Abstract
Objectives
The aim of the present study was to measure nasal mucociliary clearance (NMC) time in the patients with MS and to compare the findings with healthy population.
Methods
Totally 97 individuals including 47 patients with relapsing–remitting multiple sclerosis and 50 healthy volunteers were enrolled into the study. Saccharin clearance test was performed on both groups and NMC time was measured. Data analysis was performed by SPSS version 24.0 statistics program (SPSS Inc., Chicago, Illinois, USA). Statistical tests were interpreted at p < 0.05 significance level.
Results
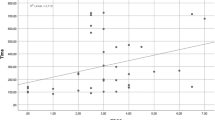
The NMC time averages in MS patients and healthy control group were 12.43 ± 4.05 min and 8.14 ± 2.87 min, respectively; the difference between the groups was significant (p < 0.001). There was a statistically strong association between NMC time values and Expanded Disability Status Scale (EDSS) values in MS patients (r = 0.817, p < 0.001).
Conclusion
We found nasal mucociliary transport time longer in MS patients than healthy population in the present study. To the best of our knowledge, there is not any study conducted about this topic in the literature. We believe that our findings would shed a light on further studies.

Similar content being viewed by others
References
Compston A, Coles A (2008) Multiplesclerosis. Lancet 372(9648):1502–1517. https://doi.org/10.1016/S0140-6736(08)61620-7
Thompson AJ, Baranzini SE, Geurts J et al (2018) Multiple sclerosis. Lancet 391:1622–1636. https://doi.org/10.1016/S0140-6736(18)30481-1
Wijnands JM, Kingwell E, Zhu F, Zhao Y, Fisk JD, Evans C, Marrie RA, Tremlett H (2017) Infection related health care utilization among people with and without multiple sclerosis. Mult Scler 23(11):1506–1516. https://doi.org/10.1177/1352458516681198
Gudis DA, Cohen NA (2010) Ciliadysfunction. Otolaryngol Clin N Am 43:461–472. https://doi.org/10.1016/j.otc.2010.02.007
Andersen I, Camner P, Jensen PL, Philipson K, Proctor DF (1974) A comparison of nasal and tracheobronchial clearance. Arch Environ Health 29(5):290–293
Thompson AJ, Banwell BL, Barkhof F, Carroll WM, Coetzee T, Comi G, Correale J, Fazekas F, Filippi M, Freedman MS (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17(2):162–173. https://doi.org/10.1016/s1474-4422(17)30470-2
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33(11):1444–1446. https://doi.org/10.1212/wnl.33.11.1444
Akcan FA, Dündar Y, Akcan HB, Uluat A, Cebeci D, Ünlü İ (2019) Evaluation of nasal mucociliary clearance time in patients with Vitamin-D deficiency. Eur Arch Otorhinolaryngol 276(4):1075–1080. https://doi.org/10.1007/s00405-019-05286-y
Montgomery S, Hillert J, Bahmanyar S (2013) Hospital admission due to infections in multiple sclerosis patients. Eur J Neurol 20:1153–1160. https://doi.org/10.1111/ene.12130
Nelson RE, Xie Y, DuVall SL, Butler J, Kamauu AW, Knippenberg K, Schuerch M, Foskett N, LaFleur J (2015) Multiple sclerosis and risk of infection-related hospitalization and death in US veterans. Int J MS Care 17:221–230. https://doi.org/10.1111/ene.12130
Sibley WA, Foley JM (1965) Infection and immunization in multiple sclerosis. Ann N Y Acad Sci 122:457–466
Steelman AJ (2015) Infection as an environmental trigger of multiple sclerosis disease exacerbation. Front Immunol 19(6):520. https://doi.org/10.3389/fimmu.2015.00520
