Abstract
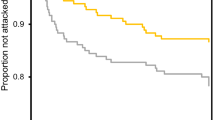
Animals often announce their unprofitability to predators through conspicuous coloured signals. Here we tested whether the apparently conspicuous colour designs of the four European Coraciiformes and Upupiformes species may have evolved as aposematic signals, or whether instead they imply a cost in terms of predation risk. Because previous studies suggested that these species are unpalatable, we hypothesized that predators could avoid targeting them based on their colours. An experiment was performed where two artificial models of each bird species were exposed simultaneously to raptor predators, one painted so as to resemble the real colour design of these birds, and the other one painted using cryptic colours. Additionally, we used field data on the black kite’s diet to compare the selection of these four species to that of other avian prey. Conspicuous models were attacked in equal or higher proportions than their cryptic counterparts, and the attack rate on the four species increased with their respective degree of contrast against natural backgrounds. The analysis of the predator’s diet revealed that the two least attacked species were negatively selected in nature despite their abundance. Both conspicuous and cryptic models of one of the studied species (the hoopoe) received fewer attacks than cryptic models of the other three species, suggesting that predators may avoid this species for characteristics other than colour. Globally, our results suggest that the colour of coraciiforms and upupiforms does not function as an aposematic signal that advises predators of their unprofitability, but also that conspicuous colours may increase predation risk in some species, supporting thus the handicap hypothesis.




Similar content being viewed by others
References
Alatalo RV, Mappes J (1996) Tracking the evolution of warning signals. Nature 382:708–710
Avilés JM (2008) Egg colour mimicry in the common cuckoo Cuculus canorus as revealed by modelling host retinal function. Proc R Soc Lond B 275:2345–2352
Avilés JM, Parejo D (1997) Dieta de los pollos de carraca (Coracias garrulus) en una zona mediterránea (Extremadura, suroeste de España). Ardeola 44:237–239
Avilés JM, Parejo D (2002) Diet and prey type selection by rollers (Coracias garrulus) during the breeding season in southwestern Iberian Peninsula. Alauda 70:227–230
Avilés JM, Soler JJ (2009) Nestling colouration is adjusted to parent visual performance in altricial birds. J Evol Biol 22:376–386
Avilés JM, Pérez-Contreras T, Navarro C, Soler JJ (2008) Dark nests and conspicuousness in color patterns of nestlings of altricial birds. Am Nat 171:327–338
Avilés JM, Vikan JR, Fossoy F, Antonov A, Moksnes A, Roskaft E, Stokke BG (2010) Avian colour perception predicts behavioural responses to experimental brood parasitism in chaffinches. J Evol Biol 23:293–301
Baker RR, Parker GA (1979) The evolution of bird colouration. Philos Trans R Soc Lond B 287:63–130
Bohlin T, Tullberg BS, Merilaita S (2008) The effect of signal appearance and distance on detection risk in an aposematic butterfly larva (Parnassius apollo). Anim Behav 76:577–584
Caro T (2005) Antipredator defenses in birds and mammals. Chicago University Press, Chicago
Cassey P, Honza M, Grim T, Hauber ME (2008) The modelling of avian visual perception predicts behavioural rejection responses to foreign egg colours. Biol Lett 4:515–517
Colombelli-Negrel D, Kleindorfer S (2010) Video nest monitoring reveals male coloration-dependant nest predation and sex differences in prey size delivery in a bird under high sexual selection. J Ornithol 151:507–512
Cott HB (1940) Adaptive coloration in animals. Methuen, London
Cott HB (1947) The edibility of birds: illustrated by 5 years’ experiments and observations (1941–1946) on the food preferences of the hornet, cat and man; and considered with special reference to the theories of adaptive coloration. Proc Zool Soc Lond 116:371–524
