Abstract
Key message
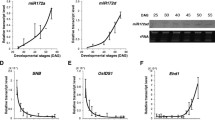
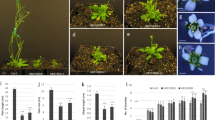
miR394 regulates Arabidopsis flowering time in a LCR-independent manner. Arabidopsis plants harboring mutations in theMIR394 genes exhibit early flowering, lower expression of floral repressor FLC and higher expression of floral integrators FT and SOC1.
Abstract
Plant development occurs throughout its entire life cycle and involves a phase transition between vegetative and reproductive phases, leading to the flowering process, fruit formation and ultimately seed production. It has been shown that the microRNA394 (miR394) regulates the accumulation of the transcript coding for LEAF CURLING RESPONSIVENESS, a member of a family of F-Box proteins. The miR394 pathway regulates several processes including leaf morphology and development of the shoot apical meristem during embryogenesis, as well as having been assigned a role in the response to biotic and abiotic stress in Arabidopsis thaliana and other species. Here, we characterized plants harboring mutations in MIR394 precursor genes and demonstrate that mir394a mir394b double mutants display an early flowering phenotype which correlates with a lower expression of FLOWERING LOCUS C earlier in development and higher expression of the floral integrators FLOWERING LOCUS T and SUPPRESSOR OF OVEREXPRESSION OF CONSTANS 1. Consequently, mutant plants produce fewer branches and exhibit lower seed production. Our work reveals previously unknown developmental aspects regulated by the miR394 pathway, in an LCR-independent manner, contributing to the characterization of the multiple roles of this versatile plant regulatory miRNA.




Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Addo-quaye C, Miller W, Axtell MJ (2009) CleaveLand: a pipeline for using degradome data to find cleaved small RNA targets. Bioinformatics 25:130–131. https://doi.org/10.1093/bioinformatics/btn604
Amasino R (2010) Seasonal and developmental timing of flowering. Plant J 61:1001–1013. https://doi.org/10.1111/j.1365-313X.2010.04148.x
Aukerman MJ, Sakai H (2003) Regulation of flowering time and floral organ identity by a microRNA and its APETALA2-like target genes. Plant Cell 15:2730–2741. https://doi.org/10.1105/tpc.016238
Ausín I, Alonso-Blanco C, Jarillo JA et al (2004) Regulation of flowering time by FVE, a retinoblastoma-associated protein. Nat Genet 36:162–166. https://doi.org/10.1038/ng1295
Chand SK, Nanda S, Joshi RK (2016) Regulation of miR394 in response to Fusarium oxysporum f. sp. cepae (FOC) infection in garlic (Allium sativum L). Front Plant Sci 7:1–12. https://doi.org/10.3389/fpls.2016.00258
Chen L, Hellmann H (2013) Plant E3 ligases: Flexible enzymes in a sessile world. Mol Plant 6:1388–1404. https://doi.org/10.1093/mp/sst005
Chen D, Yan W, Fu L-Y, Kaufmann K (2018) Architecture of gene regulatory networks controlling flower development in Arabidopsis thaliana. Nat Commun 9:4534. https://doi.org/10.1038/s41467-018-06772-3
Cheng J-Z, Zhou Y-P, Lv T-X et al (2017) Research progress on the autonomous flowering time pathway in Arabidopsis. Physiol Mol Biol Plants 23:477–485. https://doi.org/10.1007/s12298-017-0458-3
Cho L-H, Yoon J, An G (2016) The control of flowering time by environmental factors. Plant J tpj.13461:1–12. https://doi.org/10.1111/tpj.13461
Corbesier L, Coupland G (2006) The quest for florigen: a review of recent progress. J Exp Bot 57:3395–3403. https://doi.org/10.1093/jxb/erl095
