Abstract
Goals of work
Prospective clinical study to evaluate patients suffering from solid tumor using a totally implanted venous access device (TIVAD) to determine: (1) if there is a relationship between cutaneous contamination at port insertion site and catheter-related bloodstream infection (CRBI); (2) development modalities of CRBI; (3) if there is a relationship between chemotherapy administration modalities by push/bolus versus continuous infusion and CRBI.
Patients and methods
We studied 41 consecutive patients who needed a TIVAD positioned for chemotherapy administration by bolus/push or continuous infusion. In every patient, we performed blood cultures from blood samples from port catheters and cutaneous cultures from cutaneous tampons of the skin surrounding the implant area on the first (T0) and eight day (T1) postoperatively, after 1 month (T2), and after 3 months (T3) from insertion.
Main results
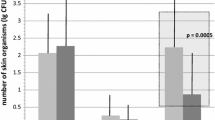
The study was completed on 40 patients; in one case, the port was removed at T2 for septic complications. We obtained four positive blood cultures (two, 5%), two in the same patient, all caused by staphylococcus. Positive cutaneous tampons were 21 (13%) in 11 patients (27%); the four CRBI occurred in this group of patients with none in the remaining 30 patients (73%) for a total number of 120 tampons (p<0.01). In two cases, the same germ was isolated from both the skin and blood. None of the patients presented a local infection of the subcutaneous pocket. Positive cutaneous cultures decrease over time: T0–T2, 24–5%; T1–T3, 20–5% (p<0.04). There were no differences in CRBI incidence and positive cutaneous tampons between the two chemotherapy administration modalities.
Conclusions
Cutaneous microbial flora has a primary role in CRBI development within TIVADs; there is a relationship between cutaneous colonization and CRBI; colonization reaches its maximum during the first days after catheterization in which the use of the system is at high risk; colonization occurs both via extraluminal and endoluminal routes; there is no difference in CRBI incidence between bolus and continuous infusion administration.

Similar content being viewed by others
References
Welling RE, Hall JM, Meyer RL Arbaugh JJ(1986) Implantable venous access device: an alternative method of extended cancer care. J Surg Oncol 33:73–75
Grannan KJ, Taylor PH (1990) Early and late complication of totally implanted venous access device.J Surg Oncol 44:52–54
Laurenzi L, Fimiani C, Faglieri N, Natoli S, Milasi G, Tirelli W, Arcuri E (1996) Complications with fully implantable venous access systems in oncologic patients. Tumori 82:232–236
Nouwen JL, van Belkum A, de Marie S, Sluijs J, Wielenga JJ, Kluytmans JA, Verbrugh HA (1998) Clonal expansion of Staphylococcus epidermidis strains causing Hickman catheter-related infections in a hemoto-oncologic departement. J Clin Microbiol 36:2696–2702
Maki DG (1992) Infections due to infusion therapy. In: Bennet JV, Brachman PS (eds) Hospital infections. Little, Brown, Boston, pp 849–898
Mermel LA, McCormik RD, Sprinman SR, Maki DG (1991) The pathogenesis and epidemiology of catheter-related infections with pulmonary artery Swan-Ganz catheters: a prospective study utilizing molecular subtyping. Am J Med 91 [Ssuppl]:S197–S205
Cooper GL, Schiller AL,Hopkins CC (1988) Possible role of capillary action in the pathogenesis of experimental catheter-associated dermal tunnel infections. J Clin Microbiol 26:8–12
Sitges-Serra A,Linares J,Garau J (1985) Catheter sepsis: the clue is the hub. Surgery 97:355–357
Raad I, Costerton W, Sabharwal U, Sacilowski M, Anaissie E, Bodey GP (1993) Ultrastuctural analysis of indwelling vascular catheter: a quantitative relationship between luminal colonization and duration of placement. J Infect Dis 168:40–47