Cramp S, Perrins C (1998) The complete birds of the Western Paleartic on CD-ROM. Oxford University Press, Oxford
Cresswell W, Quinn JL (2004) Faced with a choice, sparrowhawks more often attack the more vulnerable prey group. Oikos 104:71–76
Cresswell W, Lind J, Kaby U, Quinn JL, Jakobsson S (2003) Does a opportunistic predator preferentially attack nonvigilant prey? Anim Behav 66:643–648
Cuthill IC, Stevens M, Sheppard J, Maddocks T, Parraga CA, Troscianko TS (2005) Disruptive coloration and background pattern matching. Nature 434:72–74
Darwin C (1871) The descent of man and selection in relation to sex. Murray, London
Dumbacher JP, Pruett-Jones S (1996) Avian chemical defense. In: Nolan V, Ketterson ED (eds) Current ornithology. Plenum, New York, pp 137–174
Dumbacher JP, Beehler BM, Spande TF, Garraffo HM (1992) Homobatrachotoxin in the genus Pitohui: chemical defense in birds? Science 258:799–801
Dumbacher JP, Spande TF, Daly JW (2000) Batrachotoxin alkaloids from passerine birds: a second toxic bird genus (Ifrita kowaldi) from New Guinea. Proc Natl Acad Sci USA 97:12970–12975
Dumbacher JP, Wako A, Derrickson SR, Samuelson A, Spande TF, Daly JW (2004) Melyrid beetles (Choresine): a putative source for the batrachotoxin alkaloids found in poison-dart frogs and toxic passerine birds. Proc Natl Acad Sci USA 101:15857–15860
Edmunds M (1974) Defence in animals: a survey of antipredator defences. Longman, Harlow
Endler J (1978) A predator’s view of animal colour patterns. Evol Biol 11:319–364
Escalante P, Daly JW (1994) Alkaloids in extracts of feathers of the red-warbler. J Ornithol 135:410
Exnerova A, Svadova K, Stys P, Barcalova S, Landova E, Prokopova M, Fuchs R, Socha R (2006) Importance of colour in the reaction of passerine predators to aposematic prey: experiments with mutants of Pyrrhocoris apterus (Heteroptera). Biol J Linn Soc 88:143–153
Fry CH (2001) Family Coraciidae (rollers). In: del Hoyo J, Elliot A, Sargatal J (eds) Handbook of the birds of the world. Mousebirds to hornbills. Lynx, Barcelona, pp 342–377
Getty T (2006) Sexually selected signals are not similar to sports handicaps. Trends Ecol Evol 21:83–88
Goldsmith TH, Butler BK (2005) Color vision of the budgerigar (Melopsittacus undulatus): hue matches, tetrachromacy, and intensity discrimination. J Comp Physiol 191:933–951
Gomez D (2006) AVICOL, a program to analyse spectrometric data. http://sites.google.com/site/avicolprogram/
Götmark F (1992) Antipredator effect of conspicuous plumage in a male bird. Anim Behav 44:51–55
Götmark F (1993) Conspicuous coloration in male birds is favored by predation in some species and disfavoured in others. Proc R Soc Lond B 253:143–146
Götmark F (1994) Are bright birds distasteful? A reanalysis of H. B. Cott’s data on the edibility of birds. J Avian Biol 25:184–197
Götmark F (1996) Simulating a colour mutation: conspicuous red wings in the European blackbird reduce the risk of attacks by sparrowhawks. Funct Ecol 10:355–359
Götmark F (1997) Bright plumage in the magpie: does it increase or reduce the risk of predation? Behav Ecol Sociobiol 40:41–49
Greenberg R (1983) The role of neophobia in determining the degree of foraging specialization in some migrant warblers. Am Nat 122:444–453
Halpin CG, Skelhorn J, Rowe C (2008) Being conspicuous and defended: selective benefits for the individual. Behav Ecol 19:1012–1017
Ham AD, Ihalainen E, Lindström L, Mappes J (2006) Does colour matter? The importance of colour in avoidance learning, memorability and generalisation. Behav Ecol Sociobiol 60:482–491
Hart NS (2002) Vision in the peafowl (Aves: Pavo cristatus). J Exp Biol 205:3925–3935
Håstad O, Victorsson J, Ödeen A (2005) Differences in color vision make passerines less conspicuous in the eyes of their predators. Proc Natl Acad Sci USA 102:6391–6394
