Abstract
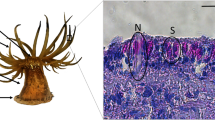
Marine bioadhesives perform in ways that manmade products simply cannot match, especially in wet environments. Despite their technological potential, bioadhesive molecular mechanisms are still largely understudied, and sea urchin adhesion is no exception. These animals inhabit wave-swept shores, relying on specialized adhesive organs, tube feet, composed by an adhesive disc and a motile stem. The disc encloses a duo-gland adhesive system, producing adhesive and deadhesive secretions for strong reversible substratum attachment. The disclosure of sea urchin Paracentrotus lividus tube foot disc proteome led to the identification of a secreted adhesion protein, Nectin, never before reported in adult adhesive organs but, that given its adhesive function in eggs/embryos, was pointed out as a putative substratum adhesive protein in adults. To further understand Nectin involvement in sea urchin adhesion, Nectin cDNA was amplified for the first time from P. lividus adhesive organs, showing that not only the known Nectin mRNA, called Nectin-1 (GenBank AJ578435), is expressed in the adults tube feet but also a new mRNA sequence, called Nectin-2 (GenBank KT351732), differing in 15 missense nucleotide substitutions. Nectin genomic DNA was also obtained for the first time, indicating that both Nectin-1 and Nectin-2 derive from a single gene. In addition, expression analysis showed that both Nectins are overexpressed in tube feet discs, its expression being significantly higher in tube feet discs from sea urchins just after collection from the field relative to sea urchin from aquarium. These data further advocate for Nectin involvement in sea urchin reversible adhesion, suggesting that its expression might be regulated according to the hydrodynamic conditions.








Similar content being viewed by others
References
Ameye L, Hernann R, Dubois P, Flammang P (2000) Ultrastructure of the echinoderm cuticle after fast-freezing /freeze substitution and conventional chemical fixations. Microsc Res Tech 48:385–393
Arendt D, Tessmar-Raible K, Snyman H, Dorresteijn AW, Wittbrodt J (2004) Ciliary photoreceptors with a vertebrate-type opsin in an invertebrate brain. Science 306:869–871
Costa C, Cavalcante C, Zito F, Yokota Y, Matranga V (2010) Phylogenetic analysis and homology modelling of Paracentrotus lividus nectin. Mol Divers 14:653–665
Del Campo A, Schwotzer W, Gorb SN, Aldred N, Santos R, Flammang P (2013) Biological and biomimetic adhesives: challenges and opportunities—preface. In: Santos R, Aldred N, Gorb S, Flammang P (eds) Biological and biomimetic adhesives: challenges and opportunities. Springer, Berlin, pp 7–17
Flammang P, Santos R, Haesaerts D (2005) Echinoderm adhesive secretions: from experimental characterization to biotechnological applications. In: Matranga V (ed) Progress in molecular and subcellular biology, marine molecular biotechnology. Echinodermata. Springer-Verlag, Berlin, pp 199–218
Flammang P, Lambert A, Bailly P, Hennebert E (2009) Polyphosphoprotein containing marine adhesives. J Adhes 85:447–464
Hennebert E, Wattiez R, Flammang P (2011) Characterisation of the carbohydrate fraction of the temporary adhesive secreted by the tube feet of the sea star Asterias rubens. Mar Biotechnol 13:484–495
Hennebert E, Wattiez R, Demeuldre M, Ladurner P, Hwang DS, Waitee JH, Flammang P (2014) Sea star tenacity mediated by a protein that fragments, then aggregates. PNAS 111(17):6317–6322
Hennebert E, Leroy B, Wattiez R, Ladurner P (2015) An integrated transcriptomic and proteomic analysis of sea star epidermal secretions identifies proteins involved in defense and adhesion. J Proteomics 128:83–91
Kamino K, Nakano M, Kanai S (2012) Significance of the conformation of building blocks in curing of barnacle underwater adhesive. FEBS J 279:1750–1760
Matranga V, Di Ferro D, Zito F, Cervello M, Nakano E (1992) A new extracellular matrix protein of the sea urchin embryo with properties of a substrate adhesion molecule. Roux’s Arch Dev Biol 201:173–178
Mistry N, Harrington W, Lasda E, Wagner EJ, Garcia-Blanco MA (2003) Of urchins and men: evolution of an alternative splicing unit in fibroblast growth factor receptor genes. RNA 9:209–217
