Abstract
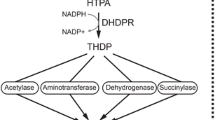
Dihydrodipicolinate synthase (DHDPS, E.C.4.2.1.52) catalyzes the first committed step in the lysine biosynthetic pathway: the condensation of (S)-aspartate semialdehyde and pyruvate to form (4S)-4-hydroxy-2,3,4,5-tetrahydro-(2S)-dipicolinic acid. Since (S)-lysine biosynthesis does not occur in animals, DHDPS is an attractive target for rational antibiotic and herbicide design. Here, we report the crystal structure of DHDPS from a hyperthermophilic bacterium Aquifex aeolicus (AqDHDPS). l-Lysine is used as an important animal feed additive where the production is at the level of 1.5 million tons per year. The biotechnological manufacture of lysine has been going for more than 50 years which includes over synthesis and reverse engineering of DHDPS. AqDHDPS revealed a unique disulfide linkage which is not conserved in the homologues of AqDHDPS. In silico mutation of C139A and intermolecular ion-pair residues and the subsequent molecular dynamics simulation of the mutants showed that these residues are critical for the stability of AqDHDPS tetramer. MD simulations of AqDHDPS at three different temperatures (303, 363 and 393 K) revealed that the molecule is stable at 363 K. Thus, this structural and in silico study of AqDHDPS likely provides additional details towards the rational and structure-based design of hyper-l-lysine producing bacterial strains.






Similar content being viewed by others
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl Acids Res 25:3389–3402
Atkinson SC, Dogovski C, Renwick CJ, Dobson RCJ, Perugini MA (2012a) Cloning, expression, purification and crystallization of dihydrodipicolinate synthase from Agrobacterium tumefaciens. Acta Crystallogr F68:1040–1047
Atkinson SC, Dogovski C, Downton MT, Pearce FG, Reboul CF, Buckle AM, Gerrard JA, Dobson RCJ, Wagner J, Perugini MA (2012b) Crystal, solution and in silico structural studies of dihydrodipicolinate synthase from the common grapevine. PLoS ONE 7:6
Azevedo RA (2001) Lysine metabolism in higher plants. Amino Acids 20:261–279
Bailey S (1994) The CCP4 suite: programs for protein crystallography. MolRep Acta Crystallogr D50:760–763
Balamurugan B, Md. Roshan MNA, Hameed BS, Sumathi K, Senthilkumar R, Udayakumar A, Babu KHV, Kalaivani M, Sowmiya G, Sivasankari P,Saravanan S, Vasuki Ranjani C, Gopalakrishnan K, Selvakumar KN, Jaikumar M, Brindha T, Michael D, Sekar K (2007) PSAP: protein structure analysis package. J Appl Crystalloger 40:773–777
Betz SF (1993) Disulfide bonds and the stability of globular proteins. Protein Sci 2:1551–1558
Blickling S, Beisel HG, Bozix D, Knablein J, Laber B, Huber R (1997) Structure of dihydrodipicolinate synthase of Nicotiana sylvestris reveals novel quaternary structure. J Mol Biol 274:608–621
Brünger AT, Adams PD, Clore GM, DeLano WL, Gros P, Grosse-Kunstleve RW, Jiang JS, Kuszewski J, Nilges M, Pannu NS, Read RJ, Rice LM, Simonson T, Warren GL (1998) Crystallography & NMR system (CNS), a new software suite for macromolecular structure determination. Acta Crystallogr D54:905–921
Burgess BR, Dobson RCJ, Bailey MF, Atkinson SC, Griffin MDW, Jameson GB, Parker MW, Gerrard JA, Perugini MA (2008) Structure and evolution of a novel dimeric enzyme from a clinically important bacterial pathogen. J Biol Chem 283:27598–27603
