Abstract
Purpose
Hyperhomocysteinemia is well recognized as an independent risk factor for the development of premature atherosclerosis. Atherosclerosis, however, may be prevented by polyphenols, potent antioxidant compounds with anti-atherogenic properties. Previously, we used cystathionine beta synthase-deficient mice [Cbs (±)] fed a high-methionine diet—a murine model of hyperhomocysteinemia—to show that daily intake of a red wine polyphenolic extract, mainly comprised of catechin and epicatechin, has a beneficial effect on aortic expression of endothelial dysfunction biomarkers and pro-inflammatory cytokines. The aim of the present study was to understand whether catechin and epicatechin, in purified forms, have anti-atherogenic effects in hyperhomocysteinemia.
Methods
Cbs (±) mice received 50 μg of catechin and/or epicatechin daily in drinking water for 1 month. Plasma homocysteine (Hcy) level and aortic expression of several endothelial dysfunction biomarkers (Vcam-1, Icam-1, E-selectin, and Lox-1) and pro-inflammatory cytokines (Tnf-α, Il-6) were assessed.
Results
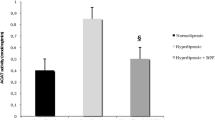
We found that both catechin and epicatechin had a beneficial effect on plasma homocysteine levels and endothelial dysfunction biomarker expression; however, only catechin had a beneficial effect on pro-inflammatory cytokine expression. Further, when both polyphenols were given, a beneficial effect was observed only on pro-inflammatory cytokine expression.
Conclusions
Catechin seems to be a more potent anti-atherogenic compound than epicatechin in hyperhomocysteinemia and should be considered as a novel therapeutic approach against endothelial dysfunction induced by this condition.

Similar content being viewed by others
References
Fortin LJ, Genest J (1995) Measurement of homocyst(e)ine in the prediction of arteriosclerosis. Clin Biochem 28(2):155–162
Weber C, Noels H (2011) Atherosclerosis: current pathogenesis and therapeutic options. Nat Med 17(11):1410–1422
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acid Res 29(9):e45
Welch GN, Loscalzo J (1998) Homocysteine and atherothrombosis. N Engl J Med 338(15):1042–1050
Selhub J (1999) Homocysteine metabolism. Annu Rev Nutr 19:217–246
Refsum H, Nurk E, Smith AD, Ueland PM, Gjesdal CG, Bjelland I, Tverdal A, Tell GS, Nygård O, Vollset SE (2006) The Hordaland homocysteine study: a community-based study of homocysteine, its determinants, and associations with disease. J Nutr 136(6 Suppl):1731S–1740S
Lentz SR (2005) Mechanisms of homocysteine-induced atherothrombosis. J Thromb Haemost 3(8):1646–1654
Noll C, Hamelet J, Matulewicz E, Paul J-L, Delabar J-M, Janel N (2009) Effects of red wine polyphenolic compounds on paraoxonase-1 and lectin-like oxidized low-density lipoprotein receptor-1 in hyperhomocysteinemic mice. J Nutr Biochem 20(8):586–596
Watanabe M, Osada J, Aratani Y, Kluckman K, Reddick R, Malinow MR, Maeda N (1995) Mice deficient in cystathionine beta-synthase: animal models for mild and severe homocyst(e)inemia. Proc Natl Acad Sci USA 92(5):1585–1589
Hamelet J, Demuth K, Dairou J, Ledru A, Paul J-L, Dupret J-M, Delabar J-M, Rodrigues-Lima F, Janel N (2007) Effects of catechin on homocysteine metabolism in hyperhomocysteinemic mice. Biochem Biophys Res Commun 355(1):221–227
Eberhardt RT, Forgione MA, Cap A et al (2000) Endothelial dysfunction in a murine model of mild hyperhomocyst(e)inemia. J Clin Investiga 106(4):483–491
Li D, Liu L, Chen H, Sawamura T, Mehta JL (2003) LOX-1, an oxidized LDL endothelial receptor, induces CD40/CD40L signaling in human coronary artery endothelial cells. Arteriosclero Thrombo Vasc Biol 23(5):816–821
Hammwöhner M, Ittenson A, Dierkes J, Bukowska A, Klein HU, Lendeckel U, Goette A (2007) Platelet expression of CD40/CD40 ligand and its relation to inflammatory markers and adhesion molecules in patients with atrial fibrillation. Exp Biol Med (Maywood) 232(4):581–589
Kassab A, Ajmi T, Issaoui M, Chaeib L, Miled A, Hammami M (2008) Homocysteine enhances LDL fatty acid peroxidation, promoting microalbuminuria in type 2 diabetes. Ann Clin Biochem 45(Pt 5):476–480
