Abstract
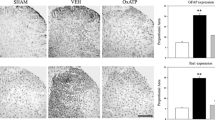
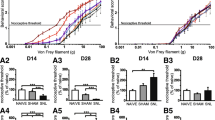
Reactive astrocytes and activated microglia are the key players in several pathophysiologic modifications of the central nervous system. We used the spared nerve injury (SNI) of the sciatic nerve to induce glial maladaptive response in the ventral horn of lumbar spinal cord and examine its role in the remodeling of the tripartite synapse plasticity. Imaging the ventral horn revealed that SNI was associated with both an early microglial and astrocytic activation, assessed, respectively, by analysis of Iba1 and GFAP expression. Microglia, in particular, localized peculiarly surrounding the motor neurons somata. Perineuronal astrocytes, which play a key role in maintaining the homeostasis of neuronal circuitry, underwent a substantial phenotypic change following peripheral axotomy, producing reactive gliosis. The gliosis was associated with the reduction of glial aminoacid transporters (GLT1 and GlyT1) and increase of neuronal glutamate transporter EAAC1. Although the expression of GABAergic neuronal marker GAD65/67 showed no change, glutamate increase, as demonstrated by HPLC analysis, shifted the excitatory/inhibitory balance as showed by the net increase of the glutamate/GABA ratio. Moreover, endogenous NGF levels were altered in SNI animals and not restored by the intrathecal NGF administration. This treatment reverted phenotypic changes associated with reactive astrocytosis, but failed to modify microglia activation. These findings on one hand confirm the correlation between gliopathy and maladaptive plasticity of the spinal synaptic circuitry, on the other hand add new data concerning the complex peculiar behavior of different glial cells in neuronal degenerative processes, defining a special role of microglia in sustaining the inflammatory response.





Similar content being viewed by others
References
Blits B, Oudega M, Boer GJ, Bartlett Bunge M, Verhaagen J (2003) Adeno-associated viral vector-mediated neurotrophin gene transfer in the injured adult rat spinal cord improves hind-limb function. Neuroscience 118(1):271–281
Bruno MA, Cuello AC (2006) Activity-dependent release of precursor nerve growth factor, conversion to mature nerve growth factor, and its degradation by a protease cascade. Proc Natl Acad Sci USA 103(17):6735–6740
Carulli D, Foscarin S, Faralli A, Pajaj E, Rossi F (2013) Modulation of semaphorin3A in perineuronal nets during structural plasticity in the adult cerebellum. Mol Cell Neurosci 57:10–22
Cavaliere C, Cirillo G, Rosaria Bianco M, Rossi F, De Novellis V, Maione S, Papa M (2007) Gliosis alters expression and uptake of spinal glial amino acid transporters in a mouse neuropathic pain model. Neuron Glia Biol 3(2):141–153
Cirillo G, Cavaliere C, Bianco MR, De Simone A, Colangelo AM, Sellitti S, Alberghina L, Papa M (2010) Intrathecal NGF administration reduces reactive astrocytosis and changes neurotrophin receptors expression pattern in a rat model of neuropathic pain. Cell Mol Neurobiol 30(1):51–62
Cirillo G, Bianco MR, Colangelo AM, Cavaliere C, de Daniele L, Zaccaro L, Alberghina L, Papa M (2011) Reactive astrocytosis-induced perturbation of synaptic homeostasis is restored by nerve growth factor. Neurobiol Dis 41(3):630–639
Cirillo G, Colangelo AM, Bianco MR, Cavaliere C, Zaccaro L, Sarmientos P, Alberghina L, Papa M (2012) BB14, a Nerve Growth Factor (NGF)-like peptide shown to be effective in reducing reactive astrogliosis and restoring synaptic homeostasis in a rat model of peripheral nerve injury. Biotechnol Adv 30(1):223–232
