Abstract
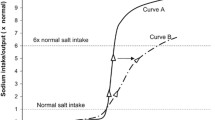
It is now accepted that early life environment can modulate adult phenotype. One of the best documented examples is the effect of prenatal environment on adult hypertension and cardiovascular morbidity. Human epidemiologic studies have been complemented with experimental models showing, for example, that maternal dietary manipulations during pregnancy in the rat can be used to induce adult hypertension in the offspring. The weight of the emerging evidence suggests that abnormal Na handling by the kidney plays an important role in the pathogenesis of the hypertension. Although the number of nephrons is modestly reduced in most experimental models, there is very little change in total glomerular filtration rate, casting doubt on the hypothesis that restricted Na filtration is the major mechanism. Recent studies have instead strongly suggested that renal tubular handling of Na is altered, resulting in an altered set-point for Na balance. The mechanism may involve intrarenal inflammation and increased oxidative stress which disrupt the tubulointerstitial microenvironment, leading to constitutively upregulated Na reabsorption in the distal tubule. The upregulation may be mediated by autocrine and paracrine factors promoting distal tubule Na reabsorption. A similar mechanism has been hypothesized to be important in other types of hypertension and may hence be a common pathway in the genesis of volume-dependent hypertension.




Similar content being viewed by others
References
Barker DJP, Osmond C, Golding J, Kuh D, Wadsworth MEJ (1989) Growth in utero, blood pressure in childhood and adult life, and mortality from cardiovascular disease. Br Med J 298:564–567
Barker DJP (1998) In utero programming of chronic disease. Clin Sci 95:115–128
Curhan GC, Chertow GM, Willett WC, Spiegelman D, Colditz GA, Manson J, Speizer FE, Stampfer MJ (1996) Birth weight and adult hypertension and obesity in women. Circulation 94:1310–1315
Law CM, Shiell AW (1996) Is blood pressure inversely related to birth weight? The strength of evidence from systematic review of the literature. J Hypertens 14:935–941
Moore VM, Cockington RA, Ryan P, Robinson JS (1999) The relationship between birth weight and blood pressure amplifies from childhood to adulthood. J Hypertens 17:883–888
Vehaskari VM, Woods LL (2005) Prenatally programmed hypertension: lessons from experimental models. J Am Soc Nephrol 16:2545–2556
Chong S, Whitelaw E (2004) Epigenetic germline inheritance. Curr Opin Genet Dev 14:692–696
Manning J, Vehaskari VM (2001) Low birth weight-associated adult hypertension in the rat. Pediatr Nephrol 16:417–422
Vehaskari VM, Manning J, Aviles DH (2001) Prenatal programming of adult hypertension in the rat. Kidney Int 59:238–245
Woods LL, Weeks DA, Rasch R (2004) Programming of adult blood pressure by maternal protein restriction: role of nephrogenesis. Kidney Int 65:1339–1348
Ortiz LA, Quan A, Zarzar F, Weinberg A, Baum M (2003) Prenatal dexamethasone programs hypertension and renal injury in the rat. Hypertension 41:328–334
Alexander BT (2003) Placental insufficiency leads to development of hypertension in growth-restricted offspring. Hypertension 41:457–462
Dodic M, May CN, Wintour EM, Coghlan JP (1998) An early prenatal exposure to excess glucocorticoid leads to hypertensive offspring in sheep. Clin Sci 94:149–155
Dodic M, Hantzis V, Duncan J, Rees S, Koukoulas I, Johnson K, Wintour EM, Moritz K (2002) Programming effects of short prenatal exposure to cortisol. FASEB J 16:1017–1026
Manning J, Vehaskari VM (2005) Postnatal modulation of prenatally programmed hypertension by dietary Na and ACE inhibition. Am J Physiol 288:R80–R84
Brenner BM, Garcia DL, Anderson S (1988) Glomeruli and blood pressure: less of one, more the other? Am J Hypertens 1:335–347
Keller G, Zimmer G, Mall G, Ritz E, Amann K (2003) Nephron number in patients with primary hypertension. N Engl J Med 348:101–108
Lifton RP, Gharavi AG, Geller DS (2001) Molecular mechanisms of human hypertension. Cell 101:545–556
Manning J, Buetler K, Knepper MA, Vehaskari VM (2002) Upregulation of BSC1 and TSC in prenatally programmed hypertension. Am J Physiol 283:F202–F206
Ahn D, Ge Y, Stricklett PK, Gill P, Taylor D, Hughes AK, Yanagisawa M, Miller L, Nelson RD, Kohan DE (2004) Collecting duct-specific knockout of endothelin-1 causes hypertension and sodium retention. J Clin Invest 114:504–511
Plato GF, Pollock DM, Garvin JF (2000) Endothelin inhibits thick ascending limb chloride flux via ETb receptor-mediated NO release. Am J Physiol 279:F326–F333
Ortiz PA, Garvin JL (2002) Role of nitric oxide in the regulation of nephron transport. Am J Physiol 282:F777–F784
Ye W, Zhang H, Hillas E, Kohan D, Miller RL, Nelson RD, Honeggar M, Yang T (2005) Expression and function of COX isoforms in renal medulla: evidence for regulation of salt sensitivity and blood pressure. Am J Physiol 290:F542–F549
Mattson DL (2003) Importance of the renal medullary circulation in the control of sodium excretion and blood pressure. Am J Physiol 284:R13–R27
Wilcox CS (2005) Oxidative stress and nitric oxide deficiency in the kidney: a critical link to hypertension? Am J Physiol 289:R913–R935
Rodriguez-Iturbe B, Quiroz Y, Herrera-Acosta J, Johnson RJ, Pons HA (2002) The role of immune cells infiltrating the kidney in the pathogenesis of salt-sensitive hypertension. J Hypertens 20:S9–S14
Rodriguez-Iturbe B, Vaziri ND, Herrera-Acosta J, Johnson RJ (2004) Oxidative stress, renal infiltration of immune cells, and salt-sensitive hypertension: all for one and one for all. Am J Physiol 286:F606–F616
Stewart T, Jung FF, Manning J, Vehaskari VM (2005) Kidney immune cell infiltration and oxidative stress contribute to prenatally programmed hypertension. Kidney Int 68:2180–2188
Kitiyakara C, Chabrashvili T, Chen Y, Blau J, Karber A, Aslam S, Welch W, Wilcox CS (2003) Salt intake, oxidative stress, and renal expression of NADPH oxidase and superoxide dismutase. J Am Soc Nephrol 14:2775–2782
Taylor NE, Glocka P, Liang M, Cowley AW Jr (2006) NADPH oxidase in the renal medulla causes oxidative stress and contributes to salt-sensitive hypertension in Dahl S rats. Hypertension 47:692–698
Alexander BT, Hendon AE, Ferril G, Dwyer TM (2005) Renal denervation abolishes hypertension in low-birth-weight offspring from pregnant rats with reduced uterine perfusion. Hypertension 45:754–758
Acknowledgements
The work in the author’s laboratory was supported by National Heart, Lung, and Blood Institute Grant 1 RO1 HL66158 and by contract HEF (2001-06)-07 from Health Excellence Fund of Louisiana Board of Regents.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vehaskari, V.M. Developmental origins of adult hypertension: new insights into the role of the kidney. Pediatr Nephrol 22, 490–495 (2007). https://doi.org/10.1007/s00467-006-0353-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-006-0353-6