Abstract
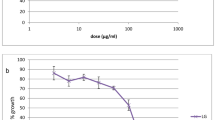
Malaria is one of the most prevalent infectious diseases in the world. Treatment for malaria is commonly inadequate due to the lack of quality assured effective drugs. The effectiveness of these drugs is declining at an ever accelerating rate, with consequent increase in malaria related morbidity and mortality. The newest antiplasmodial drug from plants is needed to overcome this problem. Numerous mangroves and mangal associates are used as folklore medicine to treat various human diseases. The mangrove plant species are a good source of potential bioactive entities which exhibits many therapeutic properties. The present study was carried out to test the antiplasmodial activity of five mangrove plant species distributed along the South East coast of India. Bruguiera cylindrica, Ceriops decandra, Lumnitzera racemosa, Rhizophora apiculata, and Rhizophora mucronata mangrove plant extracts exhibited in vitro antiplasmodial activity against chloroquine-sensitive Plasmodium falciparum. Of which, the ethanolic bark extract of R. mucronata exhibited high antiplasmodial activity (IC50 = 62.18 μg.ml−1). Statistical analysis reveals that, significant antiplasmodial activity (P < 0.05) was observed between the concentrations and time of exposure. The chemical injury to erythrocytes was also carried out and it shows that no morphological differences in erythrocytes by the ethanolic extract of mangrove plants after 48 h of incubation. The screening for phytochemical constituents in the mangrove plants were carried out and it reveals that, the presence of alkaloids, triterpenes, flavonoids, tannins, catachin, anthroquinone, phenols, sugars, and proteins. This study shows that the mangrove plants had a source of lead compounds for the development of new drugs for the treatment of malaria.
Similar content being viewed by others
References
Akalanka D, Somasundaram ST, Manavalan R (2002) Antibacterial activity of leaf and stem bark extract of Rhizophora mucronata Poir. Seshaiyana 10(2):7
Azas N, Laurencin N, Delmas F, Di Giorgio C, Gasquet M, Laget M, Timon David P (2002) Synergistic in vitro antimalarial activity of plant extracts used as traditional herbal remedies in Mali. Parasitol Res 88(2):165–171
Bandaranayake WM (2002) Bioactivities, bioactive compounds and chemical constituents of mangrove plants. Wetlands Ecol Manag 10:421–452
Basak UC, Das AB, Das P (1996) Chlorophyll, carotenoids, proteins and secondary metabolites in leaves of 14 species of mangroves. Bull Mar Sci 58:654–659
Basco LK, Eldin de Pecoulas P, Wilson C, Le Bras J, Mazabraud A (1995) Point mutation in the dihydrofolate reductase gene as the molecular basis for pyrimethamine and cycloguanil resistance in Plasmodium falciparum. Mol Biochem Parasitol 69:135–138
Chenniappan K, Kadarkarai M (2010) In vitro antimalarial activity of traditioanlly used Western Ghats plants from India and their interactions chloroquine against chloroquine- resistant Plasmodium falciparum. Parasitol Res. doi:10.1007/s00436-010-2005-9
Chou FY, Hostettmann Kubo K, Nakamshi K (1977) Isolation of an insect anti-feedent N-methyl flindesine and several Benz [c] phrnantherine alkaloids from East African plants a comment on chelethrine. Heterocyclic 7:969–977
Chung IM, Kim MY, Moon HI (2008) Antiplasmodial activity of sesquiterpene lactone from Carpesium rosulatum in mice. Parasitol Res 103:341–344
Clarkson C, Maharaj VJ, Crouch NR, Grace OM, Pillay P, Matsabisa MG, Bhagwandin N, Smith PJ, Folb PI (2004) In vitro antiplasmodial activity of medicinal plants native to or naturalized in South Africa. J Ethnopharmacol 92:177–191
Cowan MM (1999) Plants products as antimicrobial agents. Clin Microbiol Rev 12:564–582
Durand R, Ramiliarisoa O, Secardin Y, Eldin de Pecoulas P, Basco LK, Le Bras J (1997) DHFR gene point mutation as a predictor of Plasmodium falciparum resistance to cycloguanil in malaria cases from Africa imported to France. Trans R Soc Trop Med Hyg 91:460–461
