Abstract
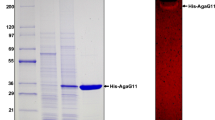
The gene for a novel β-agarase from a deep-sea Microbulbifer-like isolate was cloned and sequenced. It encoded a mature protein of 126,921 Da (1,146 amino acids), which was a modular protein including two tandem carbohydrate-binding module (CBM)-like sequences and a catalytic module. The catalytic module resembled a glycoside hydrolase family 86 β-agarase, AgrA, from Pseudoalteromonas atlantica T6c with 31% amino acid identity. Its recombinant agarase was hyper-produced extracellularly using Bacillus subtilis as the host and purified to homogeneity. The activity and stability were strongly enhanced by CaCl2. The maximal enzyme activity was observed at 45°C and pH 7.5 in the presence of 10 mM CaCl2. The enzyme was an endo-type β-agarase and degraded agarose and agarose oligosaccharides more polymerized than hexamers to yield neoagarohexaose as the main product. This is the first glycoside hydrolase family 86 enzyme to be homogeneously purified and characterized.






Similar content being viewed by others
References
Allouch J, Jam M, Helbert W, Barbeyron T, Kloareg B, Henrissat B Czjzek M (2003) The three-dimensional structures of two β-agarases. J Biol Chem 278:47171–47180
Aoki T, Araki T, Kitamikado M (1990) Purification and characterization of a novel β-agarase from Vibrio sp. AP-2. Eur J Biochem 187:461–465
Araki C (1966) Some recent studies on the polysaccharides of agarophytes. In: Young EG, Maclachan JL (eds) Proceedings international seaweed symposium 5, 1965. Pergamon, London, pp 3–17
Araki T, Lu Z, Morishita T (1998) Optimization of parameters for isolation of protoplasts from Gracilaria verrucosa (Rhodophyta). J Mar Biotechnol 6:193–197
Belas R, Bartlett D, Michael S (1988) Cloning and gene replacement mutagenesis of a Pseudomonas atlantica agarase gene. Appl Environ Microbiol 54:30–37
Birnboim HC, Doly J (1979) A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res 7:1513–1523
Black GW, Rixon JE, Clarke JH, Hazlewood GP, Theodorou MK, Morris P, Gilbert HJ (1996) Evidence that linker sequences and cellulose-binding domains enhance the activity of hemicellulases against complex substrates. Biochem J 319:515–520
Boraston AB, Notenboom V, Warren RA, Kilburn DG, Rose DR, Davies G (2003) Structure and ligand binding of carbohydrate-binding module CsCBM6-3 reveals similarities with fucose-specific lectins and “galactose-binding” domains. J Mol Biol 327:659–669
Chang S, Cohen SN (1979) High frequency transformation of Bacillus subtilis protoplasts by plasmid DNA. Mol Gen Genet 168:111–115
Duckworth M, Yaphe W (1971) Structure of agar. I. Fractionation of a complex mixture of polysaccharides. Carbohydr Res 16:189–197
Ha JC, Kim GT, Kim SK, Oh TK, Yu JH, Kong IS (1997) β-Agarase from Pseudomonas sp. W7: purification of the recombinant enzyme from Escherichia coli and the effects of salt on its activity. Biotechnol Appl Biochem 26:1–6
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166:557–580
Hatada Y, Higaki N, Saito K, Ogawa A, Sawada K, Ozawa T, Hakamada Y, Kobayashi T, Ito S (1999) Cloning and sequencing of a high-alkaline pectate lyase gene from an alkaliphilic Bacillus isolate. Biosci Biotechnol Biochem 63:998–1005
Hatada Y, Saito K, Koike K, Yoshimatsu T, Ozawa T, Kobayashi T, Ito S (2000) Deduced amino-acid sequence and possible catalytic residues of a novel pectate lyase from an alkaliphilic strain of Bacillus. Eur J Biochem 267:2268–2275
Henshaw J, Bolam DN, Pires VM, Czjzek M, Henrissat B, Ferreira LM, Fontes CM, Gilbert HJ (2004) The family 6 carbohydrate-binding module CmCBM6-2 contains two ligand-binding sites with distinct specificities. J Biol Chem 279:21552–21559
Igarashi K, Hatada Y, Hagihara H, Saeki K, Takaiwa M, Uemura T, Ara K, Ozaki K, Kawai S, Kobayashi T, Ito S (1998) Enzymatic properties of a novel liquefying β-amylase from an alkaliphilic Bacillus isolate and entire nucleotide and amino acid sequences. Appl Environ Microbiol 64:3282–3289
Kendall K, Cullum J (1984) Cloning and expression of an extracellular agarase from Streptomyces coelicolor A3(2) in Streptomyces lividans 66. Gene 29:315–321
