Abstract
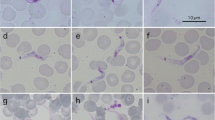
Wild rodents, as natural reservoir hosts carrying various species of pathogens, play an important role in the evolution and emergence of zoonotic diseases. In this study, protist parasites, namely Babesia sp., Trypanosoma sp. and Hepatozoon sp. were studied in rodent populations in Lithuania. Two hundred forty rodent specimens of seven species were analysed by a combined approach using polymerase chain reaction (PCR)-based techniques and traditional microscopic examination. The total prevalence of blood parasites reached 35% in rodent communities. The prevalence of Hepatozoon sp. reached the highest value (32%), followed by Trypanosoma sp. (5%) and Babesia sp. (3%). Myodes glareolus and Microtus agrestis were the most heavily infected rodent species. Comparison of microscopy and PCR-based methods showed that the two approaches might give different results and thus can lead to an underestimation of the actual prevalence and abundance of parasites. In our study, PCR-based assays were more sensitive and robust than traditional microscopy. However, precise molecular results for the estimation of the prevalence of Babesia sp. and Hepatozoon sp. were achieved only by using several sets of primers. To avoid inaccurate results, the improvement and detailed description of molecular and microscopy protocols are required.
Similar content being viewed by others
References
Azmi K, Ereqat S, Nasereddin A, Al-Jawabreh A, Baneth G, Abdeen Z (2016) Molecular detection of Theileria, Babesia, and Hepatozoon spp. in ixodid ticks from Palestine. Ticks Tick Borne Dis 7(5):734–741. https://doi.org/10.1016/j.ttbdis.2016.03.003
Bajer A, Pawelczyk A, Behnke JM, Gilbert F, Siński E (2001) Factors affecting the component community structure of haemoparasites in bank voles (Clethrionomys glareolus) from the Mazury Lake District region of Poland. Parasitology 122:43–54. https://doi.org/10.1017/S0031182000007058
Bajer A, Welc-Falęciak R, Bednarska M, Alsarraf M, Behnke-Borowczyk J, Siński E, Behnke JM (2014) Long-term spatiotemporal stability and dynamic changes in the haemoparasite community of bank voles (Myodes glareolus) in NE Poland. Microb Ecol 68:196–211. http://doi:https://doi.org/10.1007/s00248-014-0390-9
Baker JR (1963) Blood parasites of wild voles, Microtus agrestis, in England. Parasitology 53:297–301. https://doi.org/10.1017/S0031182000072772
Beck R, Vojta L, Ćurković S, Mrljak V, Margaletić J, habrun B (2011) Molecular survey of Babesia microti in wild rodents in Central Croatia. Vector Borne Zoonotic Dis 11(1):81–83. https://doi.org/10.1089/vbz.2009.0260
Beldomenico PM, Telfer S, Gebert S, Lukomski L, Bennett M, Begon M (2009) The vicious circle and infection intensity: the case of Trypanosoma microti in the field vole populations. Epidemics 1:162–167. https://doi.org/10.1016/j.epidem.2009.05.002
Blaschitz M, Narodoslavsky-Gföller M, Kanzler M, Stanek G, Walochnik J (2008) Babesia species occurring in Austrian Ixodes ricinus ticks. Appl Environ Microbiol 74(15):4841–4846. https://doi.org/10.1128/AEM.00035-08
Casati S, Sager H, Gern L, Piffaretti J-C (2006) Presence of potentially pathogenic Babesia sp. for human in Ixodes ricinus in Switzerland. Ann Agric Environ Med 13:65–70
Criado-Fornelio A, Martinez-Marcos A, Buling-Saraña A, Barba-Carretero JC (2003a) Molecular studies on Babesia, Theileria and Hepatozoon in Southern Europe part I. Epizootiological aspects. Vet Parasitol 13:189–201. https://doi.org/10.1016/S0304-4017(03)00078-5
Criado-Fornelio A, Martinez-Marcos A, Buling-Saraña A, Barba-Carretero JC (2003b) Presence of Mycoplasma haemofelis, Mycoplasma haemominutum and piroplasmids in cats from Southern Europe: a molecular study. Vet Microbiol 93:307–317. https://doi.org/10.1016/S0378-1135(03)00044-0
