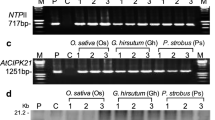
Abstract
Calcium is a ubiquitous signaling molecule and changes in cytosolic calcium concentration are involved in plant responses to various stimuli. The rice calcium-dependent protein kinase 13 (CDPK13) and calreticulin interacting protein 1 (CRTintP1) have previously been reported to be involved in cold stress response in rice. In this study, rice lines transformed with sense CDPK13 or CRTintP1 constructs were produced and used to investigate the function of these proteins. When the plants were incubated at 5°C for 3 days, leaf blades of both the sense transgenic and vector control rice plants became wilted and curled. When the plants were transferred back to non-stress conditions after cold treatment, the leaf blades died, but the sheaths remained green in the sense transgenic rice plants. Expression of CDPK13 or CRTintP1 was further examined in several rice varieties including cold-tolerant rice varieties. Accumulation of these proteins in the cold-tolerant rice variety was higher than that in rice varieties that are intermediate in their cold tolerance. To examine whether over-expression of CDPK13 and CRTintP1 would have any effect on the proteins or not, sense transgenic rice plants were analyzed using proteomics. The 2D-PAGE profiles of proteins from the vector control were compared with those of the sense transgenic rice plants. Two of the proteins that differed between these lines were calreticulins. The results suggest that CDPK13, calreticulin and CRTintP1 might be important signaling components for response to cold stress in rice.







Similar content being viewed by others
Abbreviations
- CDPK13:
-
Calcium-dependent protein kinase 13
- CRTintP1:
-
Calreticulin binding protein 1
References
Abbasi F, Onodera H, Toki S, Tanaka H, Komatsu S (2004) OsCDPK13, a calcium-dependent protein kinase gene from rice, is induced by cold and gibberellin in rice leaf sheath. Plant Mol Biol 55:541–552
Abdrakhamanova A, Wang QY, Khokhlova L, Nick P (2003) Is microtubule disassembly a trigger for cold acclimation? Plant Cell Physiol 44:676–686
Asano T, Tanaka N, Yang G, Hayashi N, Komatsu S (2005) Genome-wide identification of the rice calcium-dependent protein kinase and its closely related kinase gene families: comprehensive analysis of the CDPKs gene family in rice. Plant Cell Physiol 46:356–366
Chapman R, Sidrauski C, Walter P (1998) Intracellular signaling from the endoplasmic reticulum to the nucleus. Annu Rev Cell Dev Biol 14:459–485
Clapham DE (1995) Calcium signaling. Cell 80:259–268
Corbett EF, Michalak M (2000) Calcium, a signaling molecule in the endoplasmic reticulum? Trends Biochem Sci 25:307–311
Corbett EF, Oikawa K, Francois P, Tessier DC, Kay C, Bergeron JJ, Thomas DY, Krause KH, Michalak M (1999) Ca2+ regulation of interactions between endoplasmic reticulum chaperones. J Biol Chem 274:6203–6211
Crofts AJ, Leborgne-Castel N, Pesca M, Vitale A, Denecke J (1998) Bip and calreticulin form an abundant complex that is independent of endoplasmic reticulum stress. Plant Cell 10:813–824
Denecke J, Carlsson LE, Vidal S, Hoglund AS, Ek B, van Zeijl ML, Singorgo KMC, Palva ET (1995) The tobacco homolog of mammalian calreticulin is present in protein complexes in vivo. Plant Cell 7:391–406
Gupta A.K, Kaur N (2005) Sugar signaling and gene expression in relation to carbohydrate metabolism under abiotic stress in plants. J Biosci 30:761–776
Hamada T, Hasunuma K, Komatsu S (1999) Phosphorylation of proteins in the stem section of etiolated rice seedling irradiated with red light. Biol Pharm Bull 22:122–126
Harmon AC, Gribskov M, Harper JF (2000) CDPKs—a kinase for every Ca2+ signal? Trends Plant Sci 5:154–159
Hassan A-M, Wesson C, Trumble WR (1995) Calreticulin is the major Ca2+ storage protein in the endoplasmic reticulum of the pea plant (Pisum sativum). Biochem Biophys Res Commun 211:54–59
Hood EE, Helmer GL, Fraley RT, Chilton MD (1986) The hypervirulence of Agrobacterium tumefaciens A 281 is encoded in a region of pTiBo540 outside of T-DNA. J Bacteriol 168:1291–1301
Hrabak EM (2004) Calciudependent protein kinase and their relatives. In: Kris M, Walker JC (eds) Advances in botanical sciences, plant protein kinase, vol. 32, Academic, New York, pp 185–223
Khan M, Takasaki H, Komatsu S (2005) Comprehensive phosphoproteome analysis in rice and identification of phosphoproteins responsive to different hormones/stresses. J Proteome Res 4:1592–1599
Komatsu S, Hirano H (1993) Protein kinase activity and phosphorylation in rice (Oryza sativa L.) leaf. Plant Sci 94:127–137
Komatsu S, Masuda T, Abe K (1996) Phosphorylation of a protein (pp56) is related to the regeneration of rice cultured suspension cells. Plant Cell Physiol 37:748–753
Kwiatkowski BA, Zielinska-Kwiatkowska AG, Migdalski A, Lkeczkowski LA, Wasilewska LD (1995) Cloning of two cDNAs encoding calnexin-like and calreticulin-like proteins from maize (Zea mays) leaves: identification of potential calcium-binding domains. Gene 165:219–222