Gansane A, Sanon S, Ouattara LP, Traore A, Hutter S, Ollivier E, Azas N, Traore AS, Guissou IP, Sirima SB, Nebie I (2010) Antiplasmodial activity and toxicity of crude extracts from alternatives parts of plants widely used for the treatment of malaria in Burkina Faso: contribution for their preservation. Parasitol Res 106:335–340
Hirazumi A, Furusawa E (1999) An immunomodulatory polysaccharide rich substance from the fruit juice of Morinda citrifolio (noni) with antitumour activity. Phytother Res 13:380–387
Hoet S, Opperdoes F, Brun R, Quetin Leclercq J (2004) Natural products active against African trypanosomes: a step towards new drugs. Nat Prod Rep 21:353–364
Jongsuvat Y (1981) Investigation of anticancer from Acanthus illicifolius. MS Thesis. Chulalongkorn University, Bangkok, Thailand
Kathiresan K, Bingham BL (2001) Biology of mangroves and mangrove ecosystems. Adv Mar Biol 40:81–251
Keawpradub N, Kirby GC, Steele JCP, Houghton PJ (1999) Antiplasmodial activity of extracts and alkaloids of three Alstonia species from Thailand. Planta Med 65:690–694
Kepam W (1986) Qualitative organic analysis (Spectrochemical techniques). Ed. II. McGraw Hill, London, pp 40–58
Larson RA (1988) The antioxidants of higher plants. Phytochemical 27:969–978
Le Bras J, Durand R (2003) The mechanisms of resistance to antimalarial drugs in Plasmodium falciparum. Fundam Clin Pharmacol 17:147–153
Lee SJ, Park WH, Moon HI (2009) Bioassay-guided isolation of antiplasmodial anacardic acids derivatives from the whole plants of Viola websteri Hemsl. Parasitol Res 104:463–466
Masuda T, Yonemori S, Oyama Y, Takeda Y, Tanaka T, Andoh T, Shinohara A, Nakata M (1999) Evaluation of the antioxidant activity of environmental plants: activity of the leaf extracts from seashore plants. J Agric Food Chem 47:1749–1754
Moon HI (2007) Antiplasmodial activity of ineupatorolides A from Carpesium rosulatum. Parasitol Res 100:1147–1149
Moore GE, Gerner RE, Frankin HA (1967) Cultures of normal human leukocytes. J Am Med Assoc 199:519–524
Murakami A, Ohigashi H, Koshimizu K (1994) Possible anti-tumor promoting properties of traditional Thai foods and some of their active constituents. Asia Pac J Clin Nutr 3:185–191
Muthuraja M (2009) Effect of antibacterial bioactive compounds from Exoecaria agallocha for the management of fish and poultry diseases. M.Sc. Thesis, Alagappa University, Tamil Nadu, India
Omulokoli E, Khan B, Chhabra SC (1997) Antiplasmodial activity of four Kenyan medicinal plants. J Ethnopharmacol 56:133–137
Otoguro K, Ishiyama A, Kobayashi M, Sekiguchi H, Izuhara T, Sunazuka T, Tomoda H, Yamada H, Omura S (2004) In vitro and in vivo antimalarial activities of a carbohydrate antibiotic, prumycin against drug resistant strains of Plasmodia. J Antibiot 57(6):400–402
Ouattara Y, Sanon S, Traore Y, Mahiou V, Azas N, Sawadogo L (2006) Antimalarial activity of Swartzia madagascariensis desv. (leguminosae), combretum glutinosum guill. and perr. (combretaceae) and Tinospora bakis miers. (menispermaceae), burkina faso medicinal plants. Afr J Tradit Complement Altern Med 3(1):75–81
Padmakumar K (1988) Bioactive substances from marine algae and mangroves. Ph. D. Thesis, Annamalai University, Tamil Nadu, India
Padmakumar K, Ayyakkannu K (1997) Antiviral activity of marine plants. Indian J Virol 13:33–36
Parzy D, Doerig C, Pradines B, Rico A, Fusai T, Doury JC (1997) Proguanil resistance in Plasmodium falciparum African isolates: assessment by mutation-specific polymerase chain reaction and in vitro susceptibility testing. Am J Trop Med Hyg 57:646–650
Premanathan M, Kathiresan K, Chandra K, Bajpai SK (1993) Antiviral activity of marine plants against Newcastle disease virus. Trop Biomed 10:31–33
Premanathan M, Kathiresan K, Chandra K (1994) Antiviral activity of marine and coastal plants from India. Int J Pharm 32(4):330–336
Premanathan M, Kathiresan K, Chandra K (1995) Antiviral evaluation of some marine plants against Semliki forest virus. Int J Pharm 33(1):75–77