Criado-Fornelio A, Buling A, Cunha-Filho NA, Ruas JL, Farias NAR, Rey-Valeiron C, Pingret JL, Etievant M, Barba-Carretero JC (2007) Development and evaluation of a quantitative PCR assay for detection of Hepatozoon sp. Vet Parasitol 150:352–356. https://doi.org/10.1016/j.vetpar.2007.09.025
Duh D, Petrovec M, Trilar T, Avsic-Zupanac T (2003) The molecular evidence of Babesia microti infection in small mammals collected in Slovenia. Parasitology 126:113–117. https://doi.org/10.1017/S0031182002002743
Gimenez C, Casado N, Criado-Fornelio Á, de Miguel FÁ, Dominguez-Peñafiel G (2009) A molecular survey of Piroplasmida and Hepatozoon isolated from domestic and wild animals in Burgos (northern Spain). Vet Parasitol 162:147–150. https://doi.org/10.1016/j.vetpar.2009.02.021
Hamšikova Z, Kazimírová M, Haruštiakova D, Mahríková L, Slovák M, Berthová L, Kocianová E, Schnittger L (2016a) Babesia spp. in ticks and wildlife in different types of Slovakia. Parasit Vectors 9(1):292. https://doi.org/10.1186/s13071-016-1560-z
Hamšikova Z, Silaghi C, Rudolf I, Venclíkova K, Mahríkova L, Slovák M, Mendel J, Blažejová H, Berthová L, Kocianová E, Hubálek Z, Schnittger L, Kazimímirová M (2016b) Molecular detection and phylogenetic analysis of Hepatozoon spp. in questing Ixodes ricinus ticks and rodents from Slovakia and Czech Republic. Paerasitol Res 115(10):3897–3904. https://doi.org/10.1007/s00436-016-5156-5
Healing TD (1981) Infections with blood parasites in the small British rodents Apodemus sylvaticus, Clethrionomys glareolus and Microtus agrestis. Parasitology 83:179–189. https://doi.org/10.1017/S0031182000050149
Heidarpour Bami M, Haddadzadeh HR, Kazemi B, Khazraiinia P, Bandehpour M, Aktas M (2009) Molecular identification of ovine Theileria species by a new PCR-RFLP method. Vet Parasitol 161:171–177. https://doi.org/10.1016/j.vetpar.2009.01.035
Hellgren O, Waldenström J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon Plasmodium, and Haemoproteus from avian blood. J Parasitol 90:797–802. https://doi.org/10.1645/GE-184R1
Hojgaard A, Lukacik G, Piesman J (2014) Detection of Borrelia burgdorferi, Anaplasma phagocytophilum and Babesia microti, with two different multiplex PCR assays. Ticks Tick Borne Dis 5:349–351. https://doi.org/10.1016/j.ttbdis.2013.12.001
Inokuma H, Okuda M, Ohno K, Shimoda K, Onishi T (2002) Analysis of the 18S rRNA gene sequence of a Hepatozoon detected in two Japanese dogs. Vet Parasitol 106(3):265–271. https://doi.org/10.1016/S0304-4017(02)00065-1
Kallio ER, Begon M, Birtles RJ, Bown KJ, Koskela E, Mappes T, Watts PC (2014) First report of Anaplasma phagocytophilum and Babesia microti in rodents in Finland. Vector Borne Zoonotic Dis 14:389–393. https://doi.org/10.1089/vbz.2013.1383
Karbowiak G (2004) Zoonotic reservoir of Babesia microti in Poland. Pol J Microbiol 53:61–65
Karbowiak G, Rychlik L, Nowakowski W, Wita I (2005) Natural infections of small mammals with blood parasites on the borderland of boreal and temperate forest zones. Acta Theriol 50:31–42. https://doi.org/10.1007/BF03192616
Karbowiak G, Siński E (1996) Occurrence and morphological characteristics of a Trypanosoma evotomys strain from north Poland. Acta Parasitol 14:105–107
Karbowiak G, Stanko M, Fričova J, Wita I, Hapunik J, Peťko B (2009) Blood parasites of the striped field mouse Apodemus agrarius and their morphological characteristics. Biologia 64:1219–1224. https://doi.org/10.2478/s11756-009-0195-3
Karbowiak G, Wita I (2004) Trypanosoma (Herpetosoma) grosi kosewiense subsp. n., the parasite of the yellow-necked mouse Apodemus flavicollis (Melchior, 1834). Acta Protozool 43:173–178
Karbowiak G, Wita I, Czaplinska U (2004) Protozoan parasites in blood of social vole (Microtus socialis) in “Askania-Nova” reserve, Ukraine. Vestn Zool 18:186–187
Karesh WB, Dobson A, Lloyd-Smith JO, Lubroth J, Dixon MA, Bennett M, Aldrich S, Harrington T, Formenty P, Loh EH, Machalaba CC, Thomas MJ, Heymann DL (2012) Ecology of zoonoses: natural and unnatural histories. Lancet 380:1936–1945