Kim BJ, Kim HJ, Ha SH, Hwang SH, Byun DS, Lee TH, Kong JY (1999) Purification and characterization of β-agarase from marine bacterium Bacillus cereus ASK202. Biotechnol Lett 21:1011–1015
Kobayashi R, Takisada M, Suzuki T, Kirimura K, Usami S (1997) Neoagarobiose as a novel moisturizer with whitening effect. Biosci Biotechnol Biochem 61:162–163
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Naganuma T, Coury DA, Poline-Fuller M, Gibor A, Horikoshi K (1993) Characterization of agarolytic Microscilla isolates and their extracellular agarases. Systems Appl Microbiol 16:183–190
Ohta Y, Hatada Y, Nogi Y, Miyazaki M, Li Z, Akita M, Hidaka Y, Goda S, Ito S, Horikoshi K (2004a) Enzymatic properties and nucleotide and amino acid sequences of a thermostable β-agarase from a novel species of deep-sea Microbulbifer. Appl Microbiol Biotechnol 64:505–514
Ohta Y, Nogi Y, Miyazaki M, Li Z, Hatada Y, Ito S, Horikoshi K (2004b) Enzymatic properties and nucleotide and amino acid sequences of a thermostable β-agarase from the novel marine isolate, JAMB-A94. Biosci Biotechnol Biochem 68:1073–1081
Potin P, Richard C, Rochas C, Kloareg B (1993) Purification and Alteromonas agarlyticus (Cataldi) comb nov., strain GJ1B. Eur J Biochem 214:599–607
Saito H, Miura K (1963) Preparation of transforming deoxyribonucleic acid by phenol treatment. Biochim Biophys Acta 72:619–629
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
Schroeder DC, Jaffer, MA, Coyne VE (2003) Investigation of the role of a β (1–4) agarase produced by Pseudoalteromonas gracilis B9 in eliciting disease symptoms in the red alga Gracilaria gracilis. Microbiology 149:2919–2929
Sugano Y, Matsumoto T, Kodama H, Noma M (1993a) Cloning and sequencing of agaA, a unique agarase 0107 gene from a marine bacterium, Vibrio sp. strain JT0107. Appl Environ Microbiol 59:3750–3756
Sugano Y, Terada I, Noma M, Matsumoto T (1993b) Purification and characterization of a new agarase from a marine bacterium, Vibrio sp. strain JT0107. Appl Environ Microbiol 59:1549–1554
Sugano Y, Matsumoto T, Noma M (1994) Sequence analysis of the agaB gene encoding a new β-agarase from Vibrio sp. strain JT0107. Biochim Biophys Acta 1218:105–108
Sumitomo N, Ozaki K, Hitomi J, Kawaminami S, Kobayashi T, Kawai S, Ito S (1995) Application of the upstream region of a Bacillus endoglucanase gene to high-level expression of foreign genes in Bacillus subtilis. Biosci Biotechnol Biochem 59:2172–2175
Van der Meulen, HJ, Harder W (1975) Production and characterization of the agarase of Cytoplaga flevensis. Antonie van Leeuwenhoek 41:431–447
Vera J, Alvarez R, Murano E, Slebe JC, Leon O (1998) Identification of a marine agarolytic Pseudoalteromonas isolate and characterization of its extracellular agarase. Appl Environ Microbiol 64:4378–4383
Watanabe T, Kobori K, Miyashita K, Fujii T, Sakai H, Uchida M, Tanaka H (1993) Identification of glutamic acid 204 and aspartic acid 200 in chitinase A1 of Bacillus circulans WL-12 as essential residues for chitinase activity. J Biol Chem 268:18567–18572
Yoshizawa Y, Ametani A, Tsunehiro J, Nomura K, Itoh M, Fukui F, Kaminogawa S (1995) Macrophage stimulation activity of the polysaccharide fraction from a marine alga (Porphyra yezoensis): structure–function relationships and improved solubility. Biosci Biotechnol Biochem 59:1933–1937
Zhong Z, Toukdarian A, Helinski D, Knauf V, Sykes S, Wilkinson JE, O’Bryne C, Shea T, DeLoughery C, Caspi R (2001) Sequence analysis of a 101-kilobase plasmid required for agar degradation by a Microscilla isolate. Appl Environ Microbiol 67:5771–5779
Acknowledgements
We are grateful to Dr. William D. Grant of the University of Leicester and Dr. Y. Sakano of the Tokyo University of Agriculture and Technology for stimulating discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ohta, Y., Hatada, Y., Nogi, Y. et al. Cloning, expression, and characterization of a glycoside hydrolase family 86 β-agarase from a deep-sea Microbulbifer-like isolate. Appl Microbiol Biotechnol 66, 266–275 (2004). https://doi.org/10.1007/s00253-004-1757-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-004-1757-5