Abstract
Purpose
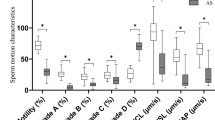
Idiopathic asthenozoospermia is considered as one of the causes of male infertility and characterized by reduced sperm motility. For a better determination of pathogenic mechanism of asthenozoospermia, the exploration of differentially expressed proteins in normal sperm motility and idiopathic asthenozoospermia was conducted in our study.
Methods
Sperm proteins were extracted and isolated by two-dimensional electrophoresis. All significantly changed protein spots were picked up from 2D gels and identified by tandem mass spectrometry. Sixteen of the thirty-three total differentially expressed protein spots were successfully identified by matrix-assisted laser desorption/ionization time-of-flight tandem mass spectrometry.
Results
Sixteen proteins identified belonged to 15 unique protein groups. GRP78, lactoferrin, SPANXB, PGK2, flagellin, DJ-1, XPA binding protein 2, CAB2, GPX4, and GAPDH were the first to be identified as differentially expressed proteins in idiopathic asthenospermia patients. Meanwhile, the analysis of quantitative RT-PCR was carried out to compare the protein levels, and the results indicated that the expression levels of the gene and protein were not entirely consistent.
Conclusions
These experimental results expand the scope of the protein database, generating targets for further investigation of the pathogenic mechanism of idiopathic asthenozoospermia.


Similar content being viewed by others
References
Liu B, Wang P, Wang Z, Jia Y, Niu X, Wang W, Zhang W (2010) Analysis and difference of voltage-dependent anion channel mRNA in ejaculated spermatozoa from normozoospermic fertile donors and infertile patients with idiopathic asthenozoospermia. J Assist Reprod Genet 27:719–724
Shen SL, Luo Y, Ning L, He DL (2007) Differential analysis of two-dimensional gel electrophoresis profiles of spermatozoal protein in human normal semen and idiopathic asthenospermia. Zhonghua Nan Ke Xue 13:50–52
Martinez-Heredia J, de Mateo S, Vidal-Taboada JM, Ballesca JL, Oliva R (2008) Identification of proteomic differences in asthenozoospermic sperm samples. Hum Reprod 23:783–791
Lu JC, Huang YF, Lu NQ (2010) WHO laboratory manual for the examination and processing of human semen: its applicability to andrology laboratories in China. Zhonghua Nan Ke Xue 16:867–871
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
Yan JX, Wait R, Berkelman T, Harry RA, Westbrook JA, Wheeler CH, Dunn MJ (2000) A modified silver staining protocol for visualization of proteins compatible with matrix-assisted laser desorption/ionization and electrospray ionization-mass spectrometry. Electrophoresis 21:3666–3672
Gharahdaghi F, Weinberg CR, Meagher DA, Imai BS, Mische SM (1999) Mass spectrometric identification of proteins from silver-stained polyacrylamide gel: a method for the removal of silver ions to enhance sensitivity. Electrophoresis 20:601–605
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 25:402–408
Yamakawa K, Yoshida K, Nishikawa H, Kato T, Iwamoto T (2007) Comparative analysis of interindividual variations in the seminal plasma proteome of fertile men with identification of potential markers for azoospermia in infertile patients. J Androl 28:858–865
Siva AB, Kameshwari DB, Singh V, Pavani K, Sundaram CS, Rangaraj N, Deenadayal M, Shivaji S (2010) Proteomics-based study on asthenozoospermia: differential expression of proteasome alpha complex. Mol Hum Reprod 16:452–462
Mobberley MA (2010) Electron microscopy in the investigation of asthenozoospermia. Br J Biomed Sci 67:92–100
Linck RW, Albertini DF, Kenney DM, Langevin GL (1982) Tektin filaments: chemically unique filaments of sperm flagellar microtubules. Prog Clin Biol Res 80:127–132
Linck RW, Amos LA, Amos WB (1985) Localization of tektin filaments in microtubules of sea urchin sperm flagella by immunoelectron microscopy. J Cell Biol 100:126–135
