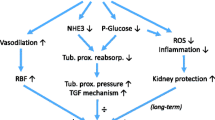
Abstract
One emerging topic in renin–angiotensin system (RAS) research is the direct local control of renin synthesis and release by endogenous metabolic intermediates. During the past few years, our laboratory has characterized the localization and signaling of the novel metabolic receptor GPR91 in the normal and diabetic kidney and established GPR91 as a new, direct link between high glucose and RAS activation in diabetes. GPR91 (also called SUCNR1) binds tricarboxylic acid (TCA) cycle intermediate succinate which can rapidly accumulate in the local tissue environment when energy supply and demand are out of balance. In a variety of physiological and pathological conditions associated with metabolic stress, succinate signaling via GPR91 appears to be an important mediator or modulator of renin secretion. This review summarizes our current knowledge on the control of renin release by molecules of endogenous metabolic pathways with the main focus on succinate/GPR91.

Similar content being viewed by others
References
Aguiar CJ, Andrade VL, Gomes ERM, Alves MNM, Ladeira MS, Pinheiro ACN, Gomes DA, Almeida AP, Goes AM, Resende RR, Guatimosim S, Leite MF (2010) Succinate modulates Ca2+ transient and cardiomyocyte viability through PKA-dependent pathway. Cell Calcium 47(1):37–46
Baumbach L, Leyssac PP, Skinner SL (1976) Studies on renin release from isolated superfused glomeruli: effects of temperature, urea, ouabain and ethacrynic acid. J Physiol 258(1):243–256
Bhuniya D, Umrani D, Dave B, Salunke D, Kukreja G, Gundu J, Naykodi M, Shaikh NS, Shitole P, Kurhade S, De S, Majumdar S, Reddy SB, Tambe S, Shejul Y, Chugh A, Palle VP, Mookhtiar KA, Cully D, Vacca J, Chakravarty PK, Nargund RP, Wright SD, Graziano MP, Singh SB, Roy S, Cai TQ (2011) Discovery of a potent and selective small molecule hGPR91 antagonist. Med Chem Lett 21(12):3596–3602
Castrop H, Höcherl K, Kurtz A, Schweda F, Todorov V, Wagner C (2010) Physiology of kidney renin. Physiol Rev 90(2):607–673
Correa PRAV, Kruglov EA, Thompson M, Leite MF, Dranoff JA, Nathanson MH (2007) Succinate is a paracrine signal for liver damage. J Hepatol 47(2):262–269
Deen PMT, Robben JH (2011) Succinate receptors in the kidney. Clin J Am Soc Nephrol 22(8):1416–1422
Fedotcheva NI, Sokolov AP, Kondrashova MN (2006) Nonezymatic formation of succinate in mitochondria under oxidative stress. Free Radic Biol Med 41(1):56–64
Gilbert RE, Wu LL, Kelly DJ, Cox A, Wilkinson-Berka JL, Johnston CI, Cooper ME (1999) Pathological expression of renin and angiotensin II in the renal tubule after subtotal nephrectomy: implications for the pathogenesis of tubulointerstitial fibrosis. Am J Pathol 155(2):429–440
Goldberg ND, Passonneau JV, Lowry OH (1966) Effects of changes in brain metabolism on the levels of citric acid cycle intermediates. J Biol Chem 241(17):3997–4003
Grobe J, Rahmouni K, Liu X, Sigmund C Metabolic rate regulation by the renin–angiotensin system: brain vs. body. Pflügers Archiv European Journal of Physiology:1-9
Hakak Y, Lehmann-Bruinsma K, Phillips S, Le T, Liaw C, Connolly DT, Behan DP (2009) The role of the GPR91 ligand succinate in hematopoiesis. J Leukoc Biol 85(5):837–843
He W, Miao FJP, Lin DCH, Schwandner RT, Wang Z, Gao J, Chen JL, Tian H, Ling L (2004) Citric acid cycle intermediates as ligands for orphan G-protein-coupled receptors. Nature 429(6988):188–193
