Abstract
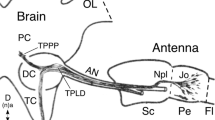
Each neural unit (cartridge) in the first optic ganglion (lamina) of the nocturnal bee Megalopta genalis contains nine receptor cell axons (6 short and 3 long visual fibres), and four different types of first-order interneurons, also known as L-fibres (L1 to L4) or lamina monopolar cells. The short visual fibres terminate within the lamina as three different types (svf 1, 2, 3). The three long visual fibres pass through the lamina without forming characteristic branching patterns and terminate in the second optic ganglion, the medulla. The lateral branching pattern of svf 2 into adjacent cartridges is unique for hymenopterans. In addition, all four types of L-fibres show dorso-ventrally arranged, wide, lateral branching in this nocturnal bee. This is in contrast to the diurnal bees Apis mellifera and Lasioglossum leucozonium, where only two out of four L-fibre types (L2 and L4) reach neighbouring cartridges. In M. genalis, L1 forms two sub-types, viz. L1-a and L1-b; L1-b in particular has the potential to contact several neighbouring cartridges. L2 and L4 in the nocturnal bee are similar to L2 and L4 in the diurnal bees but have dorso-ventral arborisations that are twice as wide. A new type of laterally spreading L3 has been discovered in the nocturnal bee. The extensive neural branching pattern of L-fibres in M. genalis indicates a potential role for these neurons in the spatial summation of photons from large groups of ommatidia. This specific adaptation in the nocturnal bee could significantly improve reliability of vision in dim light.






Similar content being viewed by others
References
Amiet F, Herrmann M, Müller A, Neumeyer R (2001) Fauna Helvetica. Apidae 3: Halictus, Lasioglossum. Schweizer Entomologische Gesellschaft, Neuchâtel
Dyer FC (1985) Nocturnal orientation by the Asian honey bee, Apis dorsata. Anim Behav 33:769–774
Fletcher DJC (1978) The African bee, Apis mellifera adansonii, in Africa. Annu Rev Entomol 23:151–171
Greiner B, Ribi WA, Warrant EJ (2004) Retinal and optical adaptations for nocturnal vision in the halictid bee Megalopta genalis. Cell Tissue Res 316:377-390
Kral K (1987) Organization of the first optic neuropil (or lamina) in different insect species. In: Gupta AP (ed) Arthropod brain: its evolution, development, structure, and functions. Wiley, New York, pp 181–201
Meinertzhagen IA, Armett-Kibel C (1982) The lamina monopolar cells in the optic lobe of the dragonfly Sympetrum. Philos Trans R Soc Lond Biol 297:27–49
Melzer RR, Zimmermann T, Smola U (1997) Modification of dispersal patterns of branched photoreceptor axons and the evolution of neural superposition. Cell Mol Life Sci 53:242–247
Meyer EP (1979) Golgi-EM study of first and second order neurons in the visual system of Cataglyphis bicolor Fabricius. Zoomorphologie 92:115–139
Nilsson D-E (1989) Optics and evolution of the compound eye. In: Stavenga DG, Hardie R (eds) Facets of vision. Springer, Berlin Heidelberg New York, pp 30–73
Nilsson D-E, Ro A-I (1994) Did neural pooling for night vision lead to the evolution of neural superposition eyes? J Comp Physiol [A] 175:289–302
Nowel MS, Shelton PMJ (1981) A Golgi-electron microscopical study of the structure and development of the lamina ganglionaris of the locust optic lobe. Cell Tissue Res 216:377–401
Ohly KP (1975) The neurons of the first synaptic region of the optic neuropil of the firefly, Phausis splendidula L. Coleoptera. Cell Tissue Res 158:89–109
Ribi WA (1975a) The first optic ganglion of the bee. I. Correlation between visual cell types and their terminals in the lamina and medulla. Cell Tissue Res 165:103–111
