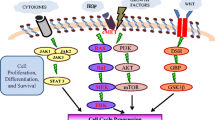
Abstract
Conventional systemic chemotherapy has been developed with so-called anti-cancer agents, essentially screened for cytotoxicity to cultured cancer cells. However, in patients with hepatocellular carcinoma (HCC), the role of chemotherapy is quite limited because most anti-cancer agents are ineffective and relatively toxic to HCC patients with chronic liver diseases. On the other hand, accumulated understanding of the molecular mechanisms regulating cancer progression has led to novel development of molecularly targeted therapies with cytostatic agents. Recently, a phase III clinical trial revealed a multi-kinase inhibitor, Sorafenib, as the first agent leading to improved overall survival of patients with advanced HCC. A new era of HCC treatment has arrived, based on identification of deranged signaling pathways of cancer cells or their microenvironment. This review summarizes the molecular hallmarks of HCC with a focus on angiogenesis, growth signals, and mitotic stress, and a novel concept “synthetic lethality” for the targeted therapy strategy.





Similar content being viewed by others
References
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100(1):57–70
Kroemer G, Pouyssegur J (2008) Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell 13(6):472–482
Luo J, Solimini NL, Elledge SJ (2009) Principles of cancer therapy: oncogene and non-oncogene addiction. Cell 136(5):823–837
Weinstein IB (2002) Cancer. addiction to oncogenes-the Achilles heal of cancer. Science 297(5578):63–64
Weinstein IB, Joe AK (2006) Mechanisms of disease: oncogene addiction—a rationale for molecular targeting in cancer therapy. Nat Clin Pract Oncol 3(8):448–457
Tanaka S, Arii S (2009) Molecularly targeted therapy for hepatocellular carcinoma. Cancer Sci 100(1):1–8
Llovet JM, Bruix J (2008) Molecular targeted therapies in hepatocellular carcinoma. Hepatology 48:1312–1327
Yau T, Chan P, Epstein R et al (2009) Management of advanced hepatocellular carcinoma in the era of targeted therapy. Liver Int 29(1):10–17 Review
Hanahan D, Folkman J (1996) Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86:353–364
Tanaka S, Arii S (2006) Current status of perspective of antiangiogenic therapy for cancer; hepatocellular carcinoma. Int J Clin Oncol 11:82–89
Ferrara N (2002) VEGF and the quest for tumour angiogenesis factors. Nat Rev Cancer 2(10):795–803
Mise M, Arii S, Higashituji H et al (1996) Clinical significance of vascular endothelial growth factor and basic fibroblast growth factor gene expression in liver tumor. Hepatology 23(3):455–464
Schmitt M, Horbach A, Kubitz R et al (2004) Disruption of hepatocellular tight junctions by vascular endothelial growth factor (VEGF): a novel mechanism for tumor invasion. J Hepatol 41(2):274–283
Kaplan RN, Riba RD, Zacharoulis S et al (2005) VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438(7069):820–827
Hiratsuka S, Watanabe A, Aburatani H et al (2006) Tumour-mediated upregulation of chemoattractants and recruitment of myeloid cells predetermines lung metastasis. Nat Cell Biol 8(12):1369–1375
Arii S (2004) Role of vascular endothelial growth factor on the invasive potential of hepatocellular carcinoma. J Hepatol 41(2):333–335
Andrae J, Gallini R, Betsholtz C (2008) Role of platelet-derived growth factors in physiology and medicine. Genes Dev 22(10):1276–1312
Kuhnert F, Tam BY, Sennino B et al (2008) Soluble receptor-mediated selective inhibition of VEGFR and PDGFRbeta signaling during physiologic and tumor angiogenesis. Proc Natl Acad Sci USA 105(29):10185–10190
Uematsu S, Higashi T, Nouso K et al (2005) Altered expression of vascular endothelial growth factor, fibroblast growth factor-2 and endostatin in patients with hepatocellular carcinoma. J Gastroenterol Hepatol 20(4):583–588
Poon RT, Ng IO, Lau C et al (2001) Correlation of serum basic fibroblast growth factor levels with clinicopathologic features and postoperative recurrence in hepatocellular carcinoma. Am J Surg 182:298–304
Llovet JM, Ricci S, Mazzaferro V et al (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359:378–390
Tanaka S, Mori M, Sakamoto Y et al (1999) Biologic significance of angiopoietin-2 expression in human hepatocellular carcinoma. J Clin Investig 103(3):341–345
Tanaka S, Wands JR, Arii S (2006) Induction of angiopoietin-2 gene expression by COX-2: A novel role for COX-2 inhibitors during hepatocarcinogenesis. J Hepatol 44(1):233–235