Hebert SC (2004) Physiology: orphan detectors of metabolism. Nature 429(6988):143–145
Kang DH, Nakagawa T, Feng L, Watanabe S, Han L, Mazzali M, Truong L, Harris R, Johnson RJ (2002) A role for uric acid in the progression of renal disease. J Am Soc Nephrol 13(12):2888–2897
Kang JJ, Toma I, Sipos A, Bansal E, Peti-Peterdi J (2007) Uric acid acutely triggers renin release and causes glomerular hyperfiltration. FASEB J 21(5):A502
Kang JJ, Toma I, Sipos A, McCulloch F, Peti-Peterdi J (2006) Imaging the renin–angiotensin system: an important target of anti-hypertensive therapy. Adv Drug Deliv Rev 58(7):824–833
Kang JJ, Toma I, Sipos A, McCulloch F, Peti-Peterdi J (2006) Quantitative imaging of basic functions in renal (patho)physiology. Am J Physiol Ren Physiol 291(2):F495–F502
Kang JJ, Toma I, Sipos A, Meer EJ, Vargas SL, Peti-Peterdi J (2008) The collecting duct is the major source of prorenin in diabetes. Hypertension 51(6):1597–1604
Koivunen P, Hirsilä M, Remes AM, Hassinen IE, Kivirikko KI, Myllyharju J (2007) Inhibition of hypoxia-inducible factor (HIF) hydroxylases by citric acid cycle intermediates. J Biol Chem 282(7):4524–4532
Krebs HA (1970) Rate control of the tricarboxylic acid cycle. Adv Enzyme Regul 8:335–353
Mori T, Ogawa S, Cowely AW Jr, Ito S (2012) Role of renal medullary oxidative and/or carbonyl stress in salt-sensitive hypertension and diabetes. Clin Exp Pharmacol Physiol 39(1):125–131
Nangaku M, Eckardt KU (2007) Hypoxia and the HIF system in kidney disease. J Mol Med 85(12):1325–1330
Palm F, Teerlink T, Hansell P (2009) Nitric oxide and kidney oxygenation. Curr Opin Nephrol Hypertens 18(1):68–73
Peti-Peterdi J (2010) High glucose and renin release: the role of succinate and GPR91. Kidney Int 78(12):1214–1217
Peti-Peterdi J, Burford JL, Hackl MJ (2012) The first decade of using multiphoton microscopy for high-power kidney imaging. Am J Physiol Ren Physiol 302:F227–F233
Peti-Peterdi J, Fintha A, Fuson AL, Tousson A, Chow RH (2004) Real-time imaging of renin release in vitro. American Journal of Physiology—Renal. Physiology 287(2):F329–F335
Peti-Peterdi J, Harris RC (2010) Macula densa sensing and signaling mechanisms of renin release. J Am Soc Nephrol 21(7):1093–1096
Peti-Peterdi J, Kang JJ, Toma I (2008) Activation of the renal renin–angiotensin system in diabetes—new concepts. Nephrol Dial Transplant 23(10):3047–3049
Peti-Peterdi J, Toma I, Sipos A, Vargas SL (2009) Multiphoton imaging of renal regulatory mechanisms. Physiology 24(2):88–96
Pluznick JL, Zou DJ, Zhang X, Yan Q, Rodriguez-Gil DJ, Eisner C, Wells E, Greer CA, Wang T, Firestein S, Schnermann J, Caplan MJ (2009) Functional expression of the olfactory signaling system in the kidney. Proc Natl Acad Sci 106(6):2059–2064
Regard JB, Sato IT, Coughlin SR (2008) Anatomical profiling of G protein-coupled receptor expression. Cell 135(3):561–571
Robben JH, Fenton RA, Vargas SL, Schweer H, Peti-Peterdi J, Deen PMT, Milligan G (2009) Localization of the succinate receptor in the distal nephron and its signaling in polarized MDCK cells. Kidney Int 76(12):1258–1267
Rubic T, Lametschwandtner G, Jost S, Hinteregger S, Kund J, Carballido-Perrig N, Schwarzler C, Junt T, Voshol H, Meingassner JG, Mao X, Werner G, Rot A, Carballido JM (2008) Triggering the succinate receptor GPR91 on dendritic cells enhances immunity. Nat Immunol 9(11):1261–1269