Deng W, Ying H, Helliwell CA et al (2011) FLOWERING LOCUS C (FLC) regulates development pathways throughout the life cycle of Arabidopsis. Proc Natl Acad Sci USA 108:6680–6685. https://doi.org/10.1073/pnas.1103175108
Dotto M, Gómez MS, Soto MS, Casati P (2018) UV-B radiation delays flowering time through changes in the PRC2 complex activity and miR156 levels in Arabidopsis thaliana. Plant Cell Environ 41:1394–1406. https://doi.org/10.1111/pce.13166
Duncan S, Holm S, Questa J et al (2015) Seasonal shift in timing of vernalization as an adaptation to extreme winter. Elife. https://doi.org/10.7554/eLife.06620
Franco-Zorrilla JM, Valli A, Todesco M et al (2007) Target mimicry provides a new mechanism for regulation of microRNA activity. Nat Genet 39:1033–1037. https://doi.org/10.1038/ng2079
Freytes SN, Canelo M, Cerdán PD (2021) Regulation of flowering time: when and where? Curr Opin Plant Biol 63:102049. https://doi.org/10.1016/j.pbi.2021.102049
Galvão VC, Fankhauser C (2015) Sensing the light environment in plants: photoreceptors and early signaling steps. Curr Opin Neurobiol 34:46–53. https://doi.org/10.1016/j.conb.2015.01.013
German MA, Luo S, Schroth G et al (2009) Construction of parallel analysis of RNA ends (PARE) libraries for the study of cleaved miRNA targets and the RNA degradome. Nat Protoc 4:356–362. https://doi.org/10.1038/nprot.2009.8
Gyula P, Baksa I, Tóth T et al (2018) Ambient temperature regulates the expression of a small set of sRNAs influencing plant development through NF-YA2 and YUC2. Plant Cell Environ 41:2404–2417. https://doi.org/10.1111/pce.13355
Helliwell CA, Wood CC, Robertson M et al (2006) The Arabidopsis FLC protein interacts directly in vivo with SOC1 and FT chromatin and is part of a high-molecular-weight protein complex. Plant J 46:183–192. https://doi.org/10.1111/j.1365-313X.2006.02686.x
Hepworth J, Dean C (2015) Flowering Locus C’s lessons: conserved chromatin switches underpinning developmental timing and adaptation. Plant Physiol 168:1237–1245. https://doi.org/10.1104/pp.15.00496
Huijser P, Schmid M (2011) The control of developmental phase transitions in plants. Development 138:4117–4129. https://doi.org/10.1242/dev.063511
Hyun Y, Richter R, Coupland G (2016) Competence to flower : age-controlled sensitivity to environmental cues. Plant Physiol 173:01523.2016. https://doi.org/10.1104/pp.16.01523
Jia XY, Shen J, Liu H et al (2015) Small tandem target mimic-mediated blockage of microRNA858 induces anthocyanin accumulation in tomato. Planta 242:283–293. https://doi.org/10.1007/s00425-015-2305-5
Jin W, Wu F (2015) Characterization of miRNAs associated with Botrytis cinerea infection of tomato leaves. BMC Plant Biol 15:1–14. https://doi.org/10.1186/s12870-014-0410-4
Jones Rhoades M, Bartel DP (2004) Computational identification of plant microRNAs and their targets, including a stress—induced. miRNA 14:787–799
Jung JH, Seo PJ, Kang SK, Park CM (2011) miR172 signals are incorporated into the miR156 signaling pathway at the SPL3/4/5 genes in Arabidopsis developmental transitions. Plant Mol Biol 76:35–45. https://doi.org/10.1007/s11103-011-9759-z
Jung J-H, Ju Y, Seo PJ et al (2012) The SOC1-SPL module integrates photoperiod and gibberellic acid signals to control flowering time in Arabidopsis. Plant J 69:577–588. https://doi.org/10.1111/j.1365-313X.2011.04813.x
Kim W, Ahn HJ, Chiou T-J, Ahn JH (2011) The role of the miR399-PHO2 module in the regulation of flowering time in response to different ambient temperatures in Arabidopsis thaliana. Mol Cells 32:83–88. https://doi.org/10.1007/s10059-011-1043-1