Tanaka S, Sugimachi K, Yamashita Yi et al (2002) Tie2 vascular endothelial receptor expression and function in hepatocellular carcinoma. Hepatology 35(4):861–867
Seegar TC, Eller B, Tzvetkova-Robev D et al (2010) Tie1–Tie2 interactions mediate functional differences between angiopoietin ligands. Mol Cell 37(5):643–655
Holash J, Maisonpierre PC, Compton D et al (1999) Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science 284(5422):1994–1998
Oliner J, Min H, Leal J et al (2004) Suppression of angiogenesis and tumor growth by selective inhibition of angiopoietin-2. Cancer Cell 6(5):507–516
Herbst RS, Hong D, Chap L et al (2009) Safety, pharmacokinetics, and antitumor activity of AMG 386, a selective angiopoietin inhibitor, in adult patients with advanced solid tumors. J Clin Oncol 27(21):3557–3565
Pawson T (2004) Specificity in signal transduction: from phosphotyrosine–SH2 domain interactions to complex cellular systems. Cell 116:191–203
Tanaka S, Sugimachi K, Maehara S et al (2002) Oncogenic signal transduction and therapeutic strategy for hepatocellular carcinoma. Surgery 131:S142–S147
Schagdarsurengin U, Wilkens L, Steinemann D et al (2003) Frequent epigenetic inactivation of the RASSF1A gene in hepatocellular carcinoma. Oncogene 22:1866–1871
Farazi PA, DePinho RA (2006) Hepatocellular carcinoma pathogenesis: from genes to environment. Nat Rev Cancer 6:674–687
Luo J, Manning BD, Cantley LC (2003) Targeting the PI3K-Akt pathway in human cancer: rationale and promise. Cancer Cell 4:257–262
Chiang GG, Abraham RT (2007) Targeting the mTOR signaling network in cancer. Trends Mol Med 13:433–442
Foster KG, Fingar DC (2010) mTOR: conducting the cellular signaling symphony. J Biol Chem
Schvartzman JM, Sotillo R, Benezra R (2010) Mitotic chromosomal instability and cancer: mouse modelling of the human disease. Nat Rev Cancer 10(2):102–115
Tanaka S, Arii S, Yasen M et al (2008) Aurora kinase B is a predictive factor for the aggressive recurrence of hepatocellular carcinoma after curative hepatectomy. Br J Surg 95:611–619
Keen N, Taylor S (2004) Aurora-kinase inhibitors as anticancer agents. Nat Rev Cancer 4:927–936
Nguyen HG, Makitalo M, Yang D et al (2009) Deregulated Aurora-B induced tetraploidy promotes tumorigenesis. FASEB J 23(8):2741–2748
Harrington EA, Bebbington D, Moore J et al (2004) VX-680, a potent and selective small-molecule inhibitor of the Aurora kinases, suppresses tumor growth in vivo. Nat Med 10(3):262–267
Aihara A, Tanaka S, Yasen M et al (2010) The selective Aurora B kinase inhibitor AZD1152 as a novel treatment for hepatocellular carcinoma. J Hepatol 52(1):63–71
Tanaka S, Mogushi K, Yasen M et al (2010) Gene-expression phenotypes for vascular invasiveness of hepatocellular carcinomas. Surgery 147(3):405–414
Luo J, Emanuele MJ, Li D et al (2009) A genome-wide RNAi screen identifies multiple synthetic lethal interactions with the Ras oncogene. Cell 137(5):835–848
Dobzhansky T (1946) Genetics of natural populations. XIII. Recombination and variability in populations of Drosophila pseudoobscura. Genetics 31:269–290
Kaelin WG Jr (2005) The concept of synthetic lethality in the context of anticancer therapy. Nat Rev Cancer 5(9):689–698
Fong PC, Boss DS, Yap TA et al (2009) Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med 361(2):123–134
Iglehart JD, Silver DP (2009) Synthetic lethality—a new direction in cancer-drug development. N Engl J Med 361(2):189–191
Bryant HE, Schultz N, Thomas HD et al (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 434(7035):913–917
Farmer H, McCabe N, Lord CJ et al (2005) Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434(7035):917–921
Ince N, Wands JR (1999) The increasing incidence of hepatocellular carcinoma. N Engl J Med 340:798–799
Arii S, Yamaoka Y, Futagawa S et al (2000) Results of surgical and nonsurgical treatment for small-sized hepatocellular carcinomas: a retrospective and nationwide survey in Japan. The Liver Cancer Study Group of Japan. Hepatology 32:1224–1229
Tanaka S, Arii S (2010) Medical treatments: in association or alone, their role and their future perspectives: novel molecular-targeted therapy for hepatocellular carcinoma. J Hepatobiliary Pancreat Surg
Conflict of interest statement
No author has any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Tanaka, S., Arii, S. Current status of molecularly targeted therapy for hepatocellular carcinoma: basic science. Int J Clin Oncol 15, 235–241 (2010). https://doi.org/10.1007/s10147-010-0083-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-010-0083-4