhout C, Illera JC, Bridgett S, Gharbi K, Emerson BC, Richardson DS (2011) Gene conversion rapidly generates major histocompatibility complex diversity in recently founded bird populations. Mol Ecol 20:5213–5225. doi:10.1111/j.1365-294X.2011.05367.x
Takahata N, Nei M (1990) Allelic genealogy under overdominant and frequency-dependent selection and polymorphism of major histocompatibility complex loci. Genetics 124:967–978
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA 6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. doi:10.1093/molbev/mst197
Vásquez-Carrillo C, Friesen F, Hall L, Peery MZ (2013) Variation in MHC class II B genes in marbled murrelets: implications for delineating conservation units. Anim Conserv 17:244–255. doi:10.1111/acv.12089
Wang Z, Zhou X, Lin Q, Fang W, Chen X (2013) Characterization, polymorphism and selection of major histocompatibility complex (MHC) DAB genes in vulnerable Chinese egret (Egretta eulophotes). PLoS One 8:e74185. doi:10.1371/journal.pone.0074185
Weber DS, Stewart BS, Schienman J, Lehman N (2004) Major histocompatibility complex variation at three class II loci in the northern elephant seal. Mol Ecol 13:711–718. doi:10.1111/j.1365-294X.2004.02095.x
Witzenberger KA, Hochkirch A (2011) Ex situ conservation genetics: a review of molecular studies on the genetic consequences of captive breeding programmes for endangered animal species. Biodivers Conserv 20:1843–1861. doi:10.1007/s10531-011-0074-4
Zeng Q, He K, Sun D, Ma M, Ge Y, Fang S, Wan Q (2016) Balancing selection and recombination as evolutionary forces caused population genetic variations in golden pheasant MHC class I genes. BMC Evol Biol 16:42. doi:10.10.1186/s12862-016-0609-0
Zhou X, Fang W, Chen X (2010) Mitochondrial DNA diversity of the vulnerable Chinese Egret (Egretta eulophotes) from China. J Ornithol 151:409–414. doi:10.1007/s10336-009-0470-7
Zhu Y, Wan Q, Yu B, Ge Y, Fang S (2013) Patterns of genetic differentiation at MHC class I genes and microsatellites identify conservation units in the giant panda. BMC Evol Biol 13:227. doi:10.1186/1471-2148-13-227
Acknowledgments
We thank Yufei Dai who helped collect some samples for this study. This work was funded by the National Natural Science Foundation of China (Grant Nos. 41476113 and 31272333) and by the Fujian Natural Science Foundation of China (2010Y2007).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
We declare that we have no conflict of interests.
Ethical approval
All procedures involving collection of animal tissue in the wild were approved by the Administration Center for Wildlife Conservation in Fujian Province (FJWCA-1208) and were in accordance with its ethical standards.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10592_2016_876_MOESM1_ESM.doc
Fig. S1 Mean of log probability of L (K) and Delta K over 10 runs for each K value. Supplementary material 1 (DOC 1230 kb)
Rights and permissions
About this article
Cite this article
Lei, W., Fang, W., Zhou, X. et al. Population genetic diversity and geographical differentiation of MHC class II DAB genes in the vulnerable Chinese egret (Egretta eulophotes). Conserv Genet 17, 1459–1468 (2016). https://doi.org/10.1007/s10592-016-0876-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-016-0876-8