Premanathan M, Nakashima H, Kathiresan K, Rajendran N, Yamamoto N (1996) In vitro anti-human immunodeficiency virus activity of mangrove plants. Indian J Med Res 103:278–281
Premanathan M, Arakaki R, Izumi H, Kathiresan K, Nakano M, Yamamoto N, Nakashima H (1999a) Antiviral properties of a mangrove plant, Rhizophora apiculata Blume, against immunodeficiency virus. Antivir Res 44:113–122
Premanathan M, Kathiresan K, Yamamoto N, Nakashima H (1999b) In vitro anti-human immunodeficiency virus activity of polysaccharide from Rhizophora mucronata Poir. Biosci Biotechnol Biochem 63(7):1187–1191
Raja M (2009) Isolation of chemical characterization of bioactive compounds from chosen mangrove plants and their antimicrobial potential against some bacterial pathogens. Ph. D. Thesis, Alagappa University, Tamil Nadu, India
Ramazani A, Sardari S, Zakeri S, Vaziri B (2010) In vitro antiplasmodial and phytochemical study of five Artemisia species from Iran and in vivo activity of two species. Parasitol Res 107:593–599
Rasoanaivo P, Ratsimamanga Urverg S, Ramanitrhasimbola D, Rafatro H, Rakoto Ratsimamanga A (1992) Criblage d’extraits de plantes de Madagascar pour recherche d’activite antipaludique et d’effet potentialisateur de la chloroquine. J Ethnopharmacol 64:117–126
Ravi Kumar S, Ramanathan G, Subhakaran M, Jacob Inbaneson S (2009) Antimicrobial compounds from marine halophytes for silkworm disease treatment. Int J Med Sci 1(5):184–191
Ravikumar S, Ramanathan G, Jacob Inbaneson S, Ramu A (2010) Antiplasmodial activity of two marine polyherbal preparations from Cheatomorpha antennina and Aegiceras corniculatum against Plasmodium falciparum. Parasitol Res. doi:10.1007/s00436-010-2041-5
Sanon S, Azas N, Gasquet M, Ollivier E, Mahiou V, Barro N, Cuzin Ouattara N, Traore AS, Esposito BG, Timon David P (2003) Antiplasmodial activity of alkaloid extracts from Pavetta crassipes (K. Schum) and Acanthospermum hispidum (DC), two plants used in traditional medicine in Burkina Faso. Parasitol Res 90:314–317
Saxena S, Pant N, Jain DC, Bhaluni RS (2003) Antimalarial agents from plant sources. Curr Sci 85:1314–1329
Scalbert A (1991) Antimicrobial properties of tannins. Phytochemical 30:3875–3883
Sherman PW, Billing J (1999) Darwinian gastronomy: why we use spices. Bioscience 49:453–463
Sivaperumal P (2009) Antibacterial sensitivity of active compounds from Exoecaria agallocha against antibiotic resistant human bacterial pathogens. M.Sc. Thesis, Alagappa University, Tamil Nadu, India
Sofowora A (1982) Medicinal plants and traditional medicine in Africa. John Wiley and Sons Ltd. Chichester, United Kingdom
Son IH, Chung IM, Lee SJ, Moon HI (2007) Antiplasmodial activity of novel stilbene derivatives isolated from Parthenocissus tricuspidata from South Korea. Parasitol Res 101:237–241
Subramonia Thangam T, Kathiresan K (1988) Toxic effect of mangrove plant extracts on mosquito larvae Anopheles stephensi L. Curr Sci 57(16):914–915
Subramonia Thangam T, Kathiresan K (1997) Mosquito Larvicidal activity of mangrove plant extracts and synergistic activity of Rhizophora apiculata with pyrethrum against Culex quinquefasciatus. Int J Pharm 35(1):69–71
Trager W (1987) The cultivation of Plasmodium falciparum: applications in basic and applied research in malaria. Ann Trop Med Parasitol 82:511–529
Waako PJ, Katuura E, Smith P, Folb P (2007) East African medicinal plants as a source of lead compounds for the development of new antimalarial drugs. Afr J Ecol 45(1):102–106
Acknowledgments
The authors are thankful to the authorities of Alagappa University for providing required facilities and also to Indian Council of Medical Research, New Delhi for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ravikumar, S., Jacob Inbaneson, S., Suganthi, P. et al. In vitro antiplasmodial activity of ethanolic extracts of mangrove plants from South East coast of India against chloroquine-sensitive Plasmodium falciparum . Parasitol Res 108, 873–878 (2011). https://doi.org/10.1007/s00436-010-2128-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-010-2128-z