Klepikova AV, Logacheva MD, Dmitriev SE, Penin AA (2015) RNA-seq analysis of an apical meristem time series reveals a critical point in Arabidopsis thaliana flower initiation. BMC Genom 16:466. https://doi.org/10.1186/s12864-015-1688-9
Knauer S, Holt AL, Rubio-Somoza I et al (2013) A protodermal miR394 signal defines a region of stem cell competence in the Arabidopsis shoot meristem. Dev Cell 24:125–132. https://doi.org/10.1016/j.devcel.2012.12.009
Komeda Y (2004) Genetic regulation of time to flower in Arabidopsis thaliana. Annu Rev Plant Biol 55:521–535. https://doi.org/10.1146/annurev.arplant.55.031903.141644
Lechner E, Achard P, Vansiri A et al (2006) F-Box proteins everywhere. Curr Opin Plant Biol 9:631–638. https://doi.org/10.1016/j.pbi.2006.09.003
Lee W-C, Hou B-H, Hou C-Y et al (2020) Widespread exon junction complex footprints in the RNA Degradome mark mRNA degradation before steady state translation. Plant Cell 32:904–922. https://doi.org/10.1105/tpc.19.00666
Li Y, Fu Y, Ji L et al (2010a) Characterization and expression analysis of the Arabidopsis mir169 family. Plant Sci 178:271–280. https://doi.org/10.1016/j.plantsci.2010.01.007
Li YF, Zheng Y, Addo-Quaye C et al (2010b) Transcriptome-wide identification of microRNA targets in rice. Plant J 62:742–759. https://doi.org/10.1111/j.1365-313X.2010.04187.x
Li F, Pignatta D, Bendix C et al (2012) MicroRNA regulation of plant innate immune receptors. Proc Natl Acad Sci U S A 109:1790–1795. https://doi.org/10.1073/pnas.1118282109
Li L, Li X, Liu Y, Liu H (2016) Flowering responses to light and temperature. Sci China Life Sci 59:403–408. https://doi.org/10.1007/s11427-015-4910-8
Litholdo CG, Parker BL, Eamens AL et al (2016) Proteomic identification of putative microRNA394 target genes in Arabidopsis thaliana identifies Major Latex Protein family members critical for normal development. Mol Cell Proteom 15:2033–2047. https://doi.org/10.1074/mcp.M115.053124
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2^(-ΔΔCT) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Llave C, Xie Z, Kasschau KD, Carrington JC (2002) Cleavage of scarecrow-like mRNA targets directed by a class of Arabidopsis miRNA. Science (80-) 297:2053–2056. https://doi.org/10.1126/science.1076311
Lopez-Vernaza M, Yang S, Müller R et al (2012) Antagonistic roles of SEPALLATA3, FT and FLC genes as targets of the polycomb group gene CURLY LEAF. PLoS ONE 7:e30715-10. https://doi.org/10.1371/journal.pone.0030715
Marquardt S, Boss PK, Hadfield J, Dean C (2006) Additional targets of the Arabidopsis autonomous pathway members, FCA and FY. J Exp Bot 57:3379–3386. https://doi.org/10.1093/jxb/erl073
Michaels SD, Amasino RM (1999) FLOWERING LOCUS C encodes a novel MADS domain protein that acts as a repressor of flowering. Plant Cell 11:949–956. https://doi.org/10.1105/tpc.11.5.949
Michaels SD, Bezerra IC, Amasino RM (2004) FRIGIDA-related genes are required for the winter-annual habit in Arabidopsis. Proc Natl Acad Sci 101:3281–3285. https://doi.org/10.1073/pnas.0306778101
Moon J, Lee H, Kim M, Lee I (2005) Analysis of flowering pathway integrators in Arabidopsis. Plant Cell Physiol 46:292–299. https://doi.org/10.1093/pcp/pci024
Mutasa-Göttgens E, Hedden P (2009) Gibberellin as a factor in floral regulatory networks. J Exp Bot 60:1979–1989. https://doi.org/10.1093/jxb/erp040
Pantaleo V, Szittya G, Moxon S et al (2010) Identification of grapevine microRNAs and their targets using high-throughput sequencing and degradome analysis. Plant J 62:960–976. https://doi.org/10.1111/j.1365-313X.2010.04208.x
Poethig RS (2013) Vegetative phase change and shoot maturation in plants, 1st edn. Elsevier Inc.