Li Z, Komatsu S (2000) Molecular cloning and characterization of calreticulin, a calcium-binding protein involved in the regeneration of rice cultured suspension cells. Eur J Biochem 267:737–745
Li Z, Onodera H, Ugaki M, Tanaka H, Komatsu S (2003) Characterization of calreticulin as a phosphoprotein interacting with cold-induced protein kinase in rice. Biol Pharm Bull 26:256–261
Liewelyn RH, Roderick H, Llewelyn DH, Campbell AK, Kendall JM (1998) Role of calreticulin in regulating intracellular Ca2+ storage and capacitative Ca2+ entry in HeLa cells. Cell Calcium 24:253–262
Maldonad R, Sanchez-Ballesta MT, Alique R, Escribano MI, Merodio C (2004) Malate metabolism and adaptation to chilling temperature storage by pretreatment with high CO2 levels in Annona cherimola fruit. J Agric Food Chem 52:4758–4763
McAinsh MR, Brownlee C, Hetherington AM (1992) Visualizing changes in cytosolic-free Ca2+ during the response of stomatal guard cells to abscisic acid. Plant Cell 4:1113–1122
Menegazzi P, Guzzo F, Baldan B, Mariani P, Treves S (1993) Purification of calreticulin-like protein(s) from spinach leaves. Biochem Biophys Res Comm 190:1130–1135
Minarik P, Tomaskova N, Kollarova M, Antalik M (2002) Malate dehydrogenases-structure and function. Gen Physiol Biophys 21:257–265
Miura K (2003) General studies on germination of seed and seedling establishment for breeding of improved rice suitable for direct seedling culture. Misc Publ Natl Inst Agrobiol Sci 2:1–44 (in Japanese)
Monroy AF, Sangwan V, Dhindsa RS (1998) Low temperature signal transduction during cold acclimation: protein phosphatase 2A as an early target for cold-inactivation. Plant J 13:653–660
Nelson DE, Glaunsinger B, Bohnert HJ (1997) Abundant accumulation of the calcium-binding molecular chaperone calreticulin in specific floral tissues of Arabidopsis thaliana. Plant Physiol 114:29–37
O’Farrell PH (1975) High resolution two-dimensional electrophoresis of protein. J Biol Chem 250:4007–4021
Ogawa H, Tarashima K (2001) A varietal difference in coleoptile growth is correlated with seedling establishment of direct seeded rice in submerged field under low-temperature conditions. Plant Prod Sci 4:166–172
Ohta S, Mita S, Hattori T, Nakamura K (1990) Construction and expression in tobacco of a β-glucuronidase (GUS) reporter gene containing an intron within the coding sequence. Plant Cell Physiol 31:805–813
Opas M, Szewczenko-Pawlikowski M, Jass GH, Mesaeli N, Michalak M (1996) Calreticulin modulates cell adhesiveness via regulation of vinculin expression. J Cell Biol 5:1–11
Scrase-Field SA, Knight MR (2003) Calcium: just a chemical switch? Curr Opin Plant Biol 6:500–506
Sharma A, Isogai M, Yamamoto T, Sakaguchi K, Hashimoto J, Komatsu S (2004) A novel interaction between calreticulin and ubiqutin-like nuclear protein in rice. Plant Cell Physiol 45:684–692
Shen S, Sharma A, Komatsu S (2003) Characterization of proteins responsive to gibberellin in the leaf-sheath of rice (Oryza sativa L.) seedling using proteome analysis. Biol Pharm Bull 26:129–136
Thomashow MF (1999) Plant cold accumulation: freezing tolerance genes and regulatory mechanisms. Annu Rev Plant Physiol Plant Mol Biol 50:571–599
Toki S, Hara N, Ono K, Onodera H, Tagiri A, Oka S, Tanaka H (2006) Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J 47:961–976
Vashisht AA, Tuteja N (2005) Cold stress-induced pea DNA helicase 47 is homologous to eIF4A and inhibited by DNA-interacting ligands. Annu Rev Plant Physiol Plant Mol Biol 440:79–90
Wanner LA, Junttila O (1999) Cold-induced freezing tolerance in Arabidopsis. Plant Physiol 120:391–399
Yang G, Shen S, Yang S, Komatsu S (2003) OsCDPK13, a calcium-dependent protein kinase gene from rice, is induced in response to cold and gibberellin. Plant Physiol Biochem 41:369–374
Yang P-F, Li X-J, Liang Y, Jing Y-X, Shen S-H., Kuang T-Y (2006) Proteomic analysis of the response of Liangyoupeijiu (super high-yield hybrid rice) seedlings to cold stress. J Integr Plant Biol 48:945–951
Zielinski RE (1998) Calmodulin and calmodulin-binding proteins in plants. Annu Rev Plant Physiol Plant Mol Biol 49:697–725
Acknowledgments
Authors thank Dr. D. He for his help for basically experiment. Authors are grateful to Dr. K. Nakamura of Nagoya University for providing pIG121-Hm vector for rice transformation, and Dr. E.E.Hood of ProdiGene for providing Agrobacterium strain EHA105. This work was supported by a grant from the Integrated Research for Providing Fresh and Delicious “Brand Nippon” Agricultural-Products, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Rights and permissions
About this article
Cite this article
Komatsu, S., Yang, G., Khan, M. et al. Over-expression of calcium-dependent protein kinase 13 and calreticulin interacting protein 1 confers cold tolerance on rice plants. Mol Genet Genomics 277, 713–723 (2007). https://doi.org/10.1007/s00438-007-0220-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-007-0220-6