Cirillo G, Colangelo AM, Berbenni M, Ippolito VM, De Luca C, Verdesca F, Savarese L, Alberghina L, Maggio N, Papa M (2014) Purinergic modulation of spinal neuroglial maladaptive plasticity following peripheral nerve injury. Mol Neurobiol. doi:10.1007/s12035-014-8943-y
Cobianchi S, Casals-Diaz L, Jaramillo J, Navarro X (2013) Differential effects of activity dependent treatments on axonal regeneration and neuropathic pain after peripheral nerve injury. Exp Neurol 240:157–167
Coderre TJ, Melzack R (1992) The contribution of excitatory amino acids to central sensitization and persistent nociception after formalin-induced tissue injury. J Neurosci 12(9):3665–3670
Colangelo AM, Bianco MR, Vitagliano L, Cavaliere C, Cirillo G, De Gioia L, Diana D, Colombo D, Redaelli C, Zaccaro L, Morelli G, Papa M, Sarmientos P, Alberghina L, Martegani E (2008) A new nerve growth factor-mimetic peptide active on neuropathic pain in rats. J Neurosci 28(11):2698–2709
Colangelo AM, Cirillo G, Lavitrano ML, Alberghina L, Papa M (2012) Targeting reactive astrogliosis by novel biotechnological strategies. Biotechnol Adv 30(1):261–271
Corvetti L, Rossi F (2005) Degradation of chondroitin sulfate proteoglycans induces sprouting of intact purkinje axons in the cerebellum of the adult rat. J Neurosci 25(31):7150–7158
Cullheim S, Thams S (2007) The microglial networks of the brain and their role in neuronal network plasticity after lesion. Brain Res Rev 55(1):89–96
Davis-López de Carrizosa MA, Morado-Díaz CJ, Tena JJ, Benítez-Temiño B, Pecero ML, Morcuende SR, de la Cruz RR, Pastor AM (2009) Complementary actions of BDNF and neurotrophin-3 on the firing patterns and synaptic composition of motoneurons. J Neurosci 29(2):575–587
Decosterd I, Woolf CJ (2000) Spared nerve injury: an animal model of persistent peripheral neuropathic pain. Pain 87(2):149–158
Foscarin S, Ponchione D, Pajaj E, Leto K, Gawlak M, Wilczynski GM, Rossi F, Carulli D (2011) Experience-dependent plasticity and modulation of growth regulatory molecules at central synapses. PLoS One 6(1):e16666
Gordon T, You S, Cassar SL, Tetzlaff W (2015) Reduced expression of regeneration associated genes in chronically axotomized facial motoneurons. Exp Neurol 264:26–32
Haftel VK, Bichler EK, Wang QB, Prather JF, Pinter MJ, Cope TC (2005) Central suppression of regenerated proprioceptive afferents. J Neurosci 25(19):4733–4742
Hua X, Malarkey EB, Sunjara V, Rosenwald SE, Li WH, Parpura V (2004) Ca(2+)-dependent glutamate release involves two classes of endoplasmic reticulum Ca(2+) stores in astrocytes. J Neurosci Res 76:86–97
Kwok JC, Dick G, Wang D, Fawcett JW (2011) Extracellular matrix and perineuronal nets in CNS repair. Dev Neurobiol 71(11):1073–1089
Liu H, Xu H, Yu T, Yao J, Zhao C, Yin ZQ (2013) Expression of perineuronal nets, parvalbumin and protein tyrosine phosphatase σ in the rat visual cortex during development and after BFD. Curr Eye Res 38(10):1083–1094
Lüscher C, Malenka RC (2012) NMDA receptor-dependent long-term potentiation and long-term depression (LTP/LTD). Cold Spring Harb Perspect Biol 4(6):a005710. doi:10.1101/cshperspect.a005710
Massey JM, Hubscher CH, Wagoner MR, Decker JA, Amps J, Silver J, Onifer SM (2006) Chondroitinase ABC digestion of the perineuronal net promotes functional collateral sprouting in the cuneate nucleus after cervical spinal cord injury. J Neurosci 26(16):4406–4414