Laakkonen J, Kallio-Kokko H, Vapalahti O, Vaheri A, Vyskočilova M, Munclinger P, Macholán M, Henttonen H (2007) The screening of parasites and viral pathogens of small mammals from a farm in Southern Finland, and genetic identification of the Finnish house mouse, Mus musculus. Ann Zool Fennici 44(3):202–208
Laakkonen J, Sukura A, Oksanen A, Henttonen H, Soveri T (2001) Haemogregarines of the genus Hepatozoon (Apicomplexa: Adeleina) in rodents from Northern Europe. Folia Parasitol 48:263–267. https://doi.org/10.14411/fp.2001.043
Li Y, Wang C, Allen KE, Little SE, Ahluwalia SK, Gao D, Macintire DK, Blagburn BL, Kaltenboeck B (2008) Diagnosis of canine Hepatozoon spp. infection by quantitative PCR. Vet Parasitol 157:50–58. https://doi.org/10.1016/j.vetpar.2008.06.027
Maia JP, Harris DJ, Carranza S, Gómez-Díaz E (2014) A comparison of multiple methods for estimating parasitemia of hemogregarine hemoparasites (Apicomplexa: Adeleorina) and its application for studying infection in natural populations. PLoS One 9:e95010. https://doi.org/10.1371/journal.pone.0095010
Najm N, Meyer-KAyser E, Hoffmann L, Herb I, Fensterer V, Pfister K, Silaghi C (2014) A molecular survey of Babesia spp. and Theileria spp. in red foxes (Vulpes vulpes) and their ticks from Thuringi, Germany. Ticks Tick Borne Dis 5(4):386–391. https://doi.org/10.1016/j.ttbdis.2014.01.005
Nakamoto K, Tokoro M, Nakamoto T, Saito-Ito A, Oikawa Y, Takada N, Matsumura T, Arayama S, Kamaruddin M (2014) Widespread distribution of Trypanosoma (Herpetosoma) grosi in Japanese field mice (Apodemus speciosus). Jpn J Vet Parasitol 13(2):67–73
Noyes HA, Ambrose P, Barker F, Begon M, Bennet M, Bown KJ, Kemp SJ (2002) Host specificity of Trypanosoma (Herpetosoma) species: evidence that bank vole (Clethrionomys glareolus) carry only one T. (H.) evotomys 18S rRNA genotype but wood mice (Apodemus sylvaticus) carry at least two polyphyletic parasite. Parsitol 124:185–190. https://doi.org/10.1017/S0031182001001019
Noyes HA, Stevens JR, Teixeira M, Phelan J, Holz P (1999) A nested PCR for the ssrRNA gene detects Trypanosoma binneyi in the platypus and Trypanosoma sp. in wombats and kangaroos in Australia. Int J Parasitol 29(2):331–339. https://doi.org/10.1016/S0020-7519(98)00167-2
Obiegala A, Pfeffer M, Pfister K, Karnath C, Silaghi C (2015) Molecular examination of Babesia microti in rodents and rodent-attached ticks from urban and sylvatic habitats in Germany. Ticks Tick Borne Dis 6(4):445–449. https://doi.org/10.1016/j.ttbdis.2015.03.005
Ord RL, Lobo CA (2015) Human babesiosis: pathogens, prevalence, diagnosis, and treatment. Curr Clin Micro RPT 2:173–181. https://doi.org/10.1007/s40588-015-0025-z
Oyamada M, Davoust B, Boni M, Dereure J, Bucheton B, Hammad A, Itamoto K, Okuda M, Inokuma H (2005) Detection of Babesia canis rossi, B. canis vogeli, and Hepatozoon canis in dogs in a village of eastern Sudan by using a screening PCR and sequencing methodologies. Clin Diagn Lab Immunol 12:1343–1346. https://doi.org/10.1128/CDLI.12.11.1343-1346.2005
Piron M, Fisa R, Casamitjana N, López-Chejade P, Puig L, Vergés M, Gascón J, Gómez i Prat J, Portús M, Sauleda S (2007) Development of a real-time PCR assay for Trypanosoma cruzi detection in blood samples. Acta Trop 103:195–200. https://doi.org/10.1016/j.actatropica.2007.05.019
Pucek Z (ed) (1981) Keys to vertebrates of Poland. Mammals. PWN Polish Scientific Publishers, Warszawa
Rigó K, Majoros G, Szekeres S, Molnár I, Jablonszky M, Majláth I, Földvári G (2016) Identification of Hepatozoon erhardovae Krampitz, 1964 from bank voles (Myodes glareolus) and fleas in southern Hungary. Parasitol Res 115(6):2409–2413. https://doi.org/10.1007/s00436-016-4992-7
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, New York.
