Abstract
Key message
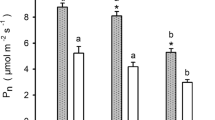
Photosynthetic capacity of yew needles depends primarily on light environment, with little evidence that developmental stage is a determinant photosynthetic performance. However, needle morphology is mostly determined by plant size.
Abstract
Populations of yew Taxus baccata are often characterized by a lack of natural regeneration and stands of this species have been afforded priority conservation status within the European Union. Light availability is known to be a key factor influencing recruitment dynamics in yew. To explore potential size-related responses to light, we investigated the distribution, photosynthetic efficiency and needle morphological traits in yew across a gradient of light availability at Atlantic oak woodland in southwest Ireland. Young plants were more common in shaded areas whereas saplings and juvenile yews were distributed in areas of higher light availability. Photosynthetic capacity of yew needles was found to depend primarily on light environment, with little evidence that the developmental stage is a determinant of photosynthetic performance. Photoinhibition was evident in needles sampled from high light environments across all demographic size classes. However, larger plants also showed particularly strong needle morphological responses (needle length, width, thickness, and specific leaf area) to light levels, which were mostly lacking in seedlings. We conclude that there is a dual control of yew light responses; the efficiency of the photosynthetic light reactions is largely linked to the light environment in which the plants grew, while needle morphology is mostly determined by the size of the plants. The findings of this study highlight the importance of considering all life-history stages and multiple traits when evaluating species response to light availability.



Similar content being viewed by others
References
Augspurger CK, Bartlett EA (2003) Differences in leaf phenology between juvenile and adult trees in a temperate deciduous forest. Tree Physiol 23(8):517
Ball M, Hodges V, Laughlin G (1991) Cold-induced photoinhibition limits regeneration of snow gum at tree-line. Funct Ecol 5:663–668
Canham CD, Denslow JS, Platt WJ, Runkle JR, Spies TA, White PS (1990) Light regimes beneath closed canopies and tree-fall gaps in temperate and tropical forests. Can J For Res 20(5):620–631
Coopman RE, Reyes-Díaz M, Briceño VF, Corcuera LJ, Cabrera HM, Bravo LA (2008) Changes during early development in photosynthetic light acclimation capacity explain the shade to sun transition in Nothofagus nitida. Tree Physiol 28(10):1561–1571
Devaney JL (2013) The natural regeneration ecology of yew Taxus baccata L., University College Cork
Devaney JL, Jansen MA, Whelan PM (2013) Spatial patterns of natural regeneration in stands of English yew (Taxus baccata L.); Negative neighbourhood effects. For Ecolog Manag 321:52–60
Dhar A, Ruprecht H, Klumpp R, Vacik H (2006) Stand structure and natural regeneration of English yew (Taxus baccata L.) at Stiwollgraben in Austria. Dendrobiology 56:19–26
Dhar A, Ruprecht H, Klumpp R, Vacik H (2007) Comparison of ecological condition and conservation status of English yew population in two Austrian gene conservation forests. J For Res 18(3):181–186
Dubreuil M, Sebastiani F, Mayol M, González-Martínez SC, Riba M, Vendramin GG (2008) Isolation and characterization of polymorphic nuclear microsatellite loci in Taxus baccata L. Conserv Genet 9(6):1665–1668
Farris E, Filigheddu R (2008) Effects of browsing in relation to vegetation cover on common yew (Taxus baccata L.) recruitment in mediterranean environments. Plant Ecol 199(2):309–318
Garcia D, Obeso RJ (2003) Facilitation by herbivore-mediated nurse plants in a threatened tree, Taxus baccata: local effects and landscape level consistency. Ecography 26(6):739–750
Garcia D, Zamora R, Hódar JA, Gómez JM, Castro J (2000) Yew (Taxus baccata L.) regeneration is facilitated by fleshy-fruited shrubs in mediterranean environments. Biol Conserv 95(1):31–38
Garcia D, Obeso JR, Martinez I (2005) Spatial concordance between seed rain and seedling establishment in bird dispersed trees: does scale matter? J Ecol 93(4):693–704
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica et Biophysica Acta (BBA)-General Subjects 990(1):87–92
Givnish TJ (1988) Adaptation to sun and shade: a whole-plant perspective. Funct Plant Biol 15(2):63–92
Hilfiker K, Gugerli F, Schütz JP, Rotach P, Holderegger R (2004) Low RAPD variation and female-biased sex ratio indicate genetic drift in small populations of the dioecious conifer Taxus baccata in Switzerland. Conserv Genet 5(3):357–365