Hill GE (2011) Condition-dependent traits as signals of the functionality of vital cellular processes. Ecol Lett 14:625–634
Jacobs J (1974) Quantitative measurement of food selection. A modification of the forage ratio and Ivlev’s electivity index. Oecologia 14:413–417
Johansen AI, Exnerova A, Svadova KH, Stys P, Gamberale-Stille G, Tullberg BS (2010) Adaptive change in protective coloration in adult striated shieldbugs Graphosoma lineatum (Heteroptera: Pentatomidae): test of detectability of two colour forms by avian predators. Ecol Entomol 35:602–610
Krištin A (2001) Family Upupidae (hoopoe). In: del Hoyo J, Elliot A, Sargatal J (eds) Handbook of the birds of the world. Mousebirds to hornbills. Lynx, Barcelona, pp 396–411
Langerhans RB (2007) Evolutionary consequences of predation: avoidance, escape, reproduction, and diversification. In: Elewa AMT (ed) Predation in organisms: a distinct phenomenon. Springer, Heidelberg, pp 177–220
Lev-Yadun S (2003) Weapon (thorn) automimicry and mimicry of aposematic colorful thorns in plants. J Theor Biol 224:183–188
Lev-Yadun S, Ne’eman G (2004) When may green plants be aposematic? Biol J Linn Soc 81:413–416
Lindstedt C, Lindström L, Mappes J (2009) Thermoregulation constrains effective warning signal expression. Evolution 63:469–478
Lindström L, Alatalo RV, Mappes J, Riipi M, Vertainen L (1999) Can aposematic signals evolve by gradual change? Nature 397:249–251
Maan ME, Cummings ME (2009) Sexual dimorphism and directional sexual selection on aposematic signals in a poison frog. Proc Natl Acad Sci USA 106:19072–19077
Mappes J, Alatalo RV (1997) Batesian mimicry and signal accuracy. Evolution 51:2050–2053
Mappes J, Marples N, Endler JA (2005) The complex business of survival by aposematism. Trends Ecol Evol 20:598–603
Martín-Platero AM, Valdivia E, Ruiz-Rodríguez M, Soler JJ, Martín-Vivaldi M, Maqueda M, Martínez-Bueno M (2006) Characterization of antimicrobial substances produced by Enterococcus faecalis MRR 10-3, isolated from the uropygial gland of the hoopoe (Upupa epops). Appl Environ Microbiol 72:4245–4249
Martín-Vivaldi M, Peña A, Peralta-Sánchez JM, Sánchez L, Ananou S, Ruiz-Rodríguez M, Soler JJ (2010) Antimicrobial chemicals in hoopoe preen secretions are produced by symbiotic bacteria. Proc R Soc Lond B 277:123–130
Masson JR, Clark L (2000) The chemical senses in birds. In: Whittow G (ed) Sturkie’s avian physiology. Academic Press, New York, pp 39–56
Mayr G (2008) Avian higher-level phylogeny: well supported clades and what we can learn from a phylogenetic analyses of 2954 morphological characters. J Zool Syst Evol Res 46:63–72
Murphy TG (2006) Dishonest “preemptive” pursuit-deterrent signal? Why the turquoise-browed motmot wags its tail before feeding nestlings. Anim Behav 73:965–970
Niskanen M, Mappes J (2005) Significance of the dorsal zigzag pattern of Vipera latastei gaditana against avian predators. J Avian Biol 74:1091–1101
Ödeen A, Håstad O (2003) Complex distribution of avian color vision systems revealed by sequencing the SWS1 opsin from total DNA. Mol Biol Evol 20:855–861
Quinn JL, Cresswell W (2004) Predator hunting behavior and prey vulnerability. J Anim Ecol 73:143–154
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org
Riipi M, Alatalo RV, Lindström L, Mappes J (2001) Multiple benefits of gregariousness cover detectability costs in aposematic aggregations. Nature 413:512–514
Rowe C, Guilford T (1999) The evolution of multimodal warning displays. Evol Ecol 13:655–671
Ruiz-Rodríguez M, Valdivia E, Soler JJ, Martín-Vivaldi M, Martín-Platero AM, Martínez-Bueno M (2009) Symbiotic bacteria living in the hoopoe’s uropygial gland prevent feather degradation. J Exp Biol 212:3621–3626
Ruxton GD, Speed M, Sherratt TN (2004) Evasive mimicry: when (if ever) could mimicry based on difficulty of capture evolve? Proc R Soc Lond B 271:2135–2142
Sergio F, Blas J, Hiraldo F (2009) Predictors of floater status in a long-lived bird: a cross-sectional and longitudinal test of hypotheses. J Anim Ecol 78:109–118