Sadagopan N, Li W, Roberds SL, Major T, Preston GM, Yu Y, Tones MA (2007) Circulating succinate is elevated in rodent models of hypertension and metabolic disease. Am J Hypertens 20(11):1209–1215
Sapieha P, Sirinyan M, Hamel D, Zaniolo K, Joyal JS, Cho JH, Honore JC, Kermorvant-Duchemin E, Varma DR, Tremblay S, Leduc M, Rihakova L, Hardy P, Klein WH, Mu X, Mamer O, Lachapelle P, Di Polo A, Beausejour C, Andelfinger G, Mitchell G, Sennlaub F, Chemtob S (2008) The succinate receptor GPR91 in neurons has a major role in retinal angiogenesis. Nat Med 14(10):1067–1076
Schnermann J, Briggs JP (2012) Synthesis and secretion of renin in mice with induced genetic mutations. Kidney Int 81(6):529–538
Schweda F, Friis U, Wagner C, Skott O, Kurtz A (2007) Renin release. Physiology 22(5):310–319
Selak MA, Armour SM, MacKenzie ED, Boulahbel H, Watson DG, Mansfield KD, Pan Y, Simon MC, Thompson CB, Gottlieb E (2005) Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-α prolyl hydroxylase. Cancer Cell 7(1):77–85
Singh P, Blantz RC, Rosenberger C, Gabbai FB, Schoeb TR, Thomson SC (2012) Aberrant tubuloglomerular feedback and HIF-1α confer resistance to ischemia after subtotal nephrectomy. J Am Soc Nephrol 23(3):483–493
Sipos A, Toma I, Kang JJ, Rosivall L, Peti-Peterdi J (2007) Advances in renal (patho)physiology using multiphoton microscopy. Kidney Int 72(10):1188–1191
Tanaka T, Nangaku M (2010) The role of hypoxia, increased oxygen consumption, and hypoxia-inducible factor-1 alpha in progression of chronic kidney disease. Curr Opin Nephrol Hypertens 19(1):43–50
Toma I, Kang JJ, Sipos A, Vargas S, Bansal E, Hanner F, Meer E, Peti-Peterdi J (2008) Succinate receptor GPR91 provides a direct link between high glucose levels and renin release in murine and rabbit kidney. J Clin Inv 118(7):2526–2534
Vargas SL, Toma I, Kang JJ, Meer EJ, Peti-Peterdi J (2009) Activation of the succinate receptor GPR91 in macula densa cells causes renin release. J Am Soc Nephrol 20(5):1002–1011
Weinberg JM, Venkatachalam MA, Roeser NF, Saikumar P, Dong Z, Senter RA, Nissim I (2000) Anaerobic and aerobic pathways for salvage of proximal tubules from hypoxia-induced mitochondrial injury. Am J Physiol Ren Physiol 279(5):F927–F943
Wiesner RJ, Kreutzer U, Rösen P, Grieshaber MK (1988) Subcellular distribution of malate-aspartate cycle intermediates during normoxia and anoxia in the heart. Biochimica et Biophysica Acta (BBA)—Bioenergetics 936 (1):114-123
Wilcox CS (2005) Oxidative stress and nitric oxide deficiency in the kidney: a critical link to hypertension? American Journal of Physiology—Regulatory. Integr Comp Physiol 289(4):R913–R935
Acknowledgments
This work was supported by DK64324 and the 1-11-BS-121 grant from the American Diabetes Association to J.P.P.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published as part of the special issue on the Renin-Angiotensin System.
Rights and permissions
About this article
Cite this article
Peti-Peterdi, J., Gevorgyan, H., Lam, L. et al. Metabolic control of renin secretion. Pflugers Arch - Eur J Physiol 465, 53–58 (2013). https://doi.org/10.1007/s00424-012-1130-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-012-1130-y