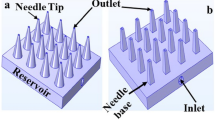
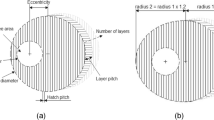
Abstract
In this paper, we present design, fabrication and coupled multifield analysis of hollow out-of-plane silicon microneedles with piezoelectrically actuated microfluidic device for transdermal drug delivery (TDD) system for treatment of cardiovascular or hemodynamic disorders such as hypertension. The mask layout design and fabrication process of silicon microneedles and reservoir involving deep reactive ion etching (DRIE) is first presented. This is followed by actual fabrication of silicon hollow microneedles by a series of combined isotropic and anisotropic etching processes using inductively coupled plasma (ICP) etching technology. Then coupled multifield analysis of a MEMS based piezoelectrically actuated device with integrated silicon microneedles is presented. The coupledfield analysis of hollow silicon microneedle array integrated with piezoelectric micropump has involved structural and fluid field couplings in a sequential structural-fluid analysis on a three-dimensional model of the microfluidic device. The effect of voltage and frequency on silicon membrane deflection and flow rate through the microneedle is investigated in the coupled field analysis using multiple code coupling method. The results of the present study provide valuable benchmark and prediction data to fabricate optimized designs of the silicon hollow microneedle based microfluidic devices for transdermal drug delivery applications.



















Similar content being viewed by others
References
Aggarwal P, Johnston CR. Geometrical effects in mechanical characterizing of microneedle for biomedical applications. Sens Actuators B. 2004;102:226–34.
Ahmadian M, Saidi M, Mehrabian A, Bazargan M, Kenarsari S. Performance of valveless diffuser micropumps under harmonic piezoelectric actuation. In: ASME conference on engineering systems design and analysis. 2006.
ANSI/IEEE Std 176. IEEE standard on piezoelectricity. IEEE; 1987. http://standards.ieee.org/reading/ieee/std_public/description/ultrasonics/176-1987_desc.html.
Aoyagi S, et al. Biodegradable polymer needle with various tip angles and consideration on insertion mechanism of mosquito’s proboscis. Sens Actuators A. 2008;143:20–8.
Arora A, Prausnitzc MR, et al. Micro-scale devices for transdermal drug delivery. Int J Pharm. 2008;364:227–36.
Barry BW. Novel mechanisms and devices to enable successful transdermal drug delivery. Eur J Pharm Sci. 2001;14:101–14.
Batchelor GK. An introduction to fluid dynamics. University of Cambridge. 1967.
BeMent SL, et al. Solid-state electrodes for multichannel multiplexed intracortical neuronal recording. IEEE Trans Biomed Eng. 1986;33(2):230–41.
Bodhale DW, Nisar A, Afzulpurkar N. Structural and microfluidic analysis of hollow side-open polymeric microneedles for transdermal drug delivery applications. Microfluid Nanofluid. 2009. doi:10.1007/s10404-009-0467-9.
Brown MB, Martin GP, et al. Dermal and transdermal drug delivery systems: current and future prospects. Drug Deliv. 2006;13:175–87.
Bussemer T, Otto I, Bodmeier R. Pulsatile drug delivery systems. Crit Rev Ther Drug Carrier Syst. 2001;18(5):433–58.
Campbell PK, et al. A silicon-based, three-dimensional neural interface: manufacturing processes for an intracortical electrode array. IEEE Trans Biomed Eng. 1991;38(8):758–68.
Chen J, et al. A multichannel neural probe for selective chemical delivery at the cellular level. IEEE Trans Biomed Eng. 1997;44(8):760–9.
Cui Q, Liu C, Xuan F. Study on a piezoelectric micropump for the controlled drug delivery system. Microfluid Nanofluidics. 2007;3(4):377–90.
Fan B, Song G, Hussain F. Simulation of piezoelectrically actuated valveless micropump. J Smart Mater Struct. 2005;14:400–5.
Frick TB, et al. Resistance forces acting on suture needles. J Biomech. 2001;34:1335–40.
Gardeniers HJGE, et al. Silicon micromachined hollow microneedles for transdermal liquid transport. J Microelctromech Syst. 2003;12(6).
Gere J, Timoshenko S. Mechanics of materials, 4th edn. 1997.
Glasgow I, Lieber S, Aubry N. Parameters influencing pulsed flow mixing in microchannels. Anal Chem. 2004a;76:4825–32.
Glasgow I, Batton J, Aubry N. Electroosmotic mixing in microchannels. Lab Chip. 2004b;4:558–62.
Griss P, Enoksson P, Tolvanen Laakso HK, Merilainen P, Ollmar S, Stemme G. Micromachined electrodes for biopotential measurements. IEEE ASME J Microelectromech Syst. 2001;10(1):10–6.
Griss P, Tolvanen Laakso H, Merilainen P, Stemme G. Characterization of micromachined spiked biopotential electrodes. IEEE Trans Biomed Eng. 2002;49(6):597–604.
Griss P, et al. Side-opened out of plane microneedles for microfluidics transdermal liquid transfer. J Microelectromech Syst. 2003;12(3):296–301.
Guo SX, Pei Z, Wang T, Ye XF. A novel type of pulseless output micropump based on magnet-solenoid actuator. In: IEEE/ICME international conference on complex medical engineering. 2007. p. 96–100.
Henry S, et al. Micro machined needles for the transdermal drug delivery of drugs. In: Proceedings of IEEE workshop MEMS. 1998. p. 494–98.