Questa J, Song J, Geraldo N et al (2016) Arabidopsis transcriptional repressor VAL1 triggers Polycomb silencing at FLC during vernalization. Science (80-) 353:485–488
Risseeuw E, Venglat P, Xiang D et al (2013) An activated form of UFO alters leaf development and produces ectopic floral and inflorescence meristems. PLoS ONE. https://doi.org/10.1371/journal.pone.0083807
Rubio-Somoza I, Weigel D (2013) Coordination of flower maturation by a regulatory circuit of three microRNAs. PLoS Genet 9:e1003374. https://doi.org/10.1371/journal.pgen.1003374
Rubio-Somoza I, Cuperus JT, Weigel D, Carrington JC (2009) Regulation and functional specialization of small RNA-target nodes during plant development. Curr Opin Plant Biol 12:622–627. https://doi.org/10.1016/j.pbi.2009.07.003
Salomé PA, Bomblies K, Laitinen RAE et al (2011) Genetic architecture of flowering time variation in Arabidopsis thaliana. Genetics 188:421–433
Schwarz S, Grande AV, Bujdoso N et al (2008) The microRNA regulated SBP-box genes SPL9 and SPL15 control shoot maturation in Arabidopsis. Plant Mol Biol 67:183–195. https://doi.org/10.1007/s11103-008-9310-z
Shu K, Yang W (2017) E3 Ubiquitin Ligases: ubiquitous actors in plant development and abiotic stress responses. Plant Cell Physiol 58:1461–1476. https://doi.org/10.1093/pcp/pcx071
Sieber P, Wellmer F, Gheyselinck J et al (2007) Redundancy and specialization among plant microRNAs: role of the MIR164 family in developmental robustness. Development 134:1051–1060. https://doi.org/10.1242/dev.02817
Simpson GG (2004) The autonomous pathway: epigenetic and post-transcriptional gene regulation in the control of Arabidopsis flowering time. Curr Opin Plant Biol 7:570–574. https://doi.org/10.1016/j.pbi.2004.07.002
Song JB, Huang SQ, Dalmay T, Yang ZM (2012) Regulation of leaf morphology by microRNA394 and its target LEAF CURLING RESPONSIVENESS. Plant Cell Physiol 53:1283–1294. https://doi.org/10.1093/pcp/pcs080
Song JB, Gao S, Sun D et al (2013) miR394 and LCR are involved in Arabidopsis salt and drought stress responses in an abscisic acid-dependent manner. BMC Plant Biol 13:210. https://doi.org/10.1186/1471-2229-13-210
Song JB, Shu XX, Shen Q et al (2015) Altered fruit and seed development of transgenic rapeseed (Brassica napus) over-expressing microRNA394. PLoS ONE 10:1–19. https://doi.org/10.1371/journal.pone.0125427
Song JB, Gao S, Wang Y et al (2016) MiR394 and its target gene LCR are involved in cold stress response in Arabidopsis. Plant Gene 5:56–64. https://doi.org/10.1016/j.plgene.2015.12.001
Spanudakis E, Jackson S (2014) The role of microRNAs in the control of flowering time. J Exp Bot 65:365–380. https://doi.org/10.1093/jxb/ert453
Srikanth A, Schmid M (2011) Regulation of flowering time: all roads lead to Rome. Cell Mol Life Sci 68:2013–2037. https://doi.org/10.1007/s00018-011-0673-y
Sundaresan V, Springer P, Volpe T et al (1995) Patterns of gene action in plant development revealed by enhancer trap and gene trap transposable elements. Genes Dev 9:1797–1810. https://doi.org/10.1101/gad.9.14.1797
Takeuchi T, Newton L, Burkhardt A et al (2014) Light and the circadian clock mediate time-specific changes in sensitivity to UV-B stress under light/dark cycles. J Exp Bot 65:6003–6012. https://doi.org/10.1093/jxb/eru339
Taylor MA, Wilczek AM, Roe JL et al (2019) Large-effect flowering time mutations reveal conditionally adaptive paths through fitness landscapes in Arabidopsis thaliana. Proc Natl Acad Sci 116:17890–17899. https://doi.org/10.1073/pnas.1902731116
Teotia S, Tang G (2015) To bloom or not to bloom: role of microRNAs in plant flowering. Mol Plant 8:359–377. https://doi.org/10.1016/j.molp.2014.12.018
Thody J, Folkes L, Medina-Calzada Z et al (2018) PAREsnip2: a tool for high-throughput prediction of small RNA targets from degradome sequencing data using configurable targeting rules. Nucleic Acids Res. https://doi.org/10.1093/nar/gky609
Tian X, Song L, Wang Y et al (2018) miR394 acts as a negative regulator of Arabidopsis resistance to B. cinerea infection by targeting LCR. Front Plant Sci 9:1–11. https://doi.org/10.3389/fpls.2018.00903