Maki DG, Jarrett F, Sarafin HW(1977) A semiquantitative culture method for identification of catheter-related infection in the burn patient. J Surg Res 22:513–520
Maki DG, Hassemer CA (1981) Endemic rate of fluid contamination and related septicemia in arterial pressure monitoring. Am J Med 70:733–738
Sitges-Serra A (1999) Strategies for prevention of catheter-related bloodstream infections. Supp Care Cancer 7:391–395
Greene FL, Moore W, Strickland G, McFarland J (1988) Comparison of a totally implantable access device for chemotherapy (Port-A Cath) and long-term percutaneous catheterisation (Broviac).South Med J 81:581–583
Groeger JS, Lucas AB, Thaler HT, Friedlander-Klar H, Brown AE, Kiehn TE, Armstrong D (1993) Infectious morbidity associated with log-term use of venous access devices in patients with cancer. Ann Intern Med 119:1168–1174
de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G, Papamichael D, Le Bail N, Louvet C, Hendler D, de Braud F, Wilson C, Morvan F, Bonetti A (2000) Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer.J Clin Oncol 18:2938–2947
Garufi C, Levi F, Giunta S, Aschelter A, Pace R, Nistico C, Terzoli E (1995) Chronomodulated 5-day infusion of floxuridine and L-folinic acid in patients with advanced malignancies: a feasibility and tolerability study. J Infus Chemother. 5(3) [Suppl 1]:134–137
Arcangeli G, Saracino B, Danesi DT, De Campora E, Giovinazzo G, Cognetti F, Carlini P, Arcangeli S, Mecozzi A (2002) Accelerated hyperfractionated radiotherapy and concurrent protracted venous infusion chemotherapy in locally advanced head and neck cancer. Am J Clin Oncol 25:431–437
Dorn GL, Smith K (1978) New centrifugation blood culture device. J Clin Microbiol 7(1):52–54
Elliott TS, Moss HA, Tebbs SE, Wilson IC, Bonser RS, Graham TR, Burke LP, Faroqui MH (1997) Novel approach to investigate a source of microbial contamination of central venous catheters. Eur J Clin Microbiol Infect Dis 16(3):210–213
Snydman DR, Gorbea HF, Pober BR, Majka JA, Murray SA, Perry LK (1982) Predictive value of surveillance skin cultures in total-parenteral-nutrition-related infection. Lancet 2:1385–1388
Raad II, Baba M, Bodey GP (1995) Diagnosis of catheter-related infections: the role of surveillance and target quantitative skin cultures. Clin Infect Dis 20:593–597
Bozzetti F (1985) Central venous catheter sepsis. Surg Gynecol Obstet 161:593–603
Andremont A, Paulet R, Nitember G, Hill C (1988) Value of semiquantitative cultures of blood drawn through catheter hubs for estimating the risk of catheter tip colonization in cancer patients. J Clin Microbiol 26:2297–2299
Brown DF, Muirhead MJ, Travis PM, Vire SR, Weller J, Hauer-Jensen M (1997) Mode of chemotherapy does not affect complications with the implantable venous access device. Cancer 80:966–972
Martens MG, Kolrud BL, Faro S, Maccato M, Hammill H (1995) Development of wound infection or separation after cesarean delivery. Prospective evaluation of 2,431 cases. J Reprod Med 40:171–175
Palmer DL, Pett SB, Akl BF (1995) Bacterial wound colonization after broad-spectrum versus narrow-spectrum antibiotics. Ann Thorac Surg 59:626–631
Gristina AG, Price JL, Hobgood CD, Webb LX, Costerton JW (1985) Bacterial colonization of percutaneous sutures. Surgery 98:12–19
Acknowledgements
Partially supported by the Italian National Institute of Health, research project N° 0201R2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laurenzi, L., Natoli, S., Benedetti, C. et al. Cutaneous bacterial colonization, modalities of chemotherapeutic infusion, and catheter-related bloodstream infection in totally implanted venous access devices. Support Care Cancer 12, 805–809 (2004). https://doi.org/10.1007/s00520-004-0607-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-004-0607-4