McRae PA, Rocco MM, Kelly G, Brumberg JC, Matthews RT (2007) Sensory deprivation alters aggrecan and perineuronal net expression in the mouse barrel cortex. J Neurosci 27(20):5405–5413
Meisner JG, Marsh AD, Marsh DR (2010) Loss of GABAergic interneurons in laminae I–III of the spinal cord dorsal horn contributes to reduced GABAergic tone and neuropathic pain after spinal cord injury. J Neurotrauma 27(4):729–737
Milligan ED, Watkins LR (2009) Pathological and protective roles of glia in chronic pain. Nat Rev Neurosci 10(1):23–36
Molteni R, Zheng JQ, Ying Z, Gómez-Pinilla F, Twiss JL (2004) Voluntary exercise increases axonal regeneration from sensory neurons. Proc Natl Acad Sci USA 101(22):8473–8478
Morris GP, Clark IA, Zinn R, Vissel B (2013) Microglia: a new frontier for synaptic plasticity, learning and memory, and neurodegenerative disease research. Neurobiol Learn Mem 105:40–53
Murdock BJ, Bender DE, Segal BM, Feldman EL (2015) The dual roles of immunity in ALS: injury overrides protection. Neurobiol Dis 77:1–12
Novikov LN, Novikova LN, Holmberg P, Kellerth J (2000) Exogenous brain-derived neurotrophic factor regulates the synaptic composition of axonally lesioned and normal adult rat motoneurons. Neuroscience 100(1):171–181
Papa M, De Luca C, Petta F, Alberghina L, Cirillo G (2014) Astrocyte-neuron interplay in maladaptive plasticity. Neurosci Biobehav Rev 42:35–54
Raineteau O, Schwab ME (2001) Plasticity of motor systems after incomplete spinal cord injury. Nat Rev Neurosci 2(4):263–273
Schwab ME, Strittmatter SM (2014) Nogo limits neural plasticity and recovery from injury. Curr Opin Neurobiol 27:53–60
Su H, Zhang W, Guo J, Guo A, Yuan Q, Wu W (2009) Neural progenitor cells enhance the survival and axonal regeneration of injured motoneurons after transplantation into the avulsed ventral horn of adult rats. J Neurotrauma 26(1):67–80
Svensson M, Eriksson P, Persson JK, Molander C, Arvidsson J, Aldskogius H (1993) The response of central glia to peripheral nerve injury. Brain Res Bull 30(3–4):499–506
Takahashi-Iwanaga H, Murakami T, Abe K (1998) Three-dimensional microanatomy of perineuronal proteoglycan nets enveloping motor neurons in the rat spinal cord. J Neurocytol 27(11):817–827
Verkhratsky A, Kirchhoff F (2007) Glutamate-mediated neuronal-glial transmission. J Anat 210(6):651–660
Wang D, Ichiyama RM, Zhao R, Andrews MR, Fawcett JW (2011) Chondroitinase combined with rehabilitation promotes recovery of forelimb function in rats with chronic spinal cord injury. J Neurosci 31(25):9332–9344
Acknowledgments
This work was supported by Grants from the Italian Minister of University and Research (PRIN2007 to M.P. and to A.M.C.); SYSBIONET—Italian ROADMAP ESFRI Infrastructures to L.A., A.M.C., M.P. (fellowship to L.S.); FIRB-ITALBIONET and NEDD to L.A.; Blueprint Pharma s.r.l.; PRIMM, s.r.l.. Research work in authors’ laboratory was funded by Grants from Regione Campania (L.R. N.5 Bando 2003 to M.P.), Regione Campania (Prog. Spec art 12 E.F. 2000 to M.P.), CNR (Neurobiotecnologie 2003 to M.P.), and Associazione Levi-Montalcini (fellowships to G.C.).
Conflict of interest
The authors declare that this article has no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Luca, C., Savarese, L., Colangelo, A.M. et al. Astrocytes and Microglia-Mediated Immune Response in Maladaptive Plasticity is Differently Modulated by NGF in the Ventral Horn of the Spinal Cord Following Peripheral Nerve Injury. Cell Mol Neurobiol 36, 37–46 (2016). https://doi.org/10.1007/s10571-015-0218-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-015-0218-2