Abstract
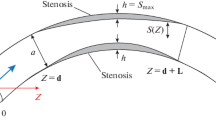
A powerful alternative means to studying hemodynamics in diseased or healthy coronary arteries can be achieved by providing a numerical model in which blood flow can be virtually simulated, for instance, using the computational fluid dynamics (CFD) method. In fact, it is well documented that CFD allows reliable physiological blood flow simulation and measurements of flow parameters. A requisite for obtaining reliable results from coronary CFD is to use exact anatomical models and realistic boundary conditions. To date, in almost all of the modeling studies on hemodynamics of stenosed coronary arteries, a velocity based boundary conditions has been assigned. The objective of this study is to show that inlet velocity actually depends on the degree of stenosis and thus for severe constriction in coronary artery, a velocity based boundary conditions cannot be realistic. We then prove that regardless of severity of stenosis in coronary arteries, the upstream pressure, systemic pressure, is always constant, thus, should be used as boundary conditions instead. The two sets of boundary conditions are implemented to demonstrate the robustness of each in modeling of stenosed coronary artery in a CFD study. These boundary conditions are applied in a stenosed cylindrical pipe including three categories of symmetrical stenosis (mild, moderate and severe stenosis starting from 15 to 95% diameter reduction) for steady state and pulsatile flow. Results strongly indicate that inlet velocity boundary conditions are no longer valid when effective diameter in stenosis goes below ~2.8 mm (a healthy diameter is considered 3.2 mm) which corresponds to 10–15% diameter reduction. Further work will determine the effect of flow reduction on the oxygen tension in blood to better define conditions for clinical symptoms such as angina.









Similar content being viewed by others
References
Back LD, Radbill JR, Crawford DW. Analysis of pulsatile, viscous blood flowthrough diseased coronary arteries of man. J Biomech. 1977;10:339–53.
Deplano V, Siouffi M. Experimental and numerical study of pulsatile flows through stenosis: wall shear stress analysis. J Biomech. 1999;32:1081–90.
Guo LR, Steinman DA, Moon BC, Wan WK, Millsap RJ. Effect of distal graft anastomosis site on retrograde perfusion and flow pattern of native coronary vasculature. Ann Thorac Surg. 2001;72:782–7.
Hu S, Wang X, Song Y, Lu F. Graft patency in off-pump and conventional coronary artery bypass grafting for treatment of triple vessel coronary diseases. Chin Med J. 2003;116:436–9.
Huang H, Modi VJ, Seymour BR. Fluid mechanics of stenosed arteries. Int J Eng Sci. 1995;33:815–28.
Ku DN, Zhu C. The mechanical environment of the artery. In: Sumpio BE, editor. Homodynamic forces and vascular cell biology. Austin, TX: R.G. Landes; 1993.
Lee JS, Fung YF. Flow in locally constricted tubes at low Reynolds numbers. ASME J Appl Mech. 1970;37:9–16.
Nichols WW, O’Rourke MF. McDonald’s blood flow in arteries. 3rd ed. Philadelphia: Lea & Febiger; 1990.
O’Brien V, Ehrlich LW. Simple pulsatile flow in an artery with a constriction. J Biomech. 1985;18:117–27.
Olifi EO, Kern MJ, Labovitz AJ, St. Vrain JA, Segal J, Aguirre FV, et al. Analysis of coronary blood flow velocity dynamics in angiographically normal and stenosed arteries before and after endolumen enlargement by angioplasty. J Am Coll Cardiol. 1993;21:308–16.
Papaharilaou Y, Doorly DJ, Sherwin SJ, Peiro J, Griffith C, Cheshire N, et al. “Combined MR imaging and numerical simulation of flow in realistic arterial bypass graft model. Biorheology. 2002;39:525–31.
Roe PL. Approximate Riemann solvers, parameter, vectors, and difference schemes. J Comput Phys. 1981;43:357–72.
Ryval J, Straatman AG, Steinman DA. Two-equation turbulence modeling of pulsatile flow in a stenosed tube. J Biomech Eng. 2004;126:625–30.
Schmidt A, Stefenelli T, Schuster E, Mayer G. Informational contribution of non-invasive screening tests for coronary artery disease in patients on chronic renal replacement therapy. Am J Kidney Dis. 2001;37:56–63.
Schoenhagen P, Nissen S. Understanding coronary artery disease: tomographic imaging with intravascular ultrasound. Heart. 2002;88:91–6.
Siouffi M, Deplano V, Pelissier R. Experimental analysis of unsteady flows through a stenosis. J Biomech. 1998;1:11–9.
Tu C, Deville M. Pulsatile flow of non-Newtonian fluids through arterial stenoses. J Biomech. 1996;29:899–908.
Tu C, Deville M, Dheur L, Vanderschuren L. Finite element simulation of pulsatile flow through arterial stenosis. J Biomech. 1992;25:1141–52.
Zendehbudi GR, Moayeri MS. Comparison of physiological and simple pulsatile flows through stenosed arteries. J Biomech. 1999;32:959–65.
Zeng D, Ding Z, Friedman MH, Ethier CR. Effect of cardiac motion on right coronary artery hemodynamics. Ann Biomed Eng. 2003;31:420–9.
Acknowledgments
The authors would like to thank the Canadian Institutes of Health Research (CIHR) for a Vascular Research Training Award.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohammadi, H., Bahramian, F. Boundary Conditions in Simulation of Stenosed Coronary Arteries. Cardiovasc Eng 9, 83–91 (2009). https://doi.org/10.1007/s10558-009-9078-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10558-009-9078-z