Abstract
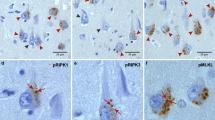
The relation of protein deposition with glial cells and oxidative stress was studied in Creutzfeldt-Jakob disease (CJD), Alzheimer’s disease (AD) and neurologically healthy control patients. Three neocortical areas, the hippocampus, and the cerebellum of 20 CJD, 10 AD and 10 control patients were immunohistochemically examined for the presence of astroglia, microglia, and protein depositions. To investigate the level of oxidative stress the percentage of neurons with cytoplasmic hydroxylated DNA was determined. Astroglia, microglia and oxidative stress were located around amyloid-β depositions and a clear quantitative relation was identified. These markers were only increased in the hippocampus of AD compared to controls. Quantitative analysis in these groups showed a correlation between the oxidative stress level and the number of microglia in the grey matter. All markers were increased in the grey matter and the cerebellum of CJD when compared to AD and controls. The highest numbers of lesions were observed in a CJD population with a rapid disease progression. Quantitative analysis showed a correlation between the oxidative stress level and all glial cells. Further analysis showed that the number of microglia was related to the intensity of the prion depositions. Glial cells in the brain are thought to be the main producers of oxidative stress, resulting in neuronal death. Our results confirm that this close relationship exists in both AD and CJD. We also show that an increased number of glial cells and therefore possibly oxidative stress is associated with the disease progression.





Similar content being viewed by others
References
Anderson I, Adinolfi C, Doctrow S, Huffman K, Joy KA, Malfroy B, et al (2001) Oxidative signalling and inflammatory pathways in Alzheimer’s disease. Biochem Soc Symp 67:141–149
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Braak H, Braak E (1996) Evolution of the neuropathology of Alzheimer’s disease. Acta Neurol Scand 165:3–12
Brown DR, Schmidt B, Kretzschmar HA (1998) A prion protein fragment primes type 1 astrocytes to proliferation signals from microglia. Neurobiol Dis 4:410–422
Colton CA, Chernyshev ON, Gilbert DL, Vitek MP (2000) Microglial contribution to oxidative stress in Alzheimer’s disease. Ann NY Acad Sci 899:292–307
Duyckaerts C, Hauw JJ (1997) Diagnosis and staging of Alzheimer disease. Neurobiol Aging 18 (Suppl 4):S33–42
Kretzschmar HA, Giese A, Brown DR, Herms J, Keller B, Schmidt B, et al (1997) Cell death in prion disease. J Neural Transm Suppl 50:191–210
Pals P, Van Everbroeck B, Sciot R, Godfraind C, Robberecht W, Dom R, et al (1999) A retrospective study of Creutzfeldt-Jakob disease in Belgium. Eur J Epidemiol 15:517–519
Peyrin JM, Lasmezas CI, Haik S, Tagliavini F, Salmona M, Williams A, et al (1999) Microglial cells respond to amyloidogenic PrP peptide by the production of inflammatory cytokines. Neuroreport 10:723–729
Prusiner SB (2001) Shattuck lecture—Neurodegenerative diseases and prions. N Engl J Med 344:1516–1526
Van Everbroeck B, Pals P, et al (2000) Phosphorylated Tau identifies a group of Creutzfeldt-Jakob patients with short disease duration. Brain Pathol 10:C631–607
Van Everbroeck B, Dewulf E, Pals P, Lubke U, Martin JJ, Cras P (2002) The role of cytokines, astrocytes, microglia and apoptosis in Creutzfeldt-Jakob disease. Neurobiol Aging 23:59–64
Van Everbroeck B, Green AJE, Vanmechelen E, Vanderstichele H, Pals P, Sanchez Valle R, et al (2002) Phosphorylated tau in cerebrospinal fluid as a marker for Creutzfeldt-Jakob disease. J Neurol Neurosurg Psychiatry 73:79–81
Vanmechelen E, Vanderstichele H, Davidsson P, Van Kerschaver E, Van Der Perre B, Sjogren M, et al (2000) Quantification of tau phosphorylated at threonine 181 in human cerebrospinal fluid: a sandwich ELISA with a synthetic phosphopeptide for standardization. Neurosci Lett 285:49–52
Veerhuis R, Hoozemans JJ, Janssen I, Boshuizen RS, Langeveld JP, Eikelenboom P (2002) Adult human microglia secrete cytokines when exposed to neurotoxic prion protein peptide: no intermediary role for prostaglandin E2. Brain Res 925:195–203
Voigtlander T, Kloppel S, Birner P, Jarius C, Flicker H, Verghese-Nikolakaki S, et al (2001) Marked increase of neuronal prion protein immunoreactivity in Alzheimer’s disease and human prion diseases. Acta Neuropathol 101:417–423
Walsh DT, Perry VH, Minghetti L (2000) Cyclooxygenase-2 is highly expressed in microglial-like cells in a murine in model of prion disease. Glia 29:392–396
Wegiel J, Wang KC, Tarnawski M, Lach B (2000) Microglia cells are the driving force in fibrillar plaque formation, whereas astrocytes are a leading factor in plague degradation. Acta Neuropathol 100:356–364
Wegiel J, Wang KC, Imaki H, Rubenstein R, Wronska A, Osuchowski M, et al (2001) The role of microglial cells and astrocytes in fibrillar plaque evolution in transgenic APP(SW) mice. Neurobiol Aging 22:49–61
Wong BS, Liu T, Li RL, Pan T, Petersen RB, Smith MA, et al (2001) Increased levels of oxidative stress markers detected in the brains of mice devoid of prion protein. J Neurochem 76:565–572
Zhang J, Perry G, Smith MA, Robertson D, Olson SJ, Graham DG, et al (1999) Parkinson’s disease is associated with oxidative damage to cytoplasmic DNA and RNA in substantia nigra neurons. Am J Pathol 154:1423–1429
Acknowledgements
The authors thank the members of the Belgian CJD surveillance network for referring CJD patients. We also thank Prof. J. Ironside (Edinburgh, UK) and Dr. R. Sanchez-Valle (Barcelona, Spain) for providing tissue slides of CJDhigh patients. The authors further thank the Fund for Scientific Research (FWO) for supporting this research. B.V.E. is a postdoctoral researcher of the Fund for Scientific Research (FWO).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Van Everbroeck, B., Dobbeleir, I., De Waele, M. et al. Extracellular protein deposition correlates with glial activation and oxidative stress in Creutzfeldt-Jakob and Alzheimer’s disease. Acta Neuropathol 108, 194–200 (2004). https://doi.org/10.1007/s00401-004-0879-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-004-0879-2