Rice-Evans C, Miller N, Paganga G (1997) Antioxidant properties of phenolic compounds. Trends Plant Sci 2(4):152–159
Sies H (2010) Polyphenols and health: update and perspectives. Arch Biochem Biophys 501(1):2–5
Mattaliano MD, Huard C, Cao W, Hill AA, Zhong W, Martinez RV, Harnish DC, Paulsen JE, Shih HH (2009) LOX-1-dependent transcriptional regulation in response to oxidized LDL treatment of human aortic endothelial cells. Am J Physiol Cell Physiol 296(6):C1329–C1337
Scalbert A, Manach C, Morand C, Rémésy C, Jiménez L (2005) Dietary polyphenols and the prevention of diseases. Crit Rev Food Sci Nutr 45(4):287–306
Stoclet J-C, Chataigneau T, Ndiaye M, Oak M-H, El Bedoui J, Chataigneau M, Schini-Kerth VB (2004) Vascular protection by dietary polyphenols. Eur J Pharmacol 500(1–3):299–313
Kaplan M, Aviram M (1999) Oxidized low density lipoprotein: atherogenic and proinflammatory characteristics during macrophage foam cell formation. An inhibitory role for nutritional antioxidants and serum paraoxonase. Clin Chem Lab Med 37(8):777–787
Kerkeni M, Addad F, Chauffert M, Chuniaud L, Miled A, Trivin F, Maaroufi K (2006) Hyperhomocysteinemia, paraoxonase activity and risk of coronary artery disease. Clin Biochem 39(8):821–825
Robert K, Chassé J-F, Santiard-Baron D, Vayssettes C, Chabli A, Aupetit J, Maeda N, Kamoun P, London J, Janel N (2003) Altered gene expression in liver from a murine model of hyperhomocysteinemia. J Biol Chem 278(34):31504–31511
Janel N, Robert K, Chabert C, Ledru A, Gouédard C, Barouki R, Delabar J-M, Chassé J-F (2004) Mouse liver paraoxonase-1 gene expression is downregulated in hyperhomocysteinemia. Thromb Haemost 92(1):221–222
Gouédard C, Barouki R, Morel Y (2004) Dietary polyphenols increase paraoxonase 1 gene expression by an aryl hydrocarbon receptor-dependent mechanism. Mol Cell Biol 24(12):5209–5222
Ou H-C, Song T-Y, Yeh Y-C et al (2010) EGCG protects against oxidized LDL-induced endothelial dysfunction by inhibiting LOX-1-mediated signaling. J Appl Physiol 108(6):1745–1756
Foncea R, Carvajal C, Almarza C, Leighton F (2000) Endothelial cell oxidative stress and signal transduction. Biol Res 33(2):89–96
Terra X, Valls J, Vitrac X et al (2007) Grape-seed procyanidins act as antiinflammatory agents in endotoxin-stimulated RAW 264.7 macrophages by inhibiting NFkB signaling pathway. J Agric Food Chem 55(11):4357–4365
Libermann TA, Baltimore D (1990) Activation of interleukin-6 gene expression through the NF-kappa B transcription factor. Mol Cell Biol 10(5):2327–2334
Raab T, Barron D, Vera FA, Crespy V, Oliveira M, Williamson G (2010) Catechin glucosides: occurrence, synthesis, and stability. J Agric Food Chem 58(4):2138–2149
Donovan JL, Bell JR, Kasim-Karakas S, German JB, Walzem RL, Hansen RJ, Waterhouse AL (1999) Catechin is present as metabolites in human plasma after consumption of red wine. J Nutr 129(9):1662–1668
Kutschera M, Engst W, Blaut M, Braune A (2011) Isolation of catechin-converting human intestinal bacteria. J Appl Microbiol 111(1):165–175
Acknowledgments
This work was supported in part by an EU grant AnEUploïdie and by Viniflhor (Office national interprofessionnel des fruits, des légumes, des vins et de l’horticulture), Ministère de l’Agriculture, programme Vin et Santé, Pathologie et biologie vasculaires. Christophe Noll is supported by a fellowship from the Ministère de l’Enseignement Supérieur et de la Recherche. We thank Dr N. Maeda (Department of Pathology, University of North Carolina, Chapel Hill, NC) for providing heterozygous Cbs mice. We thank C. Imbert, A. Djemat, and A. Kourdouli for technical assistance. We acknowledge the technical platform “Séparation, caractérisation et quantification de biomolécules” (Unité de Biologie Fonctionnelle et Adaptative, Université Paris Diderot-Paris 7, CNRS EAC 4413) for provision of Q-PCR facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noll, C., Lameth, J., Paul, JL. et al. Effect of catechin/epicatechin dietary intake on endothelial dysfunction biomarkers and proinflammatory cytokines in aorta of hyperhomocysteinemic mice. Eur J Nutr 52, 1243–1250 (2013). https://doi.org/10.1007/s00394-012-0435-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-012-0435-0