Iszkulo G (2010) Success and failure of endangered tree species: low temperatures and low light availability affect survival and growth of European yew (Taxus baccata L.) seedlings. Polish J Ecol 58(2):259–271
Iszkulo G, Boratynski A (2004) Interaction between canopy tree species and European yew Taxus baccata(Taxaceae). Polish J Ecol 52(4):523–531
Iszkulo G, Boratynski A (2006) Analysis of the relationship between photosynthetic photon flux density and natural Taxus baccata seedlings occurrence. Acta Oecol 29(1):78–84
Iszkulo G, Lewandowski A, Jasinska AK, Dering M (2007) Light limitation of growth in 10-year-old seedlings of Taxus baccata L. (European yew). Polish J Ecol 55(4):827–831
Iszkulo G, Jasinska AK, Giertych MJ, Boratynski A (2009) Do secondary sexual dimorphism and female intolerance to drought influence the sex ratio and extinction risk of Taxus baccata? Plant Ecol 200(2):229–240
Jackson G, Sheldon J (1949) The vegetation of magnesian limestone cliffs at Markland Grips near Sheffield. J Ecol 38–50
Jansen MAK, Mattoo AK, Edelman M (1999) D1-D2 protein degradation in the chloroplast. Eur J Biochem 260(2):527–532
Kobe RK, Pacala SW, Silander JA Jr, Canham CD (1995) Juvenile tree survivorship as a component of shade tolerance. Ecol Appl 5(2):517–532
Krol S (1978) An outline of ecology. In The yew—Taxus baccata L. Foreign Scientific Publication, Department of the National Centre for Scientific and Technical, and Economics Information (for the Department of Agriculture and the National Science Foundation, Washington, DC), Warsaw, Poland, pp 65–86
Kunstler G, Coomes DA, Canham CD (2009) Size dependence of growth and mortality influence the shade tolerance of trees in a lowland temperate rain forest. J Ecol 97(4):685–695
Little DJ (1994) Occurences and characteristics of podzols under oak woodland in Ireland PhD Thesis, Trinity College Dublin
Luomala E, Laitinen K, Sutinen S, Kellomäki S, Vapaavuori E (2005) Stomatal density, anatomy and nutrient concentrations of Scots pine needles are affected by elevated CO2 and temperature. Plant, Cell Environ 28(6):733–749
Lusk C (2004) Leaf area and growth of juvenile temperate evergreens in low light: species of contrasting shade tolerance change rank during ontogeny. Funct Ecol 18(6):820–828
Lusk CH, Jorgensen M (2013) The whole-plant compensation point as a measure of juvenile tree light requirements. Funct Ecol 27(6):1286–1294
Lusk C, Falster D, Jara Vergara C, Jimenez Castillo M, Saldaña Mendoza A (2008) Ontogenetic variation in light requirements of juvenile rainforest evergreens. Funct Ecol 22(3):454–459
Martínez I, García D, Obeso JR (2008) Differential seed dispersal patterns generated by a common assemblage of vertebrate frugivores in three fleshy-fruited trees. Ecoscience 15(2):189–199
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51(345):659–668
McKenna B (2003) An inventory of Arbutus unedo (Arbutus) and Taxus baccata (Yew) at Glengarriff wood nature reserve-A GIS approach Higher Diploma Dissertation, University College Cork
Messier C, Doucet R, Ruel JC, Claveau Y, Kelly C, Lechowicz MJ (1999) Functional ecology of advance regeneration in relation to light in boreal forests. Can J For Res 29(6):812–823
Mitamura M, Yamamura Y, Nakano T (2009) Large-scale canopy opening causes decreased photosynthesis in the saplings of shade-tolerant conifer Abies veitchii. Tree Physiol 29(1):137
Mitchell A (1998) Acclimation of Pacific yew (Taxus brevifolia) foliage to sun and shade. Tree Physiol 18(11):749
Mitchell A, Arnott J (1995) Effects of shade on the morphology and physiology of amabilis fir and western hemlock seedlings. New For 10(1):79–98
Monclus R, Dreyer E, Villar M, Delmotte FM, Delay D, Petit JM et al (2006) Impact of drought on productivity and water use efficiency in 29 genotypes of Populus deltoides × Populus nigra. New Phytol 169(4):765–777
Myking T, Vakkari P, Skrøppa T (2009) Genetic variation in northern marginal Taxus baccata L. populations. Implications for conservation. Forestry 82(5):529
Niinemets Ü, Valladares F (2006) Tolerance to shade, drought, and waterlogging of temperate Northern Hemisphere trees and shrubs. Ecol Monogr 76(4):521–547
O’Neill FH (2003) Regeneration of Native Oak Woodland following Rhododendron ponticum L. clearance from Glengarriff Woods Nature Reserve, Co. Cork, Ireland. PhD. Thesis, University College Cork
Oquist G, Huner N (1991) Effects of cold acclimation on the susceptibility of photosynthesis to photoinhibition in Scots pine and in winter and spring cereals: a fluorescence analysis. Funct Ecol 5:91–100