Deckert G, Warren PV, Gaasterland T, Young WG, Lenox AL, Graham DE, Overbeek R, Snead MA, Keller M, Aujay M, Hauberk R, Feldman RA, Short JM, Olsen GJ, Swanson RV (1998) The complete genome of the hyperthermophilic bacterium Aquifex aeolicus. Nature 392:353–357
Dereppe C, Bold G, Ghisalba O, Ebert E, Schar H (1992) Purification and characterization of dihydrodipicolinate synthase from pea. Plant Physiol 98:813–821
Dobson RCJ, Valega K, Gerrard JA (2004) The crystal structure of three site-directed mutants of Escherichia coli dihydrodipicolinate synthase: further evidence for a catalytic triad. J Mol Biol 338:329–339
Dobson RCJ, Devenish SR, Tumer LA, Clifford VR, Pearch FG, Jameson GB, Gerrard JA (2005a) Role of arginine 138 in the catalysis and regulation of Escherichia coli dyhydrodipicolinate synthase. Biochemistry 44:13013–133007
Dobson RCJ, Griffin MDW, Jameson GB, Gerrard JA (2005b) The crystal structures of native and (S)-lysine-bound dihydrodipicolinate synthase from Escherichia coli with improved resolution show new features of biological significance. Acta Crystallogr D61:1116–1124
Emsley P, Cowtan K (2004) Coot: model-building tools for molecular graphics. Acta Crystallogr D60:2126–2132
Geng Feng (2013) Exploring the allosteric mechanism of dihydrodipicolinate synthase by reverse engineering of the allosteric inhibitor binding sites and its application for lysine production. Appl Microbiol Biotechnol 97:1963–1971
Girish TS, Sharma E, Gopal B (2008) Structural and functional characterization of Staphylococcus aureus dihydrodipicolinate synthase. FEBS Lett 582:2923–2930
Griffin MDW, Dobson RCJ, Pearce FG, Antonio L, Whitten AE, Liew CK, Mackay JP, Trewhella J, Jameson GB, Perugini MA, Gerrard JA (2008) Evolution of quaternary structure in a homotetrameric enzyme. J Mol Biol 380:691–703
Griffin MDW, Billakanti JM, Wason A, Keller S, Mertens HDT, Atkinson SC, Dobson RCJ, Perugini MA, Gerrard JA, Pearce FG (2012) Characterisation of the first enzymes commited to lysine biosynthesis in Arabidopsis thaliana. PLoS ONE 7(7):e40318
Hess B, Kutzner C, Spoel DV, Lindahl E (2008) GROMACS 4: algorithms for highly efficient, load-balanced, and scalable molecular simulation. J Chem Theory Comput 4:435–447
Holm L, Kääriäinen S, Rosenström P, Schenkel A (2008) Searching protein structure databases with DaliLite v. 3. Struct Bioinfo 24:2780–2781
Hutton Craig A, Perugini MA, Gerrard JA (2007) Inhibition of lysine biosynthesis: an evolving antibiotic strategy. Mol BioSyst 3:458–465
Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci 8:275–282
Karsten WE (1997) Dihydrodipicolinate synthase from Escherichia coli: pH dependent changes in the kinetic mechanism and kinetic mechanism of allosteric inhibition by l-lysine. Biochemistry 36:1730–1739
Kaur N, Gautam A, Kumar S, Singh A, Singh N, Sharma S, Sharma R, Tewari R, Singh TP (2011) Biochemical studies and crystal structure determination of dihydrodipicolinate synthase from Pseudomonas aeruginosa. Int J Biol Macromol 48:779–787
Kefala G, Evans GL, Griffin MDW, Devenish SRA, Pearce FG, Perugini MA, Gerrard JA, Weiss MS, Dobson RCJ (2008) Crystal structure and kinetic study of dihydrodipicolinate synthase from Mycobacterium tuberculosis. Biochem J 411:351–360
Kumar S, Tsai CJ, Nussinov R (2000) Factors enhancing protein thermostability. Protein Eng 13:179–191
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) PROCHECK: a program to check the stereochemical quality of protein structures. J Appl Crystal 26:283–291