Varkonyi-Gasic E, Wu R, Wood M et al (2007) Protocol: a highly sensitive RT-PCR method for detection and quantification of microRNAs. Plant Methods 3:12. https://doi.org/10.1186/1746-4811-3-12
Waheed S, Zeng L (2020) The critical role of miRNAs in regulation of flowering time and flower development. Genes (basel) 11:319. https://doi.org/10.3390/genes11030319
Wang JW (2014) Regulation of flowering time by the miR156-mediated age pathway. J Exp Bot 65:4723–4730. https://doi.org/10.1093/jxb/eru246
Wang JW, Czech B, Weigel D (2009) miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138:738–749. https://doi.org/10.1016/j.cell.2009.06.014
Wu G, Poethig RS (2006) Temporal regulation of shoot development in Arabidopsis thaliana by miR156 and its target SPL3. Development 133:3539–3547. https://doi.org/10.1242/dev.02521
Wu G, Park MY, Conway SR et al (2009) The sequential action of miR156 and miR172 regulates developmental timing in Arabidopsis. Cell 138:750–759. https://doi.org/10.1016/j.cell.2009.06.031
Wu Z, Ietswaart R, Liu F et al (2016) Quantitative regulation of FLC via coordinated transcriptional initiation and elongation. Proc Natl Acad Sci 113:218–223. https://doi.org/10.1073/pnas.1518369112
Wu Z, Fang X, Zhu D, Dean C (2020) Autonomous pathway: FLOWERING LOCUS C repression through an antisense-mediated chromatin-silencing mechanism. Plant Physiol 182:27–37. https://doi.org/10.1104/pp.19.01009
Xu MY, Zhang L, Li WW et al (2014) Stress-induced early flowering is mediated by miR169 in Arabidopsis thaliana. J Exp Bot 65:89–101. https://doi.org/10.1093/jxb/ert353
Xu M, Hu T, Smith MR, Poethig RS (2015) Epigenetic regulation of vegetative phase change in Arabidopsis. Plant Cell 28:TPC2015-00854-RA. https://doi.org/10.1105/tpc.15.00854
Yamaguchi A, Abe M (2012) Regulation of reproductive development by non-coding RNA in Arabidopsis: to flower or not to flower. J Plant Res 125:693–704. https://doi.org/10.1007/s10265-012-0513-7
Yu S, Galvão VC, Zhang Y-C et al (2012) Gibberellin regulates the Arabidopsis floral transition through miR156-targeted SQUAMOSA promoter binding-like transcription factors. Plant Cell 24:3320–3332. https://doi.org/10.1105/tpc.112.101014
Yu C-W, Chang K, Wu K (2016) Genome-wide analysis of gene regulatory networks of the FVE-HDA6-FLD complex in Arabidopsis. Front Plant Sci 7:1–13. https://doi.org/10.3389/fpls.2016.00555
Yumul RE, Kim YJ, Liu X et al (2013) POWERDRESS and diversified expression of the MIR172 gene family bolster the floral stem cell network. PLoS Genet 9:e1003218. https://doi.org/10.1371/journal.pgen.1003218
Zhang J, Zhang H, Srivastava AK et al (2018) Knock-down of rice microRNA166 confers drought resistance by causing leaf rolling and altering stem xylem development. Plant Physiol. https://doi.org/10.1104/pp.17.01432
Zhang Y-Y, Hong Y-H, Liu Y-R et al (2021) Function identification of miR394 in tomato resistance to Phytophthora infestans. Plant Cell Rep 40:1831–1844. https://doi.org/10.1007/s00299-021-02746-w
Zhou C-M, Wang J-W (2013) Regulation of flowering time by microRNAs. J Genet Genom 40:211–215. https://doi.org/10.1016/j.jgg.2012.12.003
Acknowledgements
The authors thank Marja Timmermans (University of Tübingen, Germany) for providing mutant and transgenic lines analyzed in this work. This research was supported by grants from Fondo para la Investigación Científica y Tecnológica (FONCyT), PICT 2015‐0198 and PICT 2018-1090 to M. D.
Funding
This work was supported by grants from Fondo para la Investigación Científica y Tecnológica (FONCyT), PICT 2015‐0198 and PICT 2018–1090 to M. D.
Author information
Authors and Affiliations
Contributions
MD was responsible for study conception and design. Material preparation, data collection and analysis were performed by YB, MAP, FB and AV. The first draft of the manuscript was written by MD and YB, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Andrew Leigh Eamens.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bernardi, Y., Ponso, M.A., Belén, F. et al. MicroRNA miR394 regulates flowering time in Arabidopsis thaliana. Plant Cell Rep 41, 1375–1388 (2022). https://doi.org/10.1007/s00299-022-02863-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-022-02863-0