Oxborough K, Baker NR (1997) Resolving chlorophyll a fluorescence images of photosynthetic efficiency into photochemical and non-photochemical components—calculation of qP and Fv-/Fm-; without measuring Fo. Photosynth Res 54(2):135–142
Perrin PM, Mitchell FJ (2013) Effects of shade on growth, biomass allocation and leaf morphology in European yew (Taxus baccata L.). Eur J For Res 132:211–218
Perrin PM, Kelly DL, Mitchell FJG (2006) Long-term deer exclusion in yew-wood and oakwood habitats in southwest Ireland: natural regeneration and stand dynamics. For Ecol Manag 236(2–3):356–367
Piovesan G, Saba EP, Biondi F, Alessandrini A, Di Filippo A, Schirone B (2009) Population ecology of yew (Taxus baccata L.) in the Central Apennines: spatial patterns and their relevance for conservation strategies. Plant Ecol 205(1):23–46
Poorter L, Bongers F, Sterck FJ, Woell H (2005) Beyond the regeneration phase: differentiation of height–light trajectories among tropical tree species. J Ecol 93(2):256–267
Reich P, Tjoelker M, Walters M, Vanderklein D, Buschena C (1998) Close association of RGR, leaf and root morphology, seed mass and shade tolerance in seedlings of nine boreal tree species grown in high and low light. Funct Ecol 12(3):327–338
Robakowski P, Wyka T (2009) Winter photoinhibition in needles of Taxus baccata seedlings acclimated to different light levels. Photosynthetica 47(4):527–535
Rodwell JS (1991) British plant communities, woodlands and scrub. Cambridge University Press, Cambridge
Rozendaal D, Hurtado V, Poorter L (2006) Plasticity in leaf traits of 38 tropical tree species in response to light; relationships with light demand and adult stature. Funct Ecol 20(2):207–216
Smith CJ (1980) Ecology of the English chalk. Academic Press, London
Svenning J, Magård E (1999) Population ecology and conservation status of the last natural population of English yew Taxus baccata in Denmark. Biol Conserv 88(2):173–182
Takahashi S, Murata N (2008) How do environmental stresses accelerate photoinhibition? Trends Plant Sci 13(4):178–182
Thomas P, Packham JR (2007) Ecology of woodlands and forests: description, dynamics and diversity. Cambridge Univ Press, Cambridge
Thomas P, Polwart A (2003) Taxus baccata L. J Ecol 91(3):489–524
Tucker G, Emmingham W (1977) Morphological changes in leaves of residual western hemlock after clear and shelterwood cutting. For Sci 23(2):195–203
Valladares F, Niinemets Ü (2008) Shade tolerance, a key plant feature of complex nature and consequences. Annu Rev Ecol Evol Syst 39:237–257
Valladares F, Dobarro I, Sánchez-Gómez D, Pearcy RW (2005) Photoinhibition and drought in mediterranean woody saplings: scaling effects and interactions in sun and shade phenotypes. J Exp Bot 56(411):483–494
Walters MB, Reich PB (1996) Are shade tolerance, survival, and growth linked? Low light and nitrogen effects on hardwood seedlings. Ecology 77(3):841–853
Watt A (1926) Yew communities of the South Downs. J Ecol 14(2):282–316
Wayne P, Bazzaz F (1993) Birch seedling responses to daily time courses of light in experimental forest gaps and shadehouses. Ecology 1500–1515
Wyka T, Robakowski P, Zytkowiak R (2008) Leaf age as a factor in anatomical and physiological acclimative responses of Taxus baccata L. needles to contrasting irradiance environments. Photosynth Res 95(1):87–99
Author contribution statement
All authors conceived and designed the work presented in this paper, and contributed substantially to analysis and interpretation of data. J.L.D. carried out field sampling, with assistance from P.M.W. Chlorophyll fluorescence and needle morphology work was carried out and analysed by J.L.D., with assistance from M.A.K.J. The manuscript was prepared and edited by J.L.D., P.M.W., and M.A.K.J.
Acknowledgments
The authors thank Grace Cott and Alan O’Connor for their assistance in fieldwork. Acknowledgement to National Parks and Wildlife Service, Department of the Environment, Heritage and Local Government for access to some lands. Particular thanks to Declan O’Donnell and Clare Heardman for facilitating research at the Glengarriff Wood Nature Reserve. This study was funded by a University College Cork departmental postgraduate fellowship.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. Bilger.
Rights and permissions
About this article
Cite this article
Devaney, J.L., Whelan, P.M. & Jansen, M.A.K. Light responses of yew (Taxus baccata L.); does size matter?. Trees 29, 109–118 (2015). https://doi.org/10.1007/s00468-014-1095-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-014-1095-x