Ohkawa K, Nishida A, Yamamoto H, Waite JH (2004) A glycosylated byssal precursor protein from the green mussel Perna viridis with modified Dopa side-chains. Biofouling 20:101–115
Roth Z, Yehezkel G, Khalaila I (2012) Identification and quantification of protein glycosylation. Int J Carbohydr Chem 2012, 640923
Santos R, Flammang P (2006) Morphology and tenacity of the tube foot disc of three common European sea urchin species: a comparative study. Biofouling 22:187–200
Santos R, Flammang P (2007) Intra- and interspecific variation of attachment strength in sea urchins. Mar Ecol Prog Ser 332:129–142
Santos R, Flammang P (2008) Estimation of the attachment strength of the shingle sea urchin, Colobocentrotus atratus, and comparison with three sympatric echinoids. Mar Biol 154:37–49
Santos R, Gorb S, Jamar V, Flammang P (2005) Adhesion of echinoderm tube feet to rough surfaces. J Exp Biol 208:2555–2567
Santos R, da Costa G, Franco C, Gomes-Alves P, Flammang P, Coelho AV (2009) First insights into the biochemistry of tube foot adhesive from the sea urchin Paracentrotus lividus (Echinoidea, Echinodermata). Mar Biotechnol 11:686–698
Santos R, Barreto A, Franco C, Coelho AV (2013) Mapping sea urchins tube feet proteome—a unique hydraulic mechano-sensory adhesive organ. J Proteomics 79:100–113
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108
Smith AM (2006) The biochemistry and mechanics of gastropod adhesive gels. In: Smith AM, Callow JA (eds) Biological adhesives. Springer, Berlin
Stanley MS, Callow ME, Callow JA (1999) Monoclonal antibodies to adhesive cell coat glycoproteins secreted by zoospores of the green alga Enteromorpha. Planta 210:61–71
Stewart RJ, Wang CS, Shao H (2011) Complex coacervates as a foundation for synthetic underwater adhesives. Adv Colloid Interface Sci 167:85–93
Taylor CM, Wang W (2007) Histidinoalanine: a crosslinking amino acid. Tetrahedron 63:9033–9047
Waite JH, Qin X (2001) Polyphosphoprotein from the adhesive pads of Mytilus edulis. Biochemistry 40:2887–2893
Warner SC, Waite JH (1999) Expression of multiple forms of an adhesive plaque protein in an individual mussel, Mytilus edulis. Mar Biol 134:729–734
Xiang M, Bédard P-A, Wessel G, Filion M, Brandhorst BP, Klein WH (1988) Tandem duplication and divergence of a sea urchin protein belonging to the Troponin C superfamily. J Biol Chem 263:17173–17180
Yu J, Wei W, Danner E, Ashley RK, Israelachvili JN, Waite JH (2011) Mussel protein adhesion depends on thiol-mediated redox modulation. Nat Chem Biol 7:588–590
Zhao H, Sun C, Stewart RJ, Waite JH (2005) Cement proteins of the tube-building polychaete Phragmatopoma californica. J Biol Chem 280:42938–42944
Zhao H, Robertson NB, Jewhurst SA, Waite JH (2006) Probing the adhesive footprints of Mytilus californianus byssus. J Biol Chem 281:11090–11096
Zhao H, Sagert J, Hwang DS, Waite JH (2009) Glycosylated hydroxytryptophan in a mussel adhesive protein from Perna viridis. J Biol Chem 284:23344–23352
Zito F, Burke RD, Matranga V (2010) Pl-nectin, a discoidin family member, is a ligand for betaC integrins in the sea urchin embryo. Matrix Biol 29:341–345
Acknowledgments
This work was supported by Fundação para a Ciência e Tecnologia through a postdoctoral grant attributed to Duarte Toubarro (SFRH/BPD/ 77483/2011), a project research grant attributed to Analuce Gouveia, a postdoctoral grant and a research contract attributed to Gonçalo da Costa (SFRH/BPD/73779/2010, IF/00359/2014), a research contract by the Ciência 2008 program, and a postdoctoral grant attributed to Romana Santos (SFRH/BPD/109081/2015), and project grants attributed (PTDC/MAR/117360/2010), PEst-OE/QUI/UI0612/2013, UID/MULTI/00612/2013). The authors wish to acknowledge Dr. Fátima Gil and Miguel Cadete for sea urchin maintenance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOCX 17 kb)
Supplementary Table 2
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Toubarro, D., Gouveia, A., Ribeiro, R.M. et al. Cloning, Characterization, and Expression Levels of the Nectin Gene from the Tube Feet of the Sea Urchin Paracentrotus Lividus . Mar Biotechnol 18, 372–383 (2016). https://doi.org/10.1007/s10126-016-9698-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-016-9698-4