Solano-Gallego L, Sainz Á, Roura X, Estrada-Peña A, Miró G (2016). A review of canine babesiosis: the European perspective. Parasite Vector 9:336 https://doi.org/10.1186/s13071-016-1596-0
Souza SS, Bishop HS, Sprinkle P, Qvarnstrom Y (2016) Comparison of Babesia microti real-time polymerase chain reaction assays for confirmatory diagnosis of babesiosis. Am J Trop Med Hyg 95:1413–1416. https://doi.org/10.4269/ajtmh.16-0406
Smith A, Telfer S, Burthe S, Bennett M, Begon M (2005) Trypanosomes, fleas and field voles: ecological dynamics of a host–vector-parasite interaction. Parasitol 131:355–365. https://doi.org/10.1017/S0031182005007766
StatSoft, Inc. (2004) Statistica (data analysis software system), version 7. www.statsoft.com
Šebek Z (1978) Blood parasites of small mammals in Western Hungary. Parasitologia Hungaria 11:17–22
Turner CMR (1986) Seasonal and age distribution of Babesia, Hepatozoon, Trypanososma and Grahamella species in Clethrionomys glareolus and Apodemus sylvaticus populations. Parasitol 93:279–289. https://doi.org/10.1017/S0031182000051453
Turner CMR, Cox FEG (1985) Interspecific interactions between blood parasites in a wild rodent community. Ann Trop Med Parasitol 79:463–465
Valkiūnas G (2005) Avian malaria parasites and other haemosporidia. CRC Press, Boca Raton, Florida
Valkiūnas G, Iezhova TA, Križanauskienė A, Palinauskas V, Sehgal RNM, Bensch S (2008) A comparative analysis of microscopy and PCR-based detection methods for blood parasites. J Parasitol 94:1395–1401. https://doi.org/10.1645/GE-1570.1
Zábojníková L (2017) Blood parasites of small mammals (Chionomys nivalis, Myodes glareolus) living in montane zone Biele plesá, High Tatras. Oecol Mont 26:41–44
Acknowledgements
We are grateful to Dr. Ravinder Sehgal and Dr. Vaidas Palinauskas and Alan Hendrixson for suggestions to improve the manuscript.
Funding
This research was funded by a grant (No. S-MIP-17-86) from the Research Council of Lithuania.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The research was conducted according to Lithuanian laws. The use of wild mammals for research is regulated by the Republic of Lithuania Law on Wildlife (June 22, 2010, No. XI-920) and the Order of the Director of the State Food and Veterinary Service (June 30, 2011, No. D1–533/B1–310; as last amended on April 24, 2014, No. D1–369/B1–380). According to the current procedure, no permit was required to perform investigations in the current study. Investigated species are murine rodents which are not classified as protected species in accordance with the Republic of Lithuania Law on Protected Animals, Plants and Mushrooms (Order of the Director of the State Food and Veterinary Service: Chapter III, Article 50.2).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Section Editor: Nawal Hijjawi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baltrūnaitė, L., Kitrytė, N. & Križanauskienė, A. Blood parasites (Babesia, Hepatozoon and Trypanosoma) of rodents, Lithuania: part I. Molecular and traditional microscopy approach. Parasitol Res 119, 687–694 (2020). https://doi.org/10.1007/s00436-019-06577-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-019-06577-3