Ribi WA (1975b) The neurons of the first optic ganglion of the bee (Apis mellifera). Adv Anat Embryol Cell Biol 50:5–43
Ribi WA (1976) The first optic ganglion of the bee. II. Topographical relationship of the monopolar cells within and between cartridges. Cell Tissue Res 171:359–373
Ribi WA (1977) Fine structure of the first optic ganglion (lamina) of the cockroach Periplaneta americana. Tissue Cell 9:57–72
Ribi WA (1981) The first optic ganglion of the bee. IV. Synaptic fine structure and connectivity patterns of receptor cell axons and first order interneurones. Cell Tissue Res 215:443–464
Ribi WA (1983) Electron microscopy of Golgi-impregnated neurons. In: Strausfeld NJ (ed) Functional neuroanatomy. Springer, Berlin Heidelberg New York, pp 1–18
Ribi WA (1987a) Anatomical identification of spectral receptor types in the retina and lamina of the Australian orchard butterfly, Papilio aegeus aegeus D. Cell Tissue Res 247:393–407
Ribi WA (1987b) The structural basis of information processing in the visual system of the bee. In: Menzel R, Mercer A (eds) Neurobiology and behavior of honeybees. Springer, Berlin Heidelberg New York, pp 130–140
Shimohigashi M, Tominaga Y (1999) Synaptic organization in the lamina of the superposition eye of a skipper butterfly, Parnara guttata. J Comp Neurol 408:107–124
Srinivasan MV, Bernard CG (1975) The effect of motion on visual acuity of the compound eye: a theoretical analysis. Vision Res 15:515–525
Strausfeld NJ, Blest AD (1970) Golgi studies on insects. Part I. The optic lobes of Lepidoptera. Philos Trans R Soc Lond Biol 258:81–134
Warrant EJ (1993) Can neural pooling help insects see at night? Proc IEEE Int Conf Syst Man Cybern 3:150–155
Warrant EJ (1999) Seeing better at night: life style, eye design and the optimum strategy of spatial and temporal summation. Vision Res 39:1611–1630
Warrant E, Porombka T, Kirchner WH (1996) Neural image enhancement allows honeybees to see at night. Proc R Soc Lond [Biol] 263:1521–1526
Warrant EJ, Kelber A, Gislén A, Greiner B, Ribi WA, Wcislo WT (2004) Nocturnal vision and landmark orientation in a tropical halictid bee. Curr Biol 14:1309–1318
Wolburg-Buchholz K (1979) The organization of the lamina ganglionaris of the hemipteran insects, Notonecta glauca, Corixa punctata and Gerris lacustris. Cell Tissue Res 197:39–59
Acknowledgements
We thank Almut Kelber, Marie Dacke, Anna Balkenius and the two anonymous reviewers for critically reading the manuscript, Mikael Sörenson and Jan Tengö for taxonomical assistance, Victor Gonzales, Sara Juhl, Rikard Frederiksen and Mark Holdstock for help with fieldwork, the staff of the Smithonian Tropical Research Institute for their helpfulness, and the Autoridad Nacional del Ambiente of the Republic of Panama for permission to export bees.
Author information
Authors and Affiliations
Corresponding author
Additional information
B.G. is grateful for travel awards from the Royal Physiographic Society, the Per Westlings Fond, the Foundation of Dagny and Eilert Ekvall and the Royal Swedish Academy of Sciences. E.J.W. acknowledges the receipt of a Smithsonian Short-Term Research Fellowship and thanks the Swedish Research Council, the Crafoord Foundation, the Wenner–Gren Foundation and the Royal Physiographic Society of Lund for their ongoing support. W.T.W. was supported by general research funds from the Smithonian Tropical Research Institute
Rights and permissions
About this article
Cite this article
Greiner, B., Ribi, W.A., Wcislo, W.T. et al. Neural organisation in the first optic ganglion of the nocturnal bee Megalopta genalis. Cell Tissue Res 318, 429–437 (2004). https://doi.org/10.1007/s00441-004-0945-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-004-0945-z