Abstract
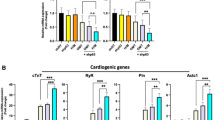
The weak contractile force exerted by engineered cardiac muscle is a big problem in cardiac muscle tissue engineering, even though the field has made great progress over the past decade. We believe that one major reason for the weak contractile force is that the expression of genes regulating cardiomyocyte differentiation and cardiac tissue syncytium may be different for in vivo and cultured cells. In the present study, we investigated the difference of mRNA expression under in vivo and culture conditions in order to seek a target for further gene transfer treatment in the process of cardiac tissue construction. To this end, mRNA expression of four major transcriptional factors (SRF, p300, Nkx2.5, and myocardin) and two intercalated disk con stituent proteins (N-cadherin and connexin43) in rat cardiomyocytes was measured by means of ratiometric reverse-transcription polymerase chain reaction. Cardiomyocytes were harvested from the hearts of 18-day (about 3 days before birth) Wistar-rat embryos (embryonic cells), 12-day neonatal rat hearts (neonatal cells), or 14-day successive dish culture of the embryonic cells harvested from 18-day embryos (cultured cells). The results indicated that, except for SRF, the mRNAs had a lower expression tendency in cultured cells than in embryonic and in neonatal cells; in particular, the mRNA expression of myocardin, Ncadherin, and connexin43 of cultured cardiomyocytes was significantly lower than that of neonatal cells. Therefore, myocardin is a candidate for forced gene up-expression during the construction of engineered cardiac tissue; in addition, a plausible reason for the weak contractile force of engineered cardiac tissue is the weak constitution of intercalated disk, because it was elucidated that mRNA expression of proteins related to intercalated disk were lower in culture.
Similar content being viewed by others
References
Carrier RL, Papadaki M, Rupnick M, Schoen FJ, Bursac N, Langer R, Freed LE, Vunjak-Novakovic G. Cardiac tissue engineering: cell seeding, cultivation parameters, and tissue construct characterization. Biotechnol Bioeng 1999;64:580–589
Leor J, Aboulafia-Etzion S, Dar A, Shapiro L, Barbash IM, Battler A, Granot Y, Cohen S. Bioengineered cardiac grafts: A new approach to repair the infarcted myocardium? Circulation 2000;102: III56–61
Zimmermann WH, Fink C, Kralisch D, Remmers U, Weil J, Eschenhagen T. Three-dimensional engineered heart tissue from neonatal rat cardiac myocytes. Biotechnol Bioeng 2000;68: 106–114
van Luyn MJ, Tio RA, Gallego y van Seijen XJ, Plantinga JA, de Leij LF, DeJongste MJ, van Wachem PB. Cardiac tissue engineering: characteristics of in-unison contracting two-and three-dimensional neonatal rat ventricle cell (co)-cultures. Biomaterials 2002;23:4793–4801
Shimizu T, Yamato M, Isoi Y, Akutsu T, Setomaru T, Abe K, Kikuchi A, Umezu M, Okano T. Fabrication of pulsatile cardiac tissue grafts using a novel 3-dimensional cell sheet manipulation technique and temperature-responsive cell culture surfaces. Circ Res 2002;90:e40
Eschenhagen T, Zimmermann WH. Engineering myocardial tissue. Circ Res 2005;97:1220–1231
Parlakian A, Tuil D, Hamard G, Tavernier G, Hentzen D, Concordet JP, Paulin D, Li Z, Daegelen D. Targeted inactivation of serum response factor in the developing heart results in myocardial defects and embryonic lethality. Mol Cell Biol 2004;24: 5281–5289
Psichari E, Balmain A, Plows D, Zoumpourlis V, Pintzas A. High activity of serum response factor in the mesenchymal transition of epithelial tumor cells is regulated by RhoA signaling. J Bio Chem 2002;277:29490–29495
Xing W, Zhang T, Cao D, Wang Z, Antos CL, Li S, Wang Y, Olson EN, Wang D. Myocardin induces cardiomyocyte hypertrophy. Circ Res 2006;98:1089–1097
Wang D, Chang PS, Wang Z, Sutherland L, Richardson JA, Small E, Krieg PA, Olson EN. Activation of cardiac gene expression by myocardin, a transcriptional cofactor for serum response factor. Cell 2001;105:851–862
Bruneau BG. Transcriptional regulation of vertebrate cardiac morphogenesis. Circ Res 2002;90:509–519
Backs J, Olson EN. Control of cardiac growth by histone acetylation/deacetylation. Circ Res 2006;98:15–24
Zuppinger C, Eberhardt ME, Eppenberger HM. N-Cadherin: structure, function and importance in the formation of new intercalated disc-like cell contacts in cardiomyocytes. Heart Fail Rev 2000;5:251–257
Salameh A, Schneider P, Muhlberg K, Hagendorff A, Dhein S, Pfeiffer D. Chronic regulation of the expression of gap junction proteins connexin40, connexin43, and connexin45 in neonatal rat cardiomyocytes. Euro J Pharmacol 2004;503:9–16
Feng Z, Matsumoto T, Nakamura T. Measurements of the mechanical properties of contracted collagen gels populated with rat fibroblasts or cardiomyocytes. J Artif Organs 2003;6(3):192–196
Sambrook J, Russell DW. Condensed protocols from molecular cloning: a laboratory manual. New York: Cold Spring Harbor, 2006
Srivastava D, Olson EN. A genetic blueprint for cardiac development. Nature 2000;407:221–226
Doevendans PA, Bilsen MV. Transcription factors and the cardiac gene programme. Int J Biochem Cell Biol 1996;28(4):387–403
Tapscott SJ. The circuitry of a master switch: Myod and the regulation of skeletal muscle gene transcription. Development 2005;132: 2685–2695
Perriard JC, Hirschy A, Ehler E. Dilated cardiomyopathy: a disease of the intercalated disc? Trends Cardiovasc Med 2003;13: 30–38
Smolich JJ. Ultrastructural and functional features of the developing mammalian heart: a brief overview. Reprod Fertil Dev 1995; 7(3):451–461
Lam ML, Bartoli M, Claycomb WC. The 21-day postnatal rat ventricular cardiac muscle cell in culture as an experimental model to study adult cardiomyocyte gene expression. Mol Cell Biochem 2002;229(1–2):51–62
Greenbaum D, Luscombe NM, Jansen R, Qian J, Gerstein M. Interrelating different types of genomic data, from proteome to secretome: oming in on function. Genome Res 2001;11: 1463–1468
Gygi SP, Rochon Y, Franza BR, Aebersold R. Correlation between protein and mRNA abundance in yeast. Mol Cell Biol 1999; 19(3):1720–1730
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakamura, T., Feng, Z., Honda, T. et al. Comparison of mRNA expression of transcriptional factors and intercalated disk constituent proteins between in vivo and cultured cardiomyocytes. J Artif Organs 11, 134–140 (2008). https://doi.org/10.1007/s10047-008-0414-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-008-0414-7