Marrodan M, Alessandro L, Farez MF, Correale J (2019) The role of infections in multiplesclerosis. Mult Scler 25(7):891–901. https://doi.org/10.1177/1352458518823940
Andersen O, Lygner PE, Bergstrom T, Andersson M, Vahlne A (1993) Viral infections trigger multiple sclerosis relapses: a prospective seroepidemiological study. J Neurol 240(7):417–422. https://doi.org/10.1007/BF00867354
Kriesel JD, White A, Hayden FG, Spruance SL, Petajan J (2004) Multiple sclerosis attacks are associated with picornavirus infections. Mult Scler 10(2):145–148. https://doi.org/10.1191/1352458504ms1005oa
Correale J, Fiol M, Gilmore W (2006) The risk of relapses in multiple sclerosis during systemic infections. Neurology 67(4):652–659. https://doi.org/10.1212/01.wnl.0000233834.09743.3b
Edwards S, Zvartau M, Clarke H, Irving W, Blumhardt LD (1998) Clinical relapses and disease activity on magnetic resonance imaging associated with viral upper respiratory tract infections in multiple sclerosis. J Neurol Neurosurg Psychiatry 64(6):736–741. https://doi.org/10.1136/jnnp.64.6.736
Winkelmann A, Loebermann M, Reisinger EC, Zettl UK (2014) Multiple sclerosis treatment and infectious issues: update 2013. Clin Exp Neuroimmunol 175:425–438. https://doi.org/10.1111/cei.12226
Stevens WW, Lee RJ, Schleimer RP, Cohen NA (2015) Chronic rhinosinusitis pathogenesis. J Allergy Clin Immunol 136:1442–1453. https://doi.org/10.1016/j.jaci.2015.10.009
Gudis D, Zhao KQ, Cohen NA (2012) Acquired cilia dysfunction in chronic rhinosinusitis. Am J Rhinol Allergy 26:1–6. https://doi.org/10.1016/j.jaci.2015.10.009
Sun SS, Hsieh JF, Tsai SC, Ho YJ, Kao CH (2000) The role of rhinoscintigraphy in the evaluation of nasal mucociliary clearance function in patients with sinusitis. Nucl Med Commun 21:1029–1032
Sadeghi J, Alizadeh N, Ahangar Oskouei M, Laghusi D, Savadi Oskouei D, Nikanfar M, Seyyed Mousavi MN (2019) Frequency of superantigen encoding genes of Staphylococcus aureus isolates collected from multiple sclerosis (MS) patients and nasal carriers. Microb Pathog 127:316–319. https://doi.org/10.1016/j.micpath.2018.12.010
Guan WJ, Peng Y, Zi XX, Tan KS, He TT, Zhong NS, Wang Y (2018) Motile ciliary disorders in chronic airway inflammatory diseases: critical target for interventions. Curr Allergy Asthma Rep 18(9):48. https://doi.org/10.1007/s11882-018-0802-x
Lovato A, Staffieri C, Ottaviano G, Cappellesso R, Giacomelli L, Bartolucci GB, Scapellato ML, Marioni G (2016) Woodworkers and the inflammatory effects of softwood/hardwood dust: evidence from nasal cytology. Eur Arch Otorhinolaryngol 273:3195–3200. https://doi.org/10.1007/s00405-016-3989-2
Staffieri C, Lovato A, Aielli F, Bortoletto M, Giacomelli L, Carrieri M, Romeo S, Boscolo-Rizzo P, Da Mosto MC, Bartolucci GB, Marioni G, Scapellato ML (2015) Investigating nasal cytology as a potential tool for diagnosing occupational rhinitis in woodworkers. Int Forum Allergy Rhinol 5:814–819. https://doi.org/10.1002/alr.21562
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Institutional Review Board of the Yozgat Bozok University.
Informed consent
Written informed consent was obtained from the patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sahin, E., Hamamcı, M. & Kantekin, Y. Measurement of mucociliary clearance in the patients with multiple sclerosis. Eur Arch Otorhinolaryngol 277, 469–473 (2020). https://doi.org/10.1007/s00405-019-05717-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-019-05717-w