Izzo I, Accoto D, Menciassi A, Schmitt L, Dario P. Modelling and experimental validation of a piezoelectric micropump with novel no-moving-part valves. Sens Actuators A. 2007;133:128–40.
Jang LS, Li YJ, Lin SJ, Hsu YC, Yao WS, Tsai MC. A stand-alone peristaltic micropump based on piezoelectric actuation. Biomed Microdevices. 2007;9(2):185–94.
Janna WS. Design of fluid thermal system. 2nd ed. Boston: PWS Pub.; 1998.
Karande P, Jain A, et al. Discovery of transdermal penetration enhancers by high- throughput screening. Nat Biotechnol. 2004;22:192–7.
Khumpuang S, et al. Design and fabrication of coupled microneedle array and insertion guide array for safe penetration through skin. In: International symposium of micromechatronics and human science. 2003.
Kim K, Park D, Lu H, Kim K-H, Lee JB. A tapered hollow metallic microneedle array using backside exposure of SU-8. J Micromech Microeng. 2004;14:597–603.
Matteucci M, et al. A compact and disposable transdermal drug delivery system. Sincrotrone Trieste, I-34012 Basovizza-Trieste, Italy. 2008.
Moon SJ, Lee SS. A novel fabrication method of a microneedle array using inclined deep x-ray exposure. J Micromech Microeng. 2005;15:903–11.
Mukherjee EV, et al. Microneedle array for transdermal biological fluid extraction and in situ analysis. Sens Actuators A. 2004;114:267–75.
Nguyen NT, Huang XY, Chuan TK. MEMS-micropumps: a review. J Fluids Eng Trans ASME. 2002;124(2):384–92.
Nisar A, Afzulpurkar N, Tuantranont A, Mahaisavariya B. Three dimensional transient multifield analysis of a piezoelectric micropump for drug delivery system for treatment of hemodynamic dysfunctions. Cardiovasc Eng. 2008;8(4):203–18.
Oka K, Aoyagi S, Arai Y, Isono Y, Hashiguchi G, Fujita H. Fabrication of a microneedle for a trace blood test. Sens Actuators A. 2002;97–98:478–85.
Paik SJ, et al. In-plane single-crystal-silicon microneedles for minimally invasive micro fluidic systems. Sens Actuators A. 2004;114:276–84.
Park JH, Davis S, Yoon YK, Allen MG, Prausnitz MR. Micromachined biodegradable microstructures. In: 16th IEEE international conference on microelectro mechanical systems. Kyoto, Japan. 2003. p. 371–74.
Park JH, Allen MG, Prausnitz MR. Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery. J Control Release. 2005;104(1):51–66.
Prausnitz MR. Microneedles for transdermal drug delivery. Adv Drug Deliv Rev. 2004;56:581–7.
Schreier H, Bouwstra J. Liposomes and niosomes as topical drug carriers-dermal and transdermal drug-delivery. J Control Release. 1994;30:1–15.
Schuetz YB, Naik A, et al. Emerging strategies for the transdermal delivery of peptide and protein drugs. Expert Opin Drug Deliv. 2005;2:533–48.
Shibata T, et al. Fabrication and mechanical characterization of microneedle array for cell surgery. In: Actuators and microsystems conference. 2007. p. 719–22.
Stoeber B, Liepmann D. Fluid injection through out-of-plane microneedles. Micro technologies in medicine and biology. In: 1st annual international conference. Berkeley, CA. 2000.
Stoeber B, Liepmann D. Design, fabrication and testing of a MEMS syringe. Berkeley sensor and actuator center, University of California at Berkeley, CA. 2002.
Sun C, Huang K. Numerical characterization of the flow rectification of dynamic microdiffusers. J Micromech Microeng. 2006;16:1331–9.
Timoshenko S, Krienger Woinowsky S. Theory of plates and shells. 2nd ed. New York: McGraw-Hill; 1995.
Wang X, et al. A novel fabrication approach for microneedles using silicon micromachining technology. In: 1st IEEE international conference on NEMS. 2006. p. 545–49.
Wang C, Leu T, Sun J. Unsteady analysis of microvalves with no moving parts. J Mech. 2007;23:9–14.
Wilke N, et al. Silicon microneedle electrode array with temperature monitoring for electroporation. Sens Actuators A. 2005a;1090(123–124):319–25.
Wilke N, et al. Process optimization and characterization of silicon microneedles fabricated by wet etch technology. Micro Electron J. 2005b;36:650–6.
Yakhot A, Arad M, Ben-Dor G. Numerical investigation of a laminar pulsating flow in a rectangular duct. Int J Numer Methods Fluids. 1999;29:935–50.
Yao Q, Xu D, Pan L, Teo A, Ho W, Lee V, et al. CFD simulations of flows in valveless micropumps. Eng Appl Comput Fluid Mech. 2007;1:181–8.
Zahn JD, et al. Micro fabricated polysilicon microneedles for minimally invasive biomedical devices. Biomed Microdevices. 2000;2:295–303.
Zahn JD, et al. Continuous on-chip micropumping for microneedle enhanced drug delivery. Biomed Microdevices. 2004;6(3):183–90.
Acknowledgments
The authors would like to thank and acknowledge K. Saejok, C. Hruanun, Atthi N. Somwamg, and J. Supadech at Thai Microelectronics Center (TMEC), Thailand for providing DRIE facility and process for microneedle fabrication.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashraf, M.W., Tayyaba, S., Nisar, A. et al. Design, Fabrication and Analysis of Silicon Hollow Microneedles for Transdermal Drug Delivery System for Treatment of Hemodynamic Dysfunctions. Cardiovasc Eng 10, 91–108 (2010). https://doi.org/10.1007/s10558-010-9100-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10558-010-9100-5