Iida H, Honda Y, Matsuyama T, Shibata Y, Inai T (2006) Tektin 4 is located on outer dense fibers, not associated with axonemal tubulins of flagella in rodent spermatozoa. Mol Reprod Dev 73:929–936
Fawcett DW (1975) The mammalian spermatozoon. Dev Biol 44:394–436
Roy A, Lin YN, Agno JE, DeMayo FJ, Matzuk MM (2007) Absence of tektin 4 causes asthenozoospermia and subfertility in male mice. FASEB J 21:1013–1025
Roy A, Lin YN, Agno JE, DeMayo FJ, Matzuk MM (2009) Tektin 3 is required for progressive sperm motility in mice. Mol Reprod Dev 76:453–459
Jin YZ, Bannai S, Dacheux F, Dacheux JL, Okamura N (1997) Direct evidence for the secretion of lactoferrin and its binding to sperm in the porcine epididymis. Mol Reprod Dev 47:490–496
Autiero M, Sansone G, Abrescia P (1991) Relative ratios of lactoferrin, albumin, and acid phosphatase seminal levels as sperm quality markers in fertile and infertile men. J Androl 12:191–200
Sawyer GM, Monzingo AF, Poteet EC, O’Brien DA, Robertus JD (2008) X-ray analysis of phosphoglycerate kinase 2, a sperm-specific isoform from Mus musculus. Proteins 71:1134–1144
Danshina PV, Geyer CB, Dai Q, Goulding EH, Willis WD, Kitto GB, McCarrey JR, Eddy EM, O’Brien DA (2010) Phosphoglycerate kinase 2 (PGK2) is essential for sperm function and male fertility in mice. Biol Reprod 82:136–145
De Maio A (1999) Heat shock proteins: facts, thoughts, and dreams. Shock 11:1–12
Erata GO, Kocak Toker N, Durlanik O, Kadioglu A, Aktan G, Aykac Toker G (2008) The role of heat shock protein 70 (Hsp 70) in male infertility: is it a line of defense against sperm DNA fragmentation? Fertil Steril 90:322–327
Haas IG (1994) BiP (GRP78), an essential hsp70 resident protein in the endoplasmic reticulum. Experientia 50:1012–1020
Delpino A, Castelli M (2002) The 78 kDa glucose-regulated protein (GRP78/BIP) is expressed on the cell membrane, is released into cell culture medium and is also present in human peripheral circulation. Biosci Rep 22:407–420
Lachance C, Bailey JL, Leclerc P (2007) Expression of Hsp60 and Grp78 in the human endometrium and oviduct, and their effect on sperm functions. Hum Reprod 22:2606–2614
Marin-Briggiler CI, Gonzalez-Echeverria MF, Munuce MJ, Ghersevich S, Caille AM, Hellman U, Corrigall VM, Vazquez-Levin MH (2010) Glucose-regulated protein 78 (Grp78/BiP) is secreted by human oviduct epithelial cells and the recombinant protein modulates sperm–zona pellucida binding. Fertil Steril 93:1574–1584
Murphy LC, Tsuyuki D, Myal Y, Shiu RP (1987) Isolation and sequencing of a cDNA clone for a prolactin-inducible protein (PIP). Regulation of PIP gene expression in the human breast cancer cell line, T-47D. J Biol Chem 262:15236–15241
Hassan MI, Waheed A, Yadav S, Singh TP, Ahmad F (2009) Prolactin inducible protein in cancer, fertility and immunoregulation: structure, function and its clinical implications. Cell Mol Life Sci 66:447–459
Witkin SS, Richards JM, Bongiovanni AM, Zelikovsky G (1983) An IgG-Fc binding protein in seminal fluid. Am J Reprod Immunol 3:23–27
Bergamo P, Balestrieri M, Cammarota G, Guardiola J, Abrescia P (1997) CD4-mediated anchoring of the seminal antigen gp17 onto the spermatozoon surface. Hum Immunol 58:30–41
Acknowledgments
The work was supported by National Natural Science Foundation (No. 81260123) and Nature Science Foundation of the Guangxi Zhuang Autonomous Region to Shulin Shen (No. Gui ke zi 0991271).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Shulin Shen and Jinzi Wang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shen, S., Wang, J., Liang, J. et al. Comparative proteomic study between human normal motility sperm and idiopathic asthenozoospermia. World J Urol 31, 1395–1401 (2013). https://doi.org/10.1007/s00345-013-1023-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-013-1023-5