Manjunath K, Kanaujia SP, Kanagaraj S, Jeyakanthan J, Sekara K (2013) Structure of SAICAR synthetase from Pyrococcus horikoshii OT3: insights into thermal stability. Int J Biol Macromol 53:7–19
McPherson A (1990) Current approaches to macromolecular crystallization. Eur J Biochem 189:1–23
Mirwaldt C, Kirndörfer I, Huber R (1995) The crystal structure of dihydrodipicolinate synthase from Escherichia coli at 2.5 Å resolutions. J Mol Biol 246:227–239
Muscroft-Taylor AC, Catchpole RJ, Dobson RCJ, Pearce FG, Perugini MA, Gerrard JA (2010) Disruption of quaternary structure in Escherichia coli dihydrodipicolinate synthase (DHDPS) generates a functional monomer that is no longer inhibited by lysine. Arch Biochem Biophys 503:202–206
Otwinowski Z, Minor W (1997) Processing of X-ray diffraction data collected in oscillation mode. Method Enzymol 276:307–326
Padmanabhan B, Strange RW, Antonyuk SV, Ellis MJ, Hasnain SS, Iino H, Agari Y, Besshoa Y, Yokoyamaa S (2009) Structure of dihydrodipicolinate synthase from Methanocaldococcus jannaschii. Acta Crystallogr F65:1222–1226
Pearce FG, Perugini MA, McKerchar HJ, Gerrard JA (2006) Dihydrodipicolinate synthase from Thermotoga maritima. Biochemistry 400:359–366
Pearce FG, Dobson RCJ, Jameson GB, Perugini MA, Gerrard JA (2011) Characterizatiom of monomeric dihydrodipicolinate synthase variant reveals the importance of substrate binding in optimizing oligomerization. BBA—Proteins Proteom 1814:1900–1909
Reboul CF, Porebski BT, Griffin MDW, Dobson RCJ, Perugini MA, Gerrard JA, Buckle AM (2012) Structural and dynamic requirements for optimal activity of the essential bacterial enzyme dihydrodipicolinate synthase. Plos Comput Biol 8:6
Sherman W, Day T, Jacobson MP, Friesner RA, Farid R (2006) Novel procedure for modeling ligand/receptor induced fit effects. J Med Chem 49:534–553
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Turner P (2007) Potential and utilization of thermophiles and thermostable enzymes in biorefining. Microb Cell Fact 6:9
Vogt G (1997) Protein thermal stability, hydrogen bonds, and ion pairs. J Mol Biol 269:631–643
Voss JE, Scally SW, Taylor NL, Atkinson SC, Griffin MDW, Hutton CA, Parker MW, Alderton MR, Gerrard JA, Dobson RCJ, Dogovski C, Perugini MA (2010) Substrate-mediated stabilization of a tetrameric drug targer reveals achilles heel in anthrax. J Biol Chem 285(8):5188–5195
Ricea EA, Bannona GA, Glenna KC, Jeonga SS, Sturmanb EJ, Rydel TJ (2008) Characterization and crystal structure of lysine insensitive Corynebacterium glutamicum dihydrodipicolinate synthase (cDHDPS) protein. Arch Biochem Biophys 480:111–121
Acknowledgments
This work was supported by the RIKEN Structural Genomic/Proteomics Initiative (RSGI), the National Project on Protein Structural and Functional Analyses, Ministry of Education, Culture, Sports, Science and Technology of Japan. US thanks the Department of Science and Technology (DST), Government of India for the financial support in the form of INSPIRE fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Atomi.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sridharan, U., Ebihara, A., Kuramitsu, S. et al. Crystal structure and in silico studies of dihydrodipicolinate synthase (DHDPS) from Aquifex aeolicus . Extremophiles 18, 973–985 (2014). https://doi.org/10.1007/s00792-014-0667-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-014-0667-4