Sergio F, Blas J, Blanco G, Tanferna A, López L, Lemus J, Hiraldo F (2011) Raptor nest decorations are a reliable threat against conspecifics. Science 331:327–330
Siefferman L, Wang YJ, Wang YP, Yuan HS (2007) Sexual dichromatism, dimorphism, and condition-dependent coloration in blue-tailed bee-eaters. Condor 109:577–584
Silva N, Avilés JM, Danchin E, Parejo D (2008) Informative content of multiple plumage-coloured traits in female and male European rollers. Behav Ecol Sociobiol 62:1969–1979
Skelhorn J, Rowe C (2006a) Avian predators taste-reject aposematic prey on the basis of their chemical defence. Biol Lett 2:348–350
Skelhorn J, Rowe C (2006b) Taste-rejection by predators and the evolution of unpalatability in prey. Behav Ecol Sociobiol 60:550–555
Sokal RR, Rohlf FJ (1995) Biometry, 3rd edn. Freeman, San Francisco
Soler JJ, Martín-Vivaldi M, Ruiz-Rodríguez M, Valdivia E, Martín-Platero AM, Martínez-Bueno M, Peralta-Sánchez JM, Méndez M (2008) Symbiotic association between hoopoes and antibiotic-producing bacteria that live in their uropygial gland. Funct Ecol 22:864–871
Stevens M (2007) Predator perception and the interrelation between different forms of protective coloration. Proc R Soc Lond B 274:1457–1464
Stevens M, Merilaita S (2009) Defining disruptive colouration and distinguishing its functions. Philos Trans R Soc Lond B 364:481–488
Thompson WL (2002) Towards reliable bird surveys: accounting for individuals present but not detected. Auk 119:18–25
Tullberg BS, Merilaita S, Wiklund C (2005) Aposematism and crypsis combined as a result of distance dependence: functional versatility of the colour pattern in the swallowtail butterfly larva. Proc R Soc Lond B 272:1315–1321
Vorobyev M, Osorio D (1998) Receptor noise as a determinant of colour thresholds. Proc R Soc Lond B 265:351–358
Vorobyev M, Osorio D, Bennett ATD, Marshall NJ, Cuthill IC (1998) Tetrachromacy, oil droplets and bird plumage colours. J Comp Physiol 183:621–633
Weldon PJ (2000) Avian chemical defense: toxic birds not of a feather. Proc Natl Acad Sci USA 97:12948–12949
Weldon PJ, Rappole JH (1997) A survey of birds odorous or unpalatable to humans: possible indications of chemical defense. J Chem Ecol 23:2609–2633
Zahavi A, Zahavi A (1999) The handicap principle: a missing piece of Darwin’s puzzle. Oxford University Press, New York
Zuk M, Kolluru GR (1998) Exploitation of sexual signals by predators and parasitoids. Q Rev Biol 73:415–438
Acknowledgments
We thank all the volunteers who helped with fieldwork: R. Barrientos, G. Calvo, F. J. Chicano, M. García, O. Gordo, F. Goytre, M. Méndez, J. M. Peralta-Sánchez, M. J. Ruiz and J. J. Soler. M. Máñez, J. L. Arroyo, I. San Martín and the Equipo de Seguimiento de Procesos Naturales of the Estación Biológica de Doñana kindly provided access to their avian surveys. We thank all the personnel of the RBD for logistical support, and J. Moya for statistical advice. We also thank Dr James Dale and two anonymous referees for their straightforward suggestions that greatly improved the manuscript. The Consejería de Medio Ambiente of the Junta de Andalucía authorized our work within the areas with restricted access in the Natural Space of Doñana. This work was funded by projects RNM-02177 and RNM-03822 of the Junta de Andalucía, and CGL2008-01781 and CGL2010-19233-C03-03 of the Ministerio de Ciencia e Innovación.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Oliver Love.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MPG 7,787 kb)
Supplementary material 2 (MPG 5,182 kb)
Rights and permissions
About this article
Cite this article
Ruiz-Rodríguez, M., Avilés, J.M., Cuervo, J.J. et al. Does avian conspicuous colouration increase or reduce predation risk?. Oecologia 173, 83–93 (2013). https://doi.org/10.1007/s00